Fluorine F Highly toxic refrigerants gas Basic Information

Fluorine (F) Highly toxic, refrigerants gas


Basic Information Name: Fluorine Symbol: F Atomic Number: 9 Atomic Mass: 18. 998 amu Electron Configuration: 2, 7 § Boiling Point: -188. 14 C § Melting Point: -219. 62 C § Diatomic element § § §

Facts § Derived from the Latin word “fleure”, which means flow § Discovery: In 1886, by Henri Moissan § Obtained from: Mineral fluorite

Description § Greenish pale yellow acidic gas § Most reactive of the non-metal → combined with most other elements § 13 th most common element in the crust

Uses § Helps other materials to burn → Rocket fuels §Alone it is poisonous → used in fluorine compound → nuclear industry § Toothpaste → Average human body contains hundredth of ounce of fluorine § Damage ozone layer

Bromine (Br) volatile, mobile, dangerous reddishbrown liquid


Basic Information Name: Bromine Symbol: Br Atomic Number: 35 Atomic Mass: 79. 904 amu Electron Configuration: 2, 8, 18, 7 § Boiling Point: 58. 78 C § Melting Point: -7. 2 C § Diatomic element § § §

Facts § Derived from the Greek word “bromos”, which means stench § Discovery: In 1826, by Antoine J. Balard from SALT WATER § Poisonous § Obtained from: SEA WATER

Description § A reddish-brown liquid § The only non-metal that is a liquid at normal room conditions. § Causes painful burns

Uses § Very poisonous → Pesticides § Photographic film § Malfunctioning of nervous system → Disturbances in genetic materials § Organic Bromine can cause damage to organs → Can even cause cancer !!!!

Iodine (I) bluish-black, lustrous solid
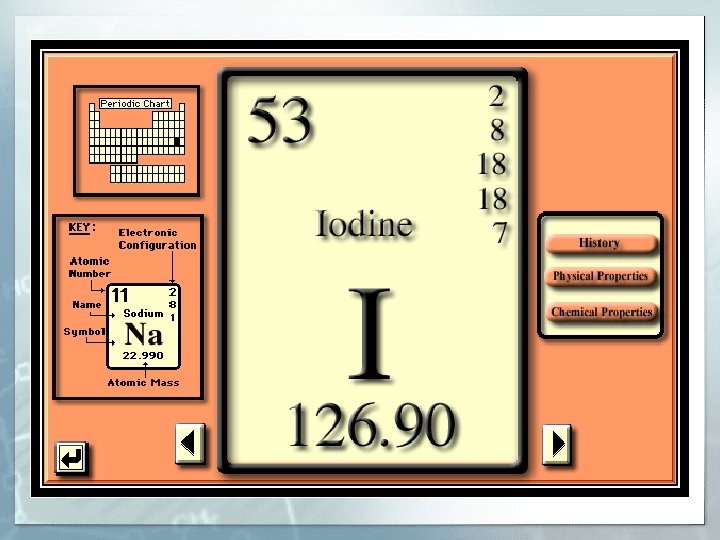

Basic Information Name: Iodine Symbol: I Atomic number: 53 Atomic mass: 126. 90 amu Electron configuration: 2, 8, 18, 7 § Boiling Point: 183 C § Melting Point: 113. 7 C § Diatomic element § § §

Facts § Derived from the Greek word iôdes, which means “violet” § Discovery: In 1811, by Bernald Courtois from SULFURIC ACID § Required in humans § Obtained from: sodium and potassium compounds

Description § When solid: looks metallic, grayish-black or violet-dark gray color. § As a gas: violet color, with a harsh, unpleasant odor. § Sublimation → Never exists as a liquid

Uses → First Aid

Iodine for body function § A naturally occurring mineral for your body to function normally. § Iodine Body Functions: Heat and Energy production Metabolism of fat Physical and Mental development Production of Thyroid hormone

Iodine Deficiency § Iodine Deficiency Disorders § For FOOD SOURCES high in IODINE try: - Seafood - Kelp - Iodized Salt

Bibliography § http: //www. chemicalelements. com/elements/br. h tml § http: //www. carondelet. pvt. k 12. ca. us/Family/Scie nce/Halogens/bromine. htm § www. webelements. com/webelements/text/I/key. html § http: //www. lm. liverpool. k 12. ny. us/Hyper. Chart/ch emicalprops/bromine. html
- Slides: 21