Fluids Behavior of Liquids and Gases 14 2

Fluids – Behavior of Liquids and Gases 14. 2 and 14. 3

Pressure • is the amount of force exerted per unit of area, or P = F/A. • measured in a unit called Pascal (Pa)

Fluid – anything that flows – gas or liquid
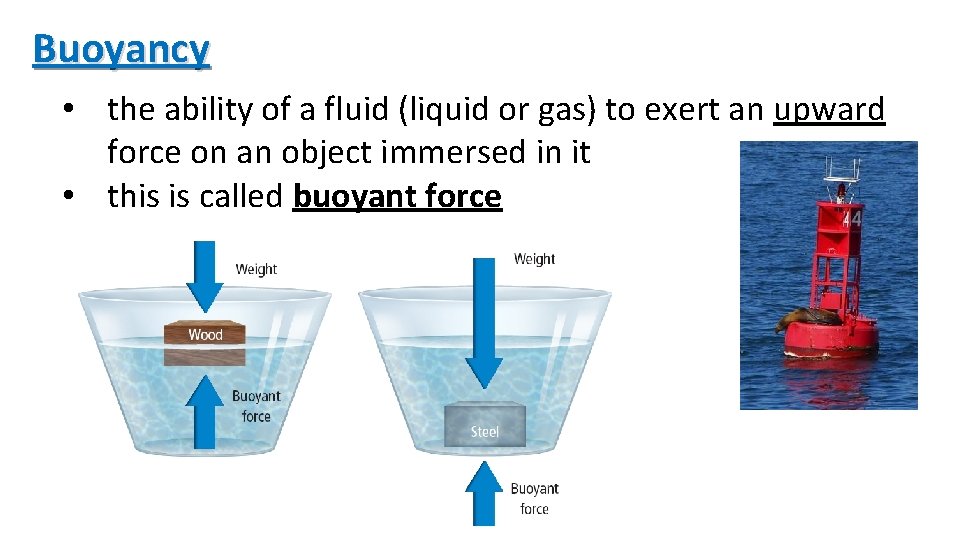
Buoyancy • the ability of a fluid (liquid or gas) to exert an upward force on an object immersed in it • this is called buoyant force
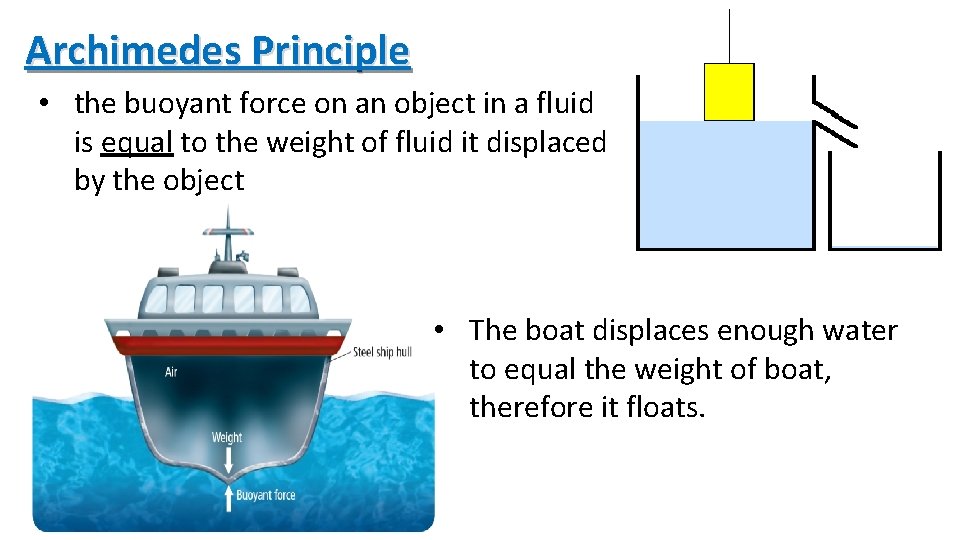
Archimedes Principle • the buoyant force on an object in a fluid is equal to the weight of fluid it displaced by the object • The boat displaces enough water to equal the weight of boat, therefore it floats.
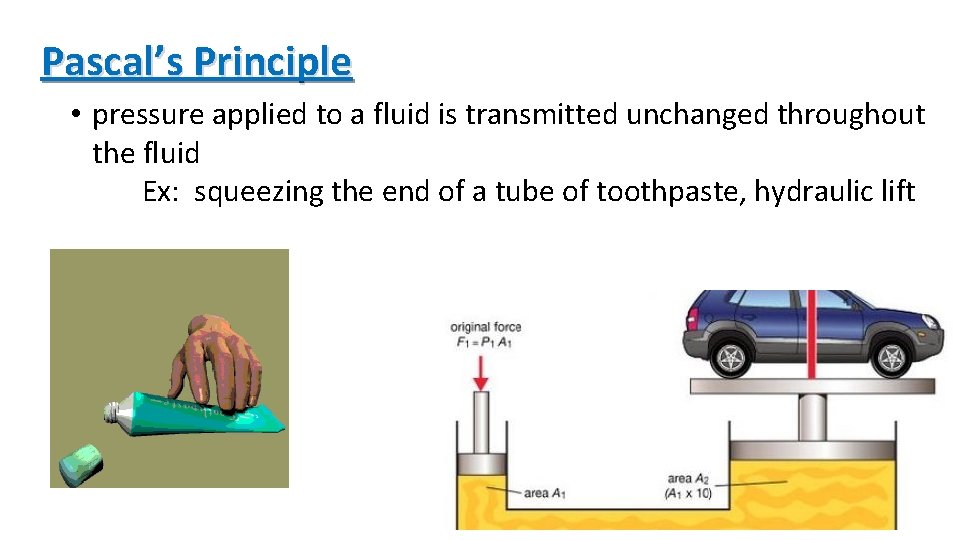
Pascal’s Principle • pressure applied to a fluid is transmitted unchanged throughout the fluid Ex: squeezing the end of a tube of toothpaste, hydraulic lift
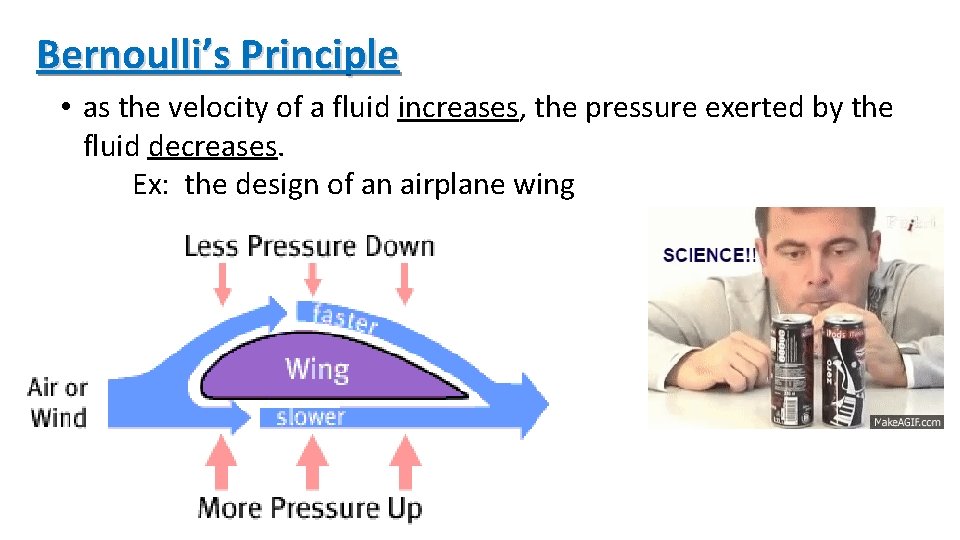
Bernoulli’s Principle • as the velocity of a fluid increases, the pressure exerted by the fluid decreases. Ex: the design of an airplane wing

Boyle’s Law • as the volume of gas decreases, the pressure of a gas increases, provided the temperature does not change. Ex: weather balloon https: //upload. wikimedia. org/wikipedia/commons/6/65/Boyle %27 s_Law_Demonstrations. webm
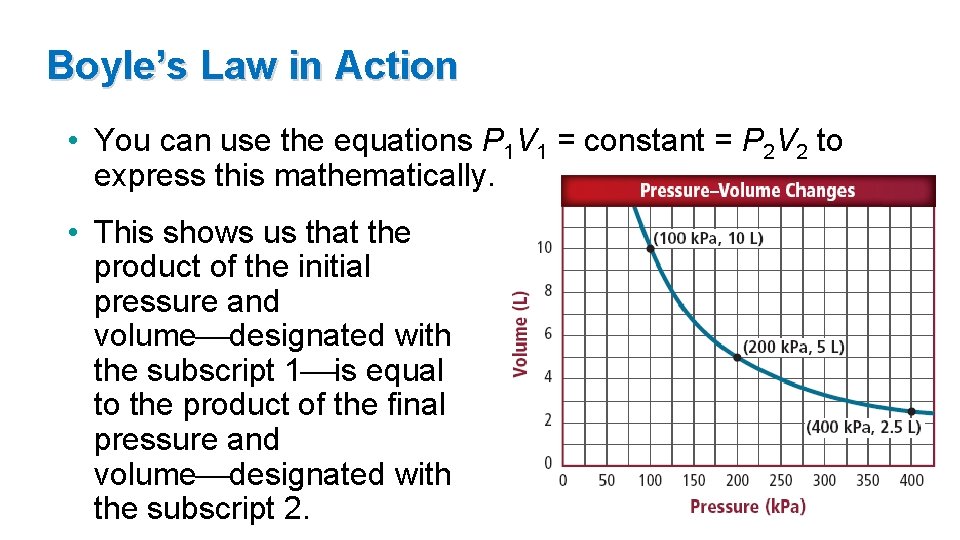
Boyle’s Law in Action • You can use the equations P 1 V 1 = constant = P 2 V 2 to express this mathematically. • This shows us that the product of the initial pressure and volume designated with the subscript 1 is equal to the product of the final pressure and volume designated with the subscript 2.

Charles’s Law • as temperature increases, the volume increases, provided pressure remains constant Ex: Hot air balloon

Using Charles’s Law • The formula that relates the variables of temperature to volume shows a direct relationship when temperature is given in Kelvin. initial volume • When using Charles’s law, the pressure must be kept constant.
In your notebook answer: Page 446 questions 13 – 17 Page 451 questions 24 – 26

Using Charles’s Law What would be the resulting volume of a 2. 0 -L balloon at 20. 0 C that was placed in a container of ice water at 3. 0 C?

Using Charles’s Law • As Charles’s law predicts, the volume decreased as the temperature of the trapped gas decreased.

Pressure – Temperature Relationship • as temperature increases, the pressure increases, provided the volume does not change Ex: popping popcorn
- Slides: 15