Fluidliquid equilibrium LIMITED MUTUAL SOLUBILITY of LIQUIDS All

Fluid-liquid equilibrium LIMITED MUTUAL SOLUBILITY of LIQUIDS All liquids in some degree are soluble in each other According to mutual degree of solubility liquids can be divided into several groups: 1. Liquids infinitely soluble in one another: methanol – water, benzene – chloroform and others

LIMITED MUTUAL SOLUBILITY of LIQUIDS 2. Liquids hardly soluble in each other: benzene – water, mercury – water and others 3. Liquids limitedly mutually soluble phenol – water, oil – water, methanol – hexane and others

LIMITED MUTUAL SOLUBILITY of LIQUIDS All kinds of mutual solubility may take place in the systems which occur during oil and gas extraction

LIMITED MUTUAL SOLUBILITY of LIQUIDS Mutual limited solubility of liquids, except of their nature, strongly depends on the temperature This influence can be different Therefore, there are three types of diagrams temperature – composition

LIMITED MUTUAL SOLUBILITY of LIQUIDS In the first case, the mutual solubility with increasing temperature increases and at a certain temperature the liquids shows unlimited solubility in each other Temperature above which liquid is unlimited soluble in each other in any ratio, called upper critical temperature of dissolution
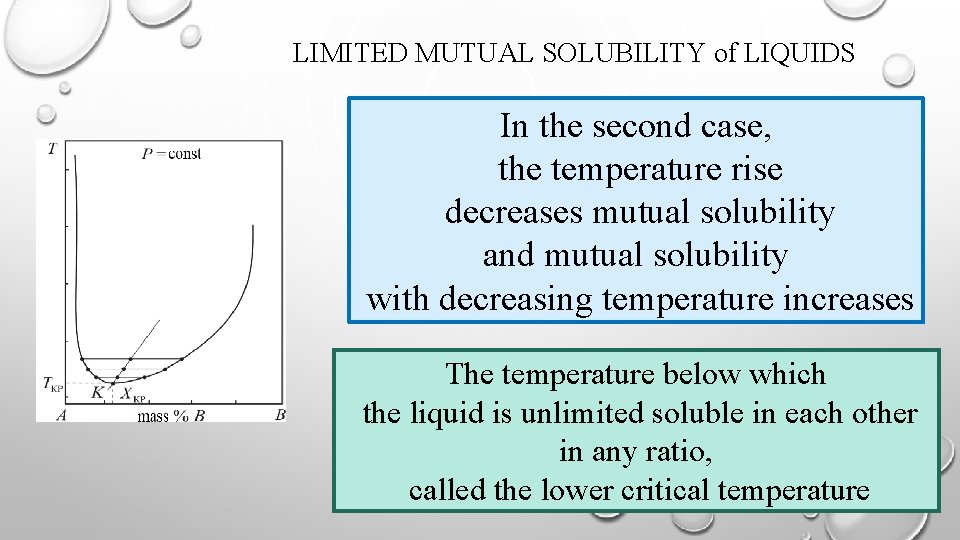
LIMITED MUTUAL SOLUBILITY of LIQUIDS In the second case, the temperature rise decreases mutual solubility and mutual solubility with decreasing temperature increases The temperature below which the liquid is unlimited soluble in each other in any ratio, called the lower critical temperature

LIMITED MUTUAL SOLUBILITY of LIQUIDS There also mixtures which have a critical temperature of the two type – top and bottom. This systems: nicotine – water, glycerol – m-toluidine, etc.

LIMITED MUTUAL SOLUBILITY of LIQUIDS Let us consider the diagram of limited solubility in detail The curve in the state diagram is the set of points that define the compositions of saturated solutions at appropriate temperatures, i. e. points at which the system goes from heterogeneous to homogeneous state or vice versa

LIMITED MUTUAL SOLUBILITY of LIQUIDS Areas of limited solubility diagrams that are outside of the of layering curve correspond to a single-phase (homogeneous) system. In areas which are within the of layering curve, a heterogeneous system consisting of two layers of saturated solutions of each other substances

LIMITED MUTUAL SOLUBILITY of LIQUIDS The point, which corresponds to the critical temperature, determined by the Alekseev’s method, according to which a straight line passing through the middle of the lines (connodes) that connect the associated phase intersects the curve of the layering at the critical point.
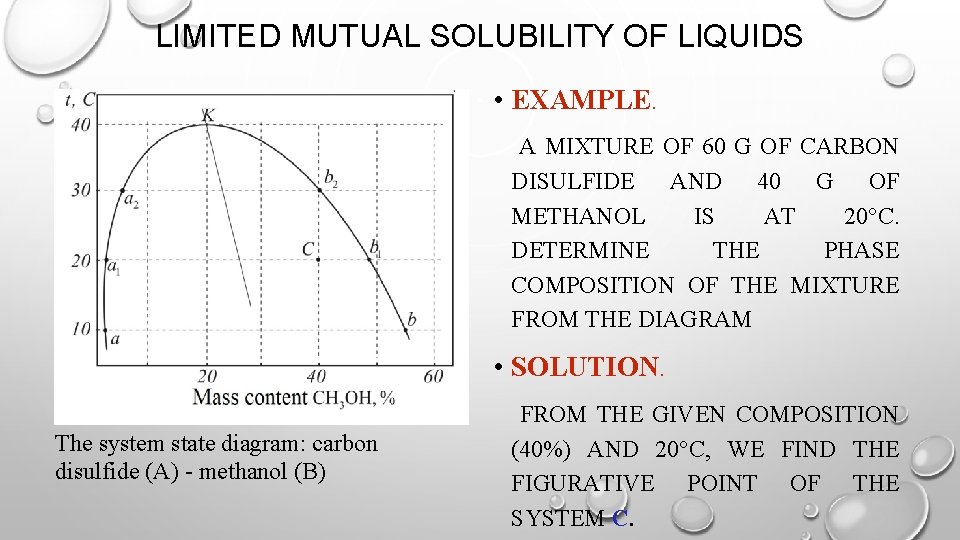
LIMITED MUTUAL SOLUBILITY OF LIQUIDS • EXAMPLE. A MIXTURE OF 60 G OF CARBON DISULFIDE AND 40 G OF METHANOL IS AT 20°C. DETERMINE THE PHASE COMPOSITION OF THE MIXTURE FROM THE DIAGRAM • SOLUTION. The system state diagram: carbon disulfide (A) - methanol (B) FROM THE GIVEN COMPOSITION (40%) AND 20°C, WE FIND THE FIGURATIVE POINT OF THE SYSTEM С.
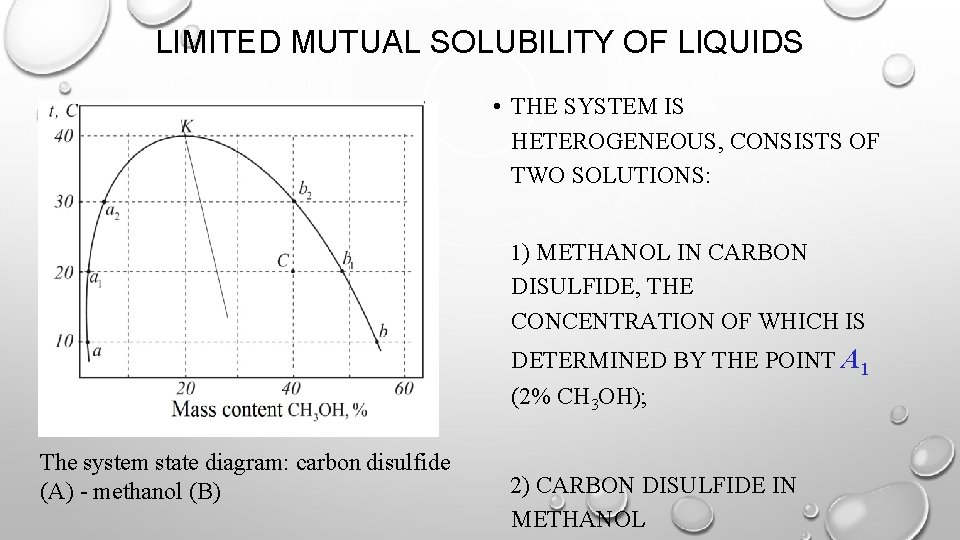
LIMITED MUTUAL SOLUBILITY OF LIQUIDS • THE SYSTEM IS HETEROGENEOUS, CONSISTS OF TWO SOLUTIONS: 1) METHANOL IN CARBON DISULFIDE, THE CONCENTRATION OF WHICH IS DETERMINED BY THE POINT A 1 (2% CH 3 OH); The system state diagram: carbon disulfide (A) - methanol (B) 2) CARBON DISULFIDE IN METHANOL

LIMITED MUTUAL SOLUBILITY OF LIQUIDS • THE NUMBER OF SOLUTIONS CAN BE DETERMINED GRAPHICALLY ACCORDING TO THE RULE OF THE LEVER: = , MI + MII = 100 MII = 4, 74 MI MI + 4, 74 MI = 100 The system state diagram: carbon disulfide (A) methanol (B) • THEREFORE, THE AMOUNT OF THE FIRST SOLUTION: 17, 4 G, AND THE AMOUNT OF THE SECOND : 100 – 17, 4 = 82, 6 G.

Thank you for attention
- Slides: 14