Fluid management Electrolyte Disturbances By Dr Wael Alham


Fluid management& Electrolyte Disturbances: By Dr. Wael Alham Mohamed lecturer of Anesthesia & ICU Sohag Faculty of Medicine

Fluid Management • Body fluid compartments (Average 70 KG ) Compartm Body ent weight(%) Volume (L) Intracell. 25 36 Extracell. Interstitial 19 Intravascul 5 Total 60 13. 5 42
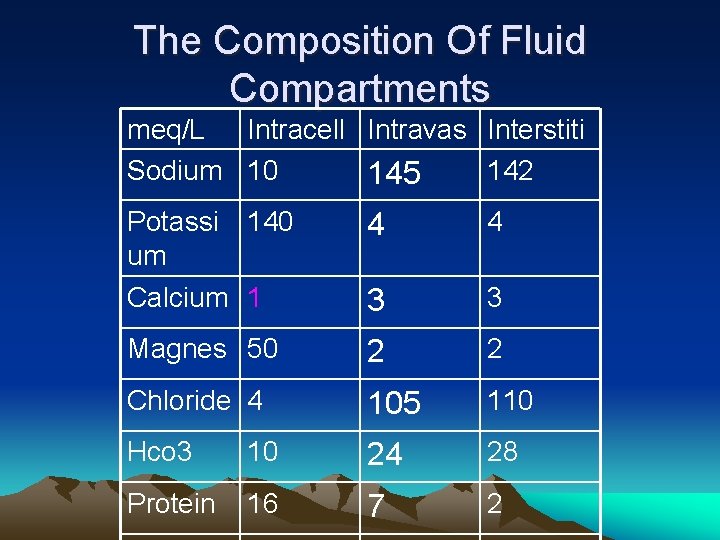
The Composition Of Fluid Compartments meq/L Intracell Intravas Interstiti Sodium 10 142 145 Potassi um 140 4 4 Calcium 1 3 3 Magnes 50 2 2 Chloride 4 105 110 Hco 3 10 24 28 Protein 16 7 2

Intracellular Fluid • *A membrane bound ATP-dependant pump exchange Na for K in a ratio 3: 2 • *Cell membrane are relatively impermeable to Na and to lesser extent to K , So K is the most important determinant of intracellular osmolality • *The impermeability of cell membrane to most proteins results in high intracellular protein concentration.

Interstitial Fluid • *Most interstitial water is in chemical association with proteoglycans forming gel * Interstitial Fluid pressure is negative (about -5 mm. Hg), as the fluid volume increase the pressure rises and become positive, free fluid increase and appear clinically as edema • *The protein content is low (2 g/L)

Intravascular Fluid **Most Its commonly referred to as plasma. electrolyte pass freely between plasma and interstitium resulting in nearly identical electrolyte composition. *Plasma proteins are the only osmotically active solute in fluid exchange between plasma and interstitium. *The interstitial compartment acts as an overflow reservoir for the intravascular compartment.
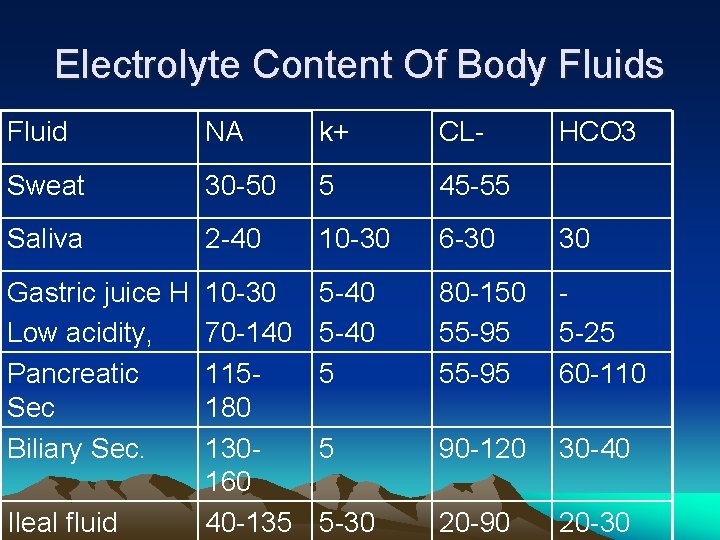
Electrolyte Content Of Body Fluids Fluid NA k+ CL- Sweat 30 -50 5 45 -55 Saliva 2 -40 10 -30 6 -30 30 Gastric juice H Low acidity, Pancreatic Sec Biliary Sec. 10 -30 70 -140 115180 130160 40 -135 5 -40 5 80 -150 55 -95 5 -25 60 -110 5 90 -120 30 -40 5 -30 20 -90 20 -30 Ileal fluid HCO 3

Replacement Type Solutions : • Normal Saline (Na 154 m. Eq/L, Cl 154 m. Eq/L) • It’s the preferred solution in hypochloremic M alkalosis • Its used for diluting packed RBCs • If its given in large amount it cause hyper chlormic M Acidosis • 3%Normal Saline : each Na and Cl 512 m. Eq/L) • Its used for ttt of symptomatic hypo natremia & hypovolemic shock • 3 -7. 5% Saline solutions can be used in hypovolemic shock (Slowly through CV catheter as it may cause hemolysis
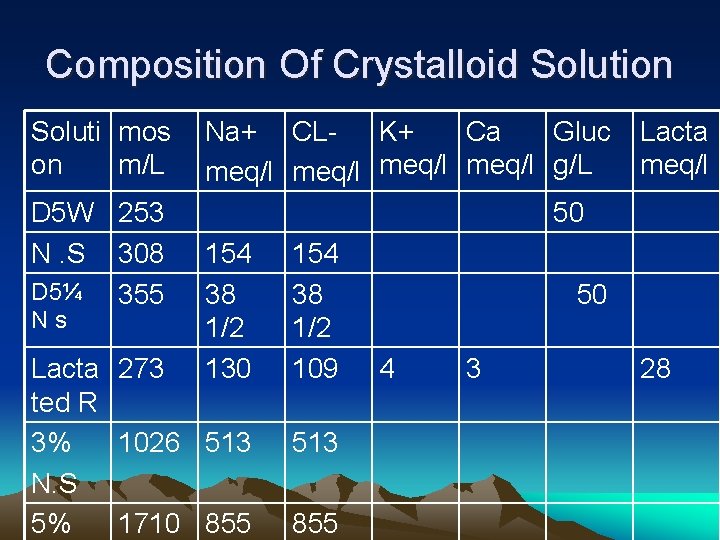
Composition Of Crystalloid Solution Soluti mos on m/L D 5 W 253 N. S 308 D 5¼ 355 Ns Na+ CL- K+ Ca Gluc meq/l g/L 50 154 38 38 50 1/2 130 109 4 3 Lacta 273 ted R 3% 1026 513 N. S 5% 1710 855 513 855 Lacta meq/l 28

Crystalloid Solutions A) Maintenance Type Solutions: Its hypotonic solutions used in absence of oral intake as fluid and electrolyte deficit can rapidly develop due to urine formation , G I T secretions , sweating and insensible loss from skin and respiratory tract. -Solutions such as D 51/4 N. S and D 51/2 N. S are commonly used because these loses are normally hypotonic (more water loss than Na loss)

Dextrose 5% • *It contain 50 gm/L of glucose , its hypotonic (253 mosm/L) • *Its used to prevent ketosis and hypoglycemia due to fasting • -For replacement of pure water deficit • -as a maintenance fluid for patient on Na restriction • -Its content of water pass freely through BBB , So volume overload from dextrose can lead to brain edema.

Maintenance fluid requirement Weight Rate For the first 10 kg 4 ml/kg For the next 1020 kg 2 ml/kg For each kg above 20 1 ml/kg

(2)Replacement Fluid Its used to replace : A) Pre existing deficit : according to the duration of the fast , estimated by: Normal maintenance rate x Length of fast (For 70 kg man fasting 8 hours= 40+20+50 ml/h x 8 hours =880 ml The fluid used should be similar to the fluid lost e. g. hypotonic solution D 5 ¼ NS or D 5 ½NS

Replacement Fluid B) Abnormal fluid losses as: -Pre operative bleeding , vomiting , diuresis or diarrhea -Occult losses due to fluid sequestration by traumatized or infected tissues or ascites -Increased insensible losses due to hyper ventilation , fever and sweating Ideally, all deficits should be replaced preoperatively in all patients by fluids similar to that lost

Surgical Fluid Losses • Blood Loss : • -Measurement in surgical suction container • -Blood on surgical sponges and laparotomy pads (fully soaked sponge 4 x 4 hold about 10 ml blood and a soaked pad holds 100 -150 ml • -Serial hematocrite or hemoglobin concentrations reflect the ratio of blood cells to plasma (Rapid fluid shift and fluid replacement affect measurements

Replacing Blood Loss • Ideally, blood loss should be replaced with crystalloid or colloid solutions to maintain normovolemia until the danger of anemia outweigh the risks of transfusion. • -At that point , further blood loss is replaced with RBCs to maintain Hb concent. or hematocrite at that level. • This point corresponds to 7 -10 gm Hb/dl, or hematocrit of 21 -30%.

Replacing Blood Loss • -We give lactated ringer 3 -4 times the volume lost or colloid in a 1: 1 ratio until transfusion point is reached, then blood is replaced unit for unit as its lost either with whole blood or reconstituted packed RBCs. • -One unit of RBCs will increase hemoglobin 1 g/L or hematocrite 2 -3% in adult and 10 ml/kg of RBCs will increase hemoglobin 3 g/L and hematocrite by 10%

Other Fluid Losses • Evaporative fluid losses are apparent with large wound and directly proportionate to surface area exposed and duration of surgery • Internal redistribution (third spacing): • Surgical dissections, inflamed, infected or traumatized tissue can sequester large amount of fluid in its interstitial space, across serosal surface (ascites), or into bowel lumen , which become non functioning as it does not equilibrate with the rest of extracellular compartments.

Replacing Redistributive & Evaporative Losses • Procedures can be classified according the degree of tissue trauma : Degree of tissue trauma Fluid requirement Minimal (e. g. , 0 -2 m L /kg/h herniorrhapy ) Moderate (e. g. 2 -4 m. L/kg/h , cholycystectomy) Severe (e. g. , bowel resection) 4 -8 ml/kg/h

Replacement Type Solutions • *Ringer lactate solution. Its the most physiologic solution , and it has the least effect on extra cellular fluid. • *Normal Saline. Its the preferred solution for hypochloremic metabolic acidosis and for dilution packed RBCs prior to transfusion , when its given in large amount , it produce dilutional hyperchloremic acidosis, as plasma Hco 3 decrease as Cl- concent. Increase. • *Hypertonic 3% saline is used for severe symptomatic hyponatremia&hypovolemic shock

Potassium Balance Daily Requirements • 1 meq/kg/day • 1 meq of K+ per inch of banana • If the average person weighs 70 kg then to fulfill your necessary daily requirements you need to eat a 6 feet banana

Disorders Of Potassium Balance • Intracellular K is 140 meq / L • Extra cellular K is 4 meq/ L (3. 5 -5. 5 meq/L), reflecting the balance between K intake and excretion • Extra cellular K is regulated by : 1 - Cell membrane Na-K-ATPase activity. 2 -Serum K determine its urinary excretion

Inter Compartmental shift of K • 1 - PH acidosis increase serum K Alkalosis decrease serum K ( Kchanges 0. 6 meq/l per 0. 1 unit change in arterial PH) 2 - Circulating Insulin decrease serum K 3 -Sympathetic stimulation alpha stim. increase serum K B 2 stim. Decrease serum K

Inter Compartmental shift of K 4 -Hypothermia dec. serum K due to cellular uptake , Re warming reverse this shift 5 - Plasma osmolality e. g. hyperglycemia , hyper natremia , mannitol administ. Increase serum K 6 - Exercise inc serum K

Urinary Excretion • Increased serum K leads to increased aldosteron excretion from adrenal gland leading to excretion of K Also decrease serum K decrease its excretion • Increase renal tubular flow in distal nephrone increase K secretion

Hypokalemia

Definition • Normal serum potassium 3. 5 -5. 5 m. Eq/L • Hyp. Okalemia is a serum potassium less than 3. 5 m. Eq/L

Causes Of Hypokalemia • I- Inter compartmental shift : B 2 agonist Alkalosis Insulin Hypothermia (transient) • 2 - K Depletion: Extra renal e g diarrhea nasogastric suction Renal e g Diuretics , Mg depletion prevent reabsorption of k in renal tubule • 3 -Decreased K intake

Clinical manifestation Of Hypokalemia • I-CVS : ECG – P wave amplitude increase Prolonged P-R interval depressed S-T segment flattened or inverted T wave prominent U wave &QRS normal

CVS Effects • Dysrhythmia , myocardial dysfunction , myocardial fibrosis in chronic hypokalemia , orthostatic hypotension Increased digitalis toxicity • Neuromuscular : Sk ms weakness , Ileus , ms cramp , tetany

3 - Renal : • Nephrogenic D. I leads to polyurea • Alkalosis due to inc. HCO 3 re absorption • Increase ammonia production leads to encephalopathy when liver function is impaired 4 - Hormonal : decrease sec of aldosteron , insulin , GH. 5 - Metabolic : encephalopathy when liver function is impaired

Management of Hypokalemia • Check urine output before correction. • Oral replacement is safest if there is no serous organ dysfunction (60 -80 meq/day • I. V. replacement for serous cardiac manifestation , ms weakness or severe hypokalemia with : ECG monitoring - avoid dextrose sol. – - peripheral vein 8 meq/h , central vein 10 -20 meq/h

Causes of Hyperkalemia • 1 - Inter compartmental shift e. g. acidosis , B blockers, tissue breakdown , hemolysis, … • 2 -Decrease renal excretion e. g. renal failure , dec mineral corticoid activity , K sparing diuretics , ACEI, NSAI • 3 - Increased K intake e. g. old blood transfusion, salt substitute

Clinical manifestation Of Hyperkalemia • I- Skeletal ms weakness not seen until serum K> 7 meq/L due : - sustained depolarization - inactivation of Na channel • 2 - Cardiac (when serum K>7 meq/L)

ECG : Loss of P wave – prolonged P-R depressed S-T segment Tall peaked T wave –Wide QRS Short q-T may progress to V. F. , a systole accentuated by by acidosis , hyponatremia , hypocalcaemia
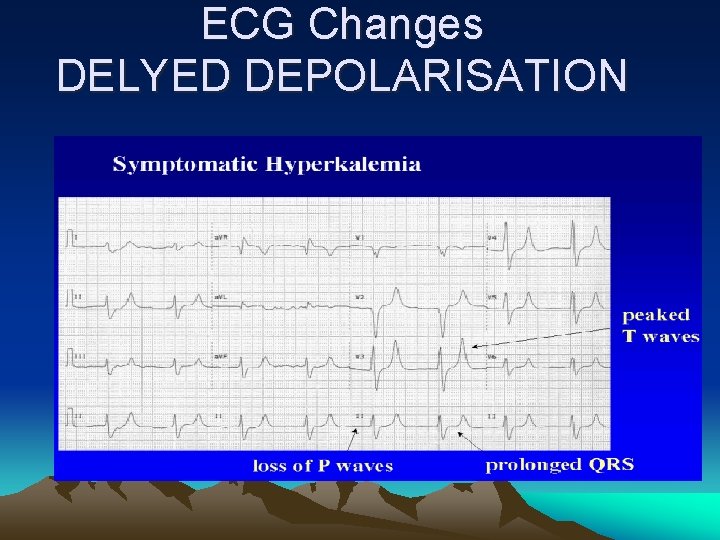
ECG Changes DELYED DEPOLARISATION
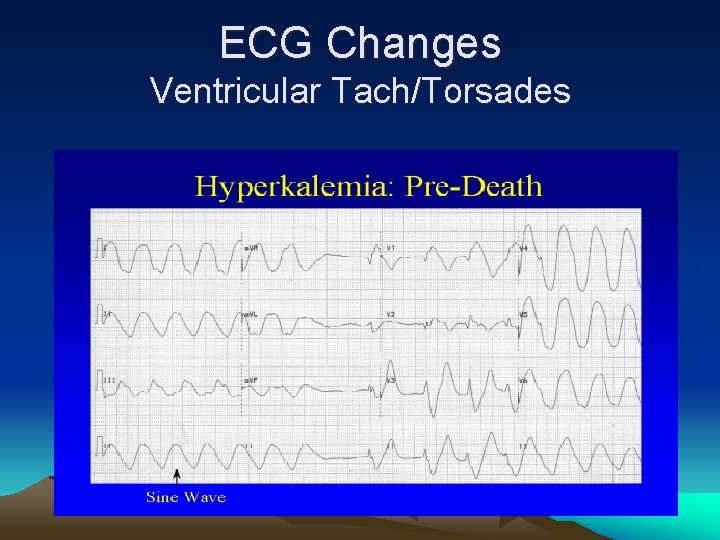
ECG Changes Ventricular Tach/Torsades
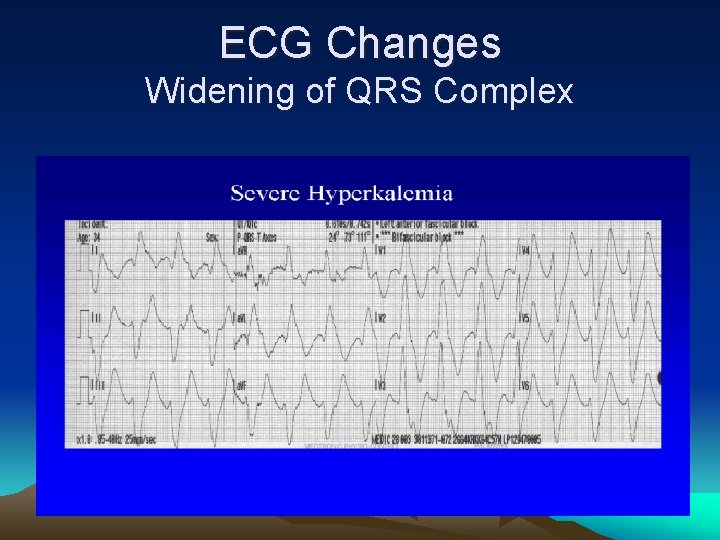
ECG Changes Widening of QRS Complex

Management of Hyperkalemia • Stop source of K intake , drugs contribute to hyperkalemia Mineral corticoid replacement. 1 - calcium to antagonize cardiac effects 3 -5 ml Ca. Cl 10% (acts immediately) or 5 -10 ml Ca gluconate 10% 2 - Na. HCO 3 in presence of M. acidosis ( It decrease serum K within 15 minutes ) 3 - Glucose & Insulin 50 gm+10 u( dec serum K 1 meq/h within one h

Management of Hyperkalemia • • 4 - Low dose epinephrine 0. 5 -2 ug/min 5 - B 2 agonist 6 - Lasix diuretics 7 -Potassium exchange resins as oral or rectal sodium polysterene sulphonate • 8 -Dialysis : hemodialysis is more effective than peritoneal dialysis
Treatment Removal of Potassium From the Body • Hemodialysis • Peritoneal Dialysis

- Slides: 43