FLUID ELECTROLYTE THERAPY Assoc Prof Dr Suat Bier

FLUID, ELECTROLYTE THERAPY Assoc. Prof. Dr. Suat Biçer Yeditepe University Faculty of Medicine Department of Child Health and Pediatrics

Regulation of Body Fluids, Electrolytes, & Tonicity Total body water (TBW) constitutes 50%– 75% of the total body mass, depending on age, sex, and fat content. After an initial postnatal diuresis, the TBW slowly decreases to the adult range near puberty (Figure – 1). TBW is divided into the intracellular and extracellular spaces. Intracellular fluid (ICF) accounts for twothirds of the TBW and extracellular fluid (ECF), for one-third. The ECF is further compartmentalized into plasma (intravascular) volume and interstitial fluid (ISF).
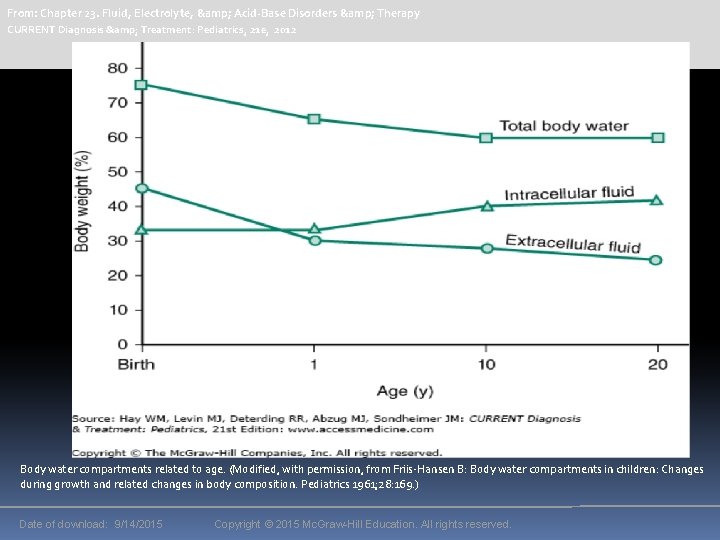
From: Chapter 23. Fluid, Electrolyte, & Acid-Base Disorders & Therapy CURRENT Diagnosis & Treatment: Pediatrics, 21 e, 2012 Body water compartments related to age. (Modified, with permission, from Friis-Hansen B: Body water compartments in children: Changes during growth and related changes in body composition. Pediatrics 1961; 28: 169. ) Date of download: 9/14/2015 Copyright © 2015 Mc. Graw-Hill Education. All rights reserved.

The principal constituents of plasma are sodium, chloride, bicarbonate, and protein (primarily albumin). The ISF is similar to plasma but lacks significant amounts of protein (Figure – 2). Conversely, the ICF is rich in potassium, magnesium, phosphates, sulfates, and protein.
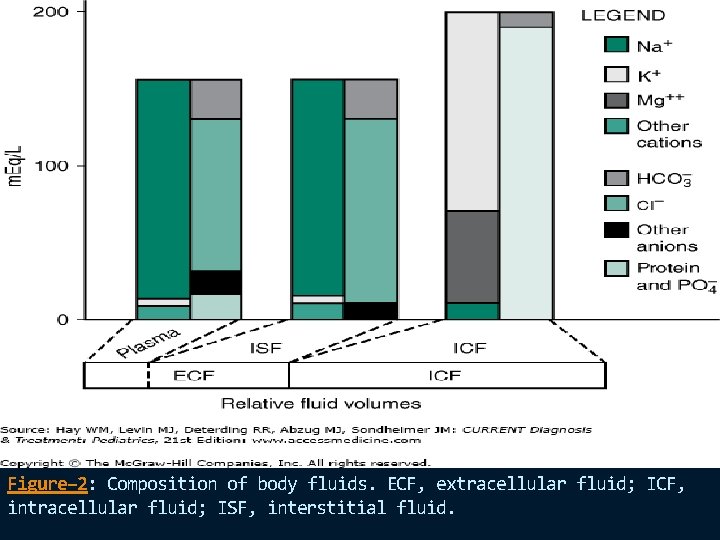
Figure– 2: Composition of body fluids. ECF, extracellular fluid; ICF, intracellular fluid; ISF, interstitial fluid.

Osmotic shifts between the ECF and ICF Na. Cl, mannitol, and glucose (in the setting of hyperglycemia) remain restricted to the ECF space and contribute effective osmoles by obligating water to remain in the ECF compartment. In contrast, a freely permeable solute such as urea does not contribute effective osmoles because it is not restricted to the ECF and readily crosses cell membranes.

Osmolality may be estimated by the following formula: +

Oncotic pressure, or colloid osmotic pressure, represents the osmotic activity of macromolecular constituents such as albumin in the plasma and body fluids. The principal mechanisms that regulate ECF volume and tonicity are antidiuretic hormone (ADH), thirst, aldosterone, and atrial natriuretic factor (ANF).

Fluid & Electrolyte Management Therapy of fluid and electrolyte disorders: - providing maintenance fluid and electrolyte requirements, - replenishing prior losses, - replacing persistent abnormal losses.

Therapy should be phased to (1) rapidly expand the ECF volume and restore tissue perfusion, (2) replenish fluid and electrolyte deficits while correcting attendant acid-base abnormalities, (3) meet the patient’s nutritional needs, (4) replace ongoing losses.

maintenance fluid and electrolyte requirements Provision of enough water, glucose, and electrolytes to prevent deterioration of body stores for a euvolemic patient.

calculation of maintenance requirements based on: body surface area, weight, caloric expenditure A system based on caloric expenditure is most helpful, because 1 m. L of water is needed for each kilocalorie expended.
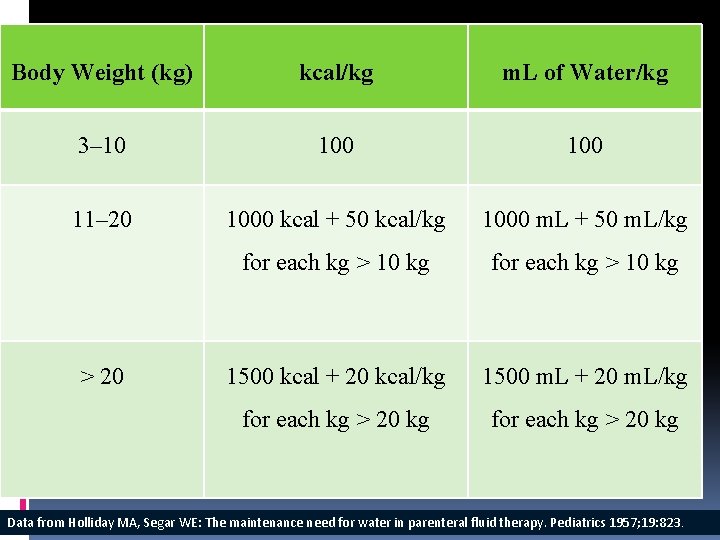
Body Weight (kg) kcal/kg m. L of Water/kg 3– 10 100 11– 20 1000 kcal + 50 kcal/kg 1000 m. L + 50 m. L/kg for each kg > 10 kg 1500 kcal + 20 kcal/kg 1500 m. L + 20 m. L/kg for each kg > 20 Data from Holliday MA, Segar WE: The maintenance need for water in parenteral fluid therapy. Pediatrics 1957; 19: 823.
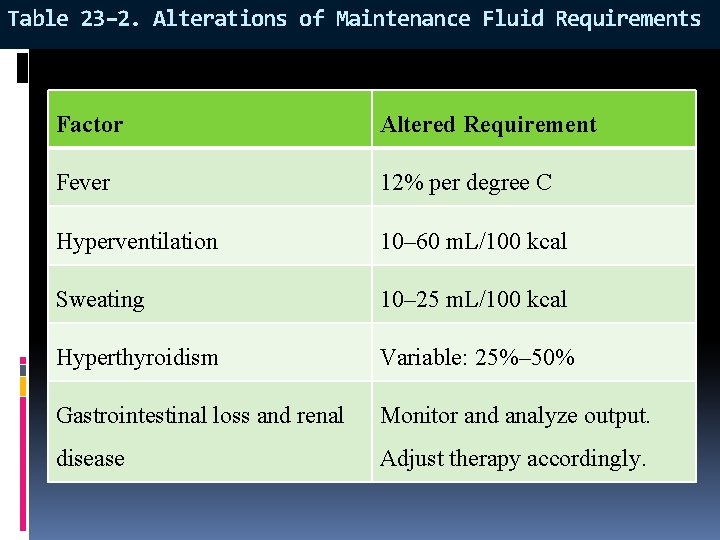
Table 23– 2. Alterations of Maintenance Fluid Requirements Factor Altered Requirement Fever 12% per degree C Hyperventilation 10– 60 m. L/100 kcal Sweating 10– 25 m. L/100 kcal Hyperthyroidism Variable: 25%– 50% Gastrointestinal loss and renal Monitor and analyze output. disease Adjust therapy accordingly.

Electrolyte losses occur primarily through the urinary tract and to a lesser degree via the skin and stool. maintenance needs are 3– 4 m. Eq Na+/100 kcal and 2 m. Eq K+/100 kcal or 30– 40 m. Eq Na+/L and 20 m. Eq K+/L,

monitor the patient’s daily weight, urinary output, fluid input, and urine specific gravity.

Dehydration Children have a high incidence of gastrointestinal diseases, including gastroenteritis, and may demonstrate gastrointestinal symptoms in nongastrointestinal conditions, such as pneumonia or meningitis. Infants and young children often decrease their oral intake when ill, and their high ratio of surface area to weight promotes significant evaporative losses. Renal concentrating mechanisms do not maximally conserve water in early life, and fever may significantly increase fluid needs.

Dehydration decreases ECF volume, leading to decreased tissue perfusion, progressive uremia and abnormal renal function studies, compensatory tachycardia, and lactic acidosis. The clinical effects of dehydration relate to the degree of dehydration and to the relative amounts of salt and water lost.

Caregivers must be particularly aware of dehydration occurring in breast-fed newborn infants who go home soon after birth and whose mothers fail to produce enough milk. This problem is more common in the hot summer months and has been associated with severe dehydration, brain damage, and death.

The clinical evaluation of a child with dehydration should focus on the composition and volume of fluid intake; the frequency and amount of vomiting, diarrhea, and urine output; the degree and duration of fever; the nature of any administered medications; and the existence of underlying medical conditions.

A recently recorded weight, if known, can be very helpful in calculating the magnitude of dehydration. Important clinical features in estimating the degree of dehydration include the capillary refill time, postural blood pressure, and heart rate changes; dryness of the lips and mucous membranes; lack of tears; lack of external jugular venous filling when supine; a sunken fontanelle in an infant; oliguria; and altered mental status (Table – 3).
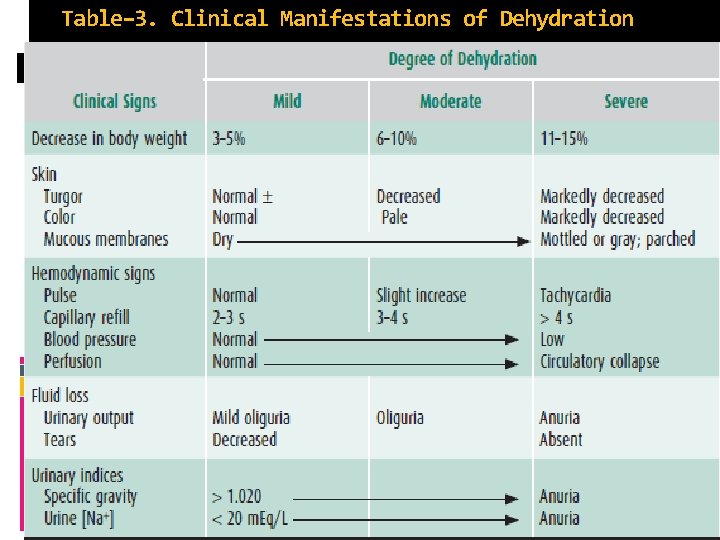
Table– 3. Clinical Manifestations of Dehydration

Children generally respond to a decrease in circulating volume with a compensatory increase in pulse rate and may maintain their blood pressure in the face of severe dehydration. A low or falling blood pressure is, therefore, a late sign of shock in children, and when present should prompt emergent treatment.

Salient laboratory parameters include: a high urine specific gravity (in the absence of an underlying renal concentrating defect as seen in diabetes insipidus or chronic obstructive or reflux nephropathy), a relatively greater elevation in blood urea nitrogen than in serum creatinine, a low urinary [Na+] excretion (< 20 m. Eq/L), an elevated hematocrit or serum albumin level secondary to hemoconcentration.

Emergent intravenous therapy is indicated when there is evidence of compromised perfusion (inadequate capillary refill, tachycardia, poor color, oliguria, or hypotension). The initial goal is to rapidly expand the plasma volume and to prevent circulatory collapse. Either colloid (5% albumin) or crystalloid (normal saline or Ringer lactate) may be used. Colloid is particularly useful in hypernatremic patients in shock, in malnourished infants, and in neonates.

If no intravenous site is available, fluid may be administered intraosseously through the marrow space of the tibia. If there is no response to the first fluid bolus a second bolus may be given When adequate tissue perfusion is demonstrated by improved capillary refill, decreased pulse rate, urine output, improved mental status

If adequate perfusion is not restored after 40 m. L/kg of isotonic fluids, other pathologic processes must be considered such as sepsis, occult hemorrhage, cardiogenic shock

Isotonic dehydration may be treated by providing half of the remaining fluid deficit over 8 hours and the second half over the ensuing 16 hours in the form of 5% dextrose with 0. 45% saline containing 20 m. Eq/L of KCl. In the presence of metabolic acidosis, potassium acetate may be considered. Maintenance fluids and replacement of ongoing losses should also be provided. If the patient is unable to eat for a prolonged period, nutritional needs must be met through hyperalimentation or enteral tube feedings.
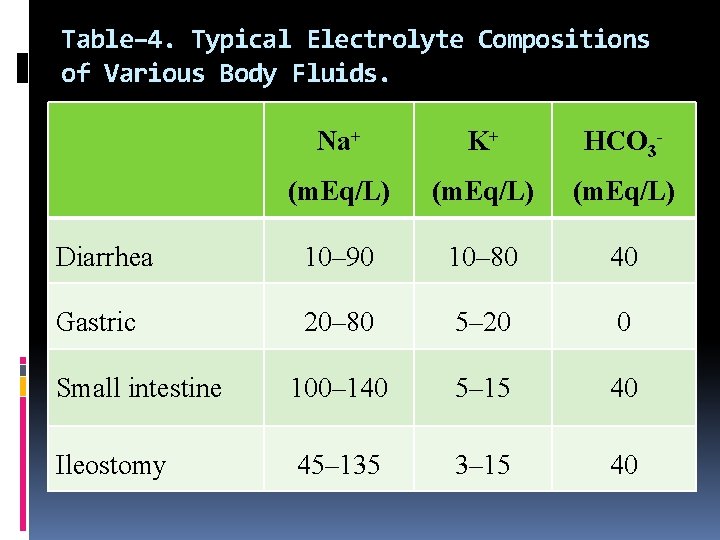
Table– 4. Typical Electrolyte Compositions of Various Body Fluids. Na+ K+ HCO 3 - (m. Eq/L) Diarrhea 10– 90 10– 80 40 Gastric 20– 80 5– 20 0 Small intestine 100– 140 5– 15 40 Ileostomy 45– 135 3– 15 40

Oral Rehidration Therapy (ORT) mild to moderate dehydration Clear liquid beverages found in the home, such as broth, soda, juice, and tea are inappropriate for the treatment of dehydration. Commercially available ORT solutions provide 45– 75 m. Eq/L of Na+, 20– 25 m. Eq/L of K+, 30– 34 m. Eq/L of citrate or bicarbonate, and 2%– 2. 5% of glucose.

ORT Frequent small aliquots (5– 15 m. L) should be given to provide approximately 50 m. L/kg over 4 hours for mild dehydration up to 100 m. L/kg over 6 hours for moderate dehydration.

Oral rehydration is contraindicated in children with: altered levels of consciousness, respiratory distress who cannot drink freely; in children suspected of having an acute surgical abdomen; in infants with greater than 10% volume depletion; in children with hemodynamic instability; in the setting of severe hyponatremia ([Na+] < 120 m. Eq/L) hypernatremia ([Na+] > 160 m. Eq/L). • Failure of oral rehydration due to persistent vomiting or inability to keep up with losses mandates intravenous therapy.
![The type of dehydration is characterized by the serum [Na+]. If relatively more The type of dehydration is characterized by the serum [Na+]. If relatively more](http://slidetodoc.com/presentation_image_h2/6a5e50a40f85a34afc2a5ef476347acc/image-33.jpg)
The type of dehydration is characterized by the serum [Na+]. If relatively more solute is lost than water, the [Na+] falls, and hyponatremic dehydration ([Na+] < 130 m. Eq/L) ensues. Tissue perfusion is more significantly impaired for a given degree of hyponatremic dehydration than for a comparable degree of isotonic or hypertonic dehydration. It is important to note, however, that significant solute losses also occur in hypernatremic dehydration. Furthermore, because plasma volume is somewhat protected in hypernatremic dehydration, it poses the risk of the clinician underestimating the severity of dehydration.
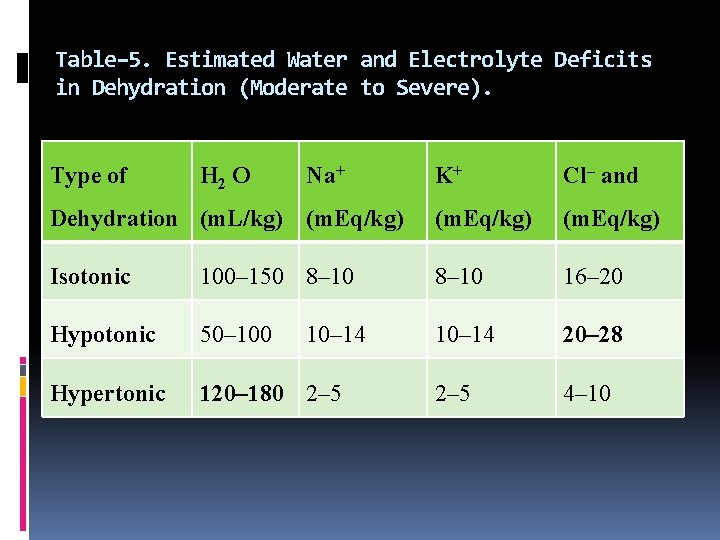
Table– 5. Estimated Water and Electrolyte Deficits in Dehydration (Moderate to Severe). Type of H 2 O Na+ K+ Cl– and Dehydration (m. L/kg) (m. Eq/kg) Isotonic 100– 150 8– 10 16– 20 Hypotonic 50– 100 10– 14 20– 28 Hypertonic 120– 180 2– 5 4– 10 10– 14

Hyponatremia • Hyponatremia may be factitious in the presence of high plasma lipids or proteins, which decrease the percentage of plasma volume that is water. • Hyponatremia in the absence of hypotonicity also occurs when an osmotically active solute, such as glucose or mannitol, is added to the ECF. • Water drawn from the ICF dilutes the serum [Na+] despite isotonicity or hypertonicity.

Patients with hyponatremic dehydration generally demonstrate typical signs and symptoms of dehydration, because the vascular space is compromised as water leaves the ECF to maintain osmotic neutrality. The treatment of hyponatremic dehydration is fairly straightforward. The magnitude of the sodium deficit may be calculated by the following formula

Half of the deficit is replenished in the first 8 hours of therapy, and the remainder is given over the following 16 hours. Maintenance and replacement fluids should also be provided. The deficit plus maintenance calculations generally approximate 5% dextrose with 0. 45% saline. The rise in serum [Na+] should not exceed 0. 5– 1. 0 m. Eq/L/h or more than 20 m. Eq/L/24 h unless the patient demonstrates central nervous system symptoms that warrant more rapid initial correction. The dangers of too rapid correction of hyponatremia include cerebral dehydration and injury due to fluid shifts from the ICF compartment.

Hypovolemic hyponatremia also occurs in cerebral salt wasting associated with CNS insults, a condition characterized by high urine output and elevated urinary [Na+] (> 80 m. Eq/L) due to an increase in ANF. This must be distinguished from the syndrome of inappropriate secretion of ADH (SIADH), which may also become manifest in CNS conditions and certain pulmonary disorders. In contrast to cerebral salt wasting, SIADH is characterized by euvolemia or mild volume expansion and relatively low urine output due to ADH-induced water retention. Urinary [Na+] is high in both conditions, though generally not as high as in SIADH. It is important to distinguish between these two conditions, because the treatment of the former involves replacement of urinary salt and water losses, whereas the treatment of SIADH involves water restriction.
![In cases of severe hyponatremia (serum [Na+] < 120 m. Eq/L) with CNS In cases of severe hyponatremia (serum [Na+] < 120 m. Eq/L) with CNS](http://slidetodoc.com/presentation_image_h2/6a5e50a40f85a34afc2a5ef476347acc/image-39.jpg)
In cases of severe hyponatremia (serum [Na+] < 120 m. Eq/L) with CNS symptoms, intravenous 3% Na. Cl may be given over 1 hour to raise the [Na+] to 120 m. Eq/L, to alleviate CNS manifestations and sequelae. In general, 6 m. L/kg of 3% Na. Cl will raise the serum [Na+] by about 5 m. Eq/L. Further correction should proceed slowly, as outlined earlier.

Hypervolemic hyponatremia May occur in edematous disorders such as nephrotic syndrome, congestive heart failure, and cirrhosis, wherein water is retained in excess of salt. Treatment involves restriction of Na+ and water and correction of the underlying disorder. Hypervolemic hyponatremia due to water intoxication is characterized by a maximally dilute urine (specific gravity < 1. 003) and is also treated with water restriction.

Hypernatremia Although diarrhea is commonly associated with hyponatremic or isonatremic dehydration, hypernatremia may develop in the presence of persistent fever or decreased fluid intake or in response to improperly mixed rehydration solutions. Extreme care is required to treat hypernatremic dehydration appropriately. If the serum [Na+] falls precipitously, the osmolality of the ECF drops more rapidly than that of the CNS. Water shifts from the ECF compartment into the CNS to maintain osmotic neutrality.
![If hypertonicity is corrected too rapidly (a drop in [Na +] of greater If hypertonicity is corrected too rapidly (a drop in [Na +] of greater](http://slidetodoc.com/presentation_image_h2/6a5e50a40f85a34afc2a5ef476347acc/image-42.jpg)
If hypertonicity is corrected too rapidly (a drop in [Na +] of greater than 0. 5– 1 m. Eq/L/h), cerebral edema, seizures, and CNS injury may occur. Thus, following the initial restoration of adequate tissue perfusion using isotonic fluids, a gradual decrease in serum [Na+] is desired (10– 15 m. Eq/L/d). This is commonly achieved using 5% dextrose with 0. 45% saline to replace the calculated fluid deficit over 48 hours. Maintenance and replacement fluids should also be provided. If the serum [Na+] is not correcting appropriately, the free water deficit may be estimated as 4 m. L/kg of free water for each milliequivalent of serum [Na+] above 145 m. Eq/L and provided as 5% dextrose over 48 hours.

If metabolic acidosis is also present, it must be corrected slowly to avoid CNS irritability. Potassium is provided as indicated—as the acetate salt if necessary. Electrolyte concentrations should be assessed every 2 hours in order to control the decline in serum [Na+]. Elevations of blood glucose and blood urea nitrogen may worsen the hyperosmolar state in hypernatremic dehydration and should also be monitored closely. Hyperglycemia is often associated with hypernatremic dehydration and may necessitate lower intravenous glucose concentrations (eg, 2. 5%).

Patients with diabetes insipidus, whether nephrogenic or central in origin, are prone to develop profound hypernatremic dehydration as a result of unremitting urinary -free water losses (urine specific gravity < 1. 010), particularly during superimposed gastrointestinal illnesses associated with vomiting or diarrhea. Treatment involves restoration of fluid and electrolyte deficits as described earlier as well as replacement of excessive water losses. Formal water deprivation testing to distinguish responsiveness to ADH should only be done during daylight hours after restoration of normal fluid volume status.

However, if a child presents with marked dehydration and a serum Na+ greater than 150 m. Eq/L, it may prove helpful and timely to obtain a plasma vasopressin level at the time of their initial presentation. Hypervolemic hypernatremia (salt poisoning), associated with excess total body salt and water, may occur as a consequence of providing improperly mixed formula, excessive Na. Cl or Na. HCO 3 administration, or as a feature of primary hyperaldosteronism. Treatment includes the use of diuretics, and potentially, concomitant water replacement or even dialysis.

Potassium Disorders The predominantly intracellular distribution of potassium is maintained by the actions of Na+-K+-ATPase in the cell membranes. Potassium is shifted into the ECF and plasma by acidemia and into the ICF in the setting of alkalosis, hypochloremia, or in conjunction with insulin-induced cellular glucose uptake. The ratio of intracellular to extracellular K+ is the major determinant of the cellular resting membrane potential and contributes to the action potential in neural and muscular tissue.

Abnormalities of K+ balance are potentially life- threatening. In the kidney, K+ is filtered at the glomerulus, reabsorbed in the proximal tubule, and excreted in the distal tubule. Distal tubular K+ excretion is regulated primarily by aldosterone. Renal K+ excretion continues for significant periods even after the intake of K+ is decreased. Thus, by the time urinary [K+] decreases, the systemic K+ pool has been depleted significantly.

The causes of net K+ loss are primarily renal in origin. Gastrointestinal losses through nasogastric suction or vomiting reduce total body K+ to some degree. However, the resultant volume depletion results in an increase in plasma aldosterone, promoting renal excretion of K+ in exchange for Na+ reclamation to preserve circulatory volume. Diuretics (especially thiazides), mineralocorticoids, and intrinsic renal tubular diseases (eg, Bartter syndrome) enhance the renal excretion of K+.

Hypokalaemia is defined as a potassium level < 3. 5 mmol/L Moderate hypokalaemia is a serum level of < 3. 0 mmol/L Severe hypokalaemia is defined as a level < 2. 5 mmol/L

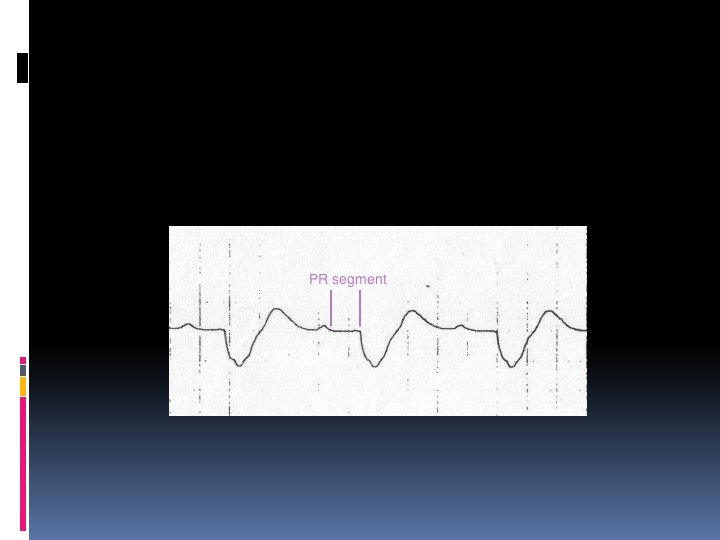
Clinically, hypokalemia is associated with neuromuscular excitability, decreased peristalsis or ileus, hyporeflexia, paralysis, rhabdomyolysis, and arrhythmias. Electrocardiographic changes include flattened T waves, a shortened PR interval, and the appearance of U waves. Arrhythmias associated with hypokalemia include premature ventricular contractions; atrial, nodal, or ventricular tachycardia; and ventricular fibrillation. Hypokalemia increases responsiveness to digitalis and may precipitate overt digitalis toxicity. In the presence of arrhythmias, extreme muscle weakness, or respiratory compromise, intravenous K+ should be given.

Hypokalaemia is often associated with hypomagnesaemia, which increases the risk of malignant ventricular arrhythmias Check potassium and magnesium in any patient with an arrhythmia Top up the potassium to 4. 0 -4. 5 mmol/l and the magnesium to > 1. 0 mmol/l to stabilise the myocardium and protect against arrhythmias – this is standard practice in most CCUs and ICUs If the patient is hypophosphatemic ([ PO 43 -] < 2 mg/d. L), a phosphate salt may be used.

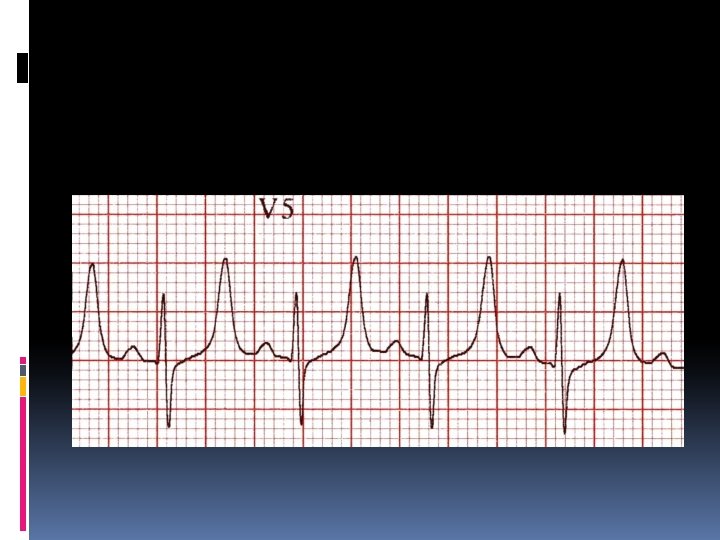
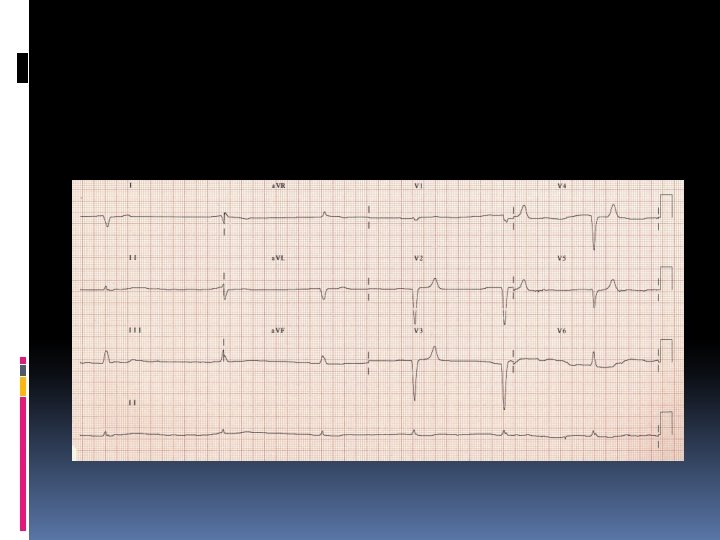
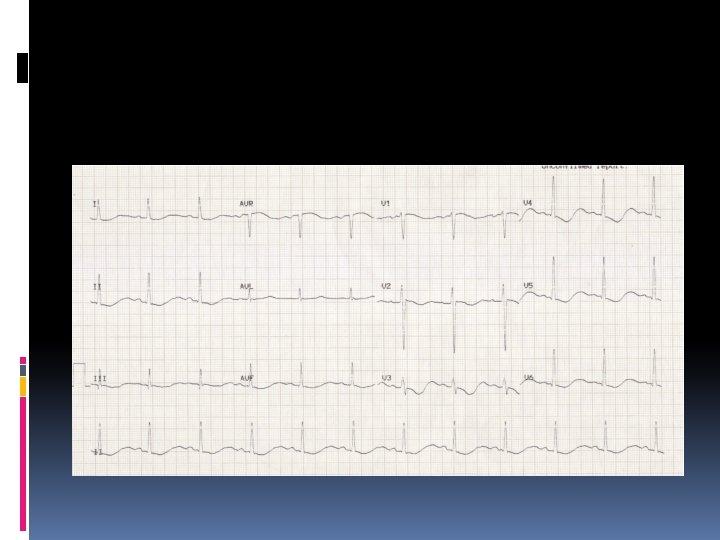
Increased amplitude and width of the P wave Prolongation of the PR interval T wave flattening and inversion ST depression Prominent U waves (best seen in the precordial leads) Apparent long QT interval due to fusion of the T and U waves (= long QU interval) Frequent supraventricular and ventricular ectopics Supraventricular tachyarrhythmias: AF, atrial flutter, atrial tachycardia Potential to develop life-threatening ventricular arrhythmias, e. g. VT, VF and Torsades de Pointes
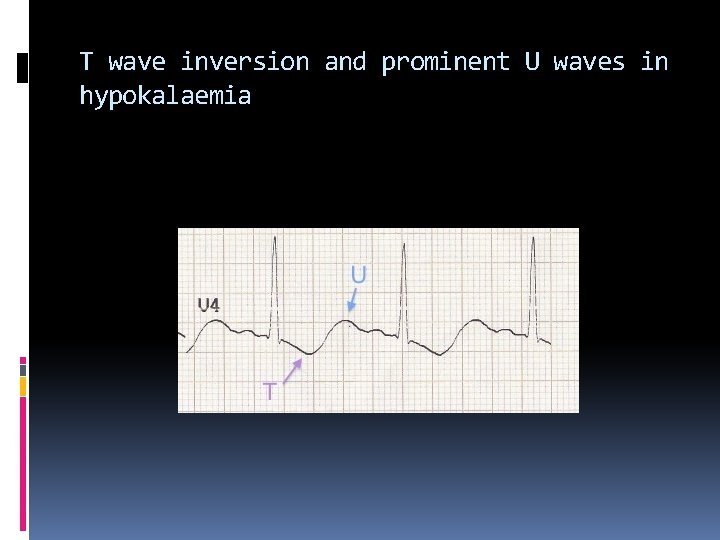
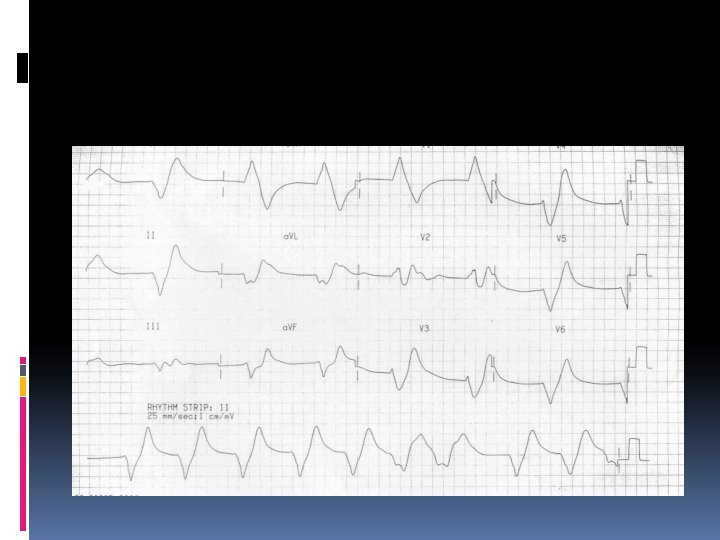
T wave inversion and prominent U waves in hypokalaemia
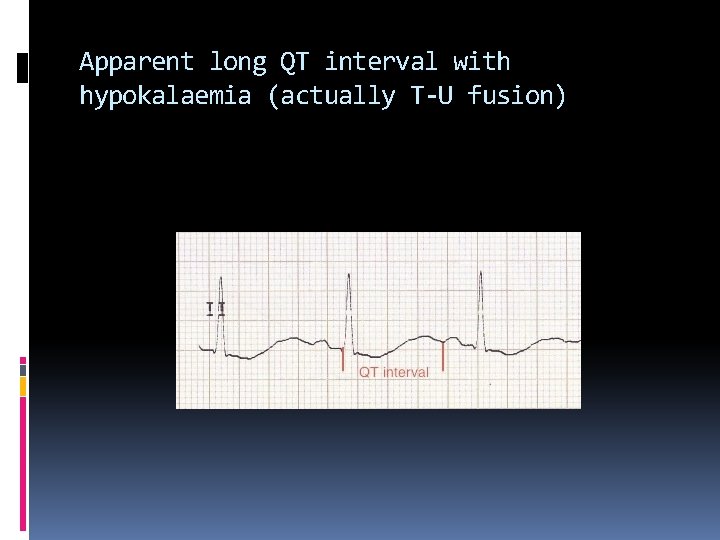
Apparent long QT interval with hypokalaemia (actually T-U fusion)

The first priority in the treatment of hypokalemia is the restoration of an adequate serum [K+]. Providing maintenance amounts of K+ is usually sufficient; however, when the serum [K+] is dangerously low and K+ must be administered intravenously, it is imperative that the patient have a cardiac monitor. Intravenous K+ should generally not be given faster than at a rate of 0. 3 m. Eq/kg/h. Oral K+ supplements may be needed for weeks to replenish depleted body stores.

Hyperkalemia Due to decreased renal K+ excretion, Mineralocorticoid deficiency or unresponsiveness, K+ release from the ICF compartment

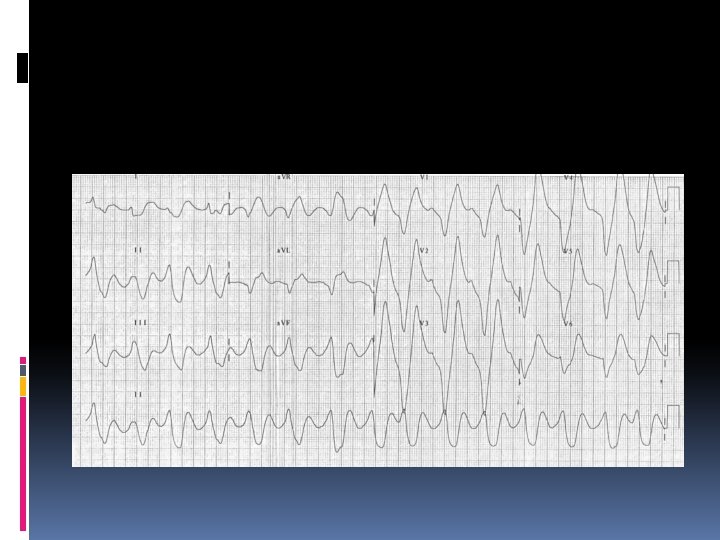
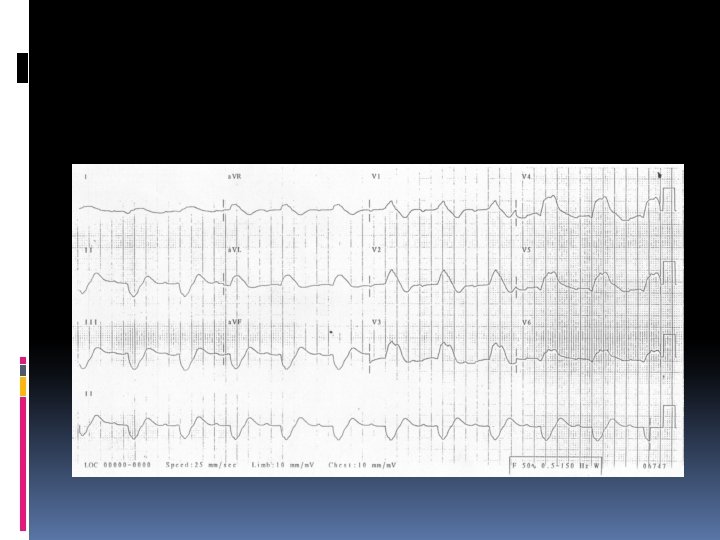
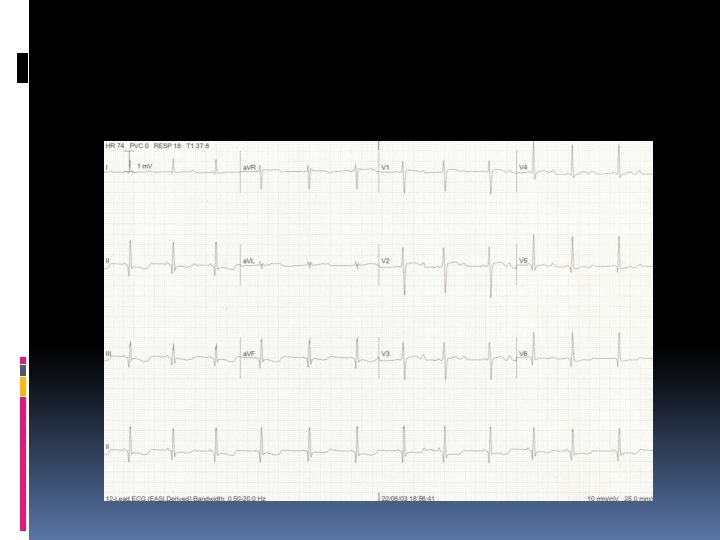
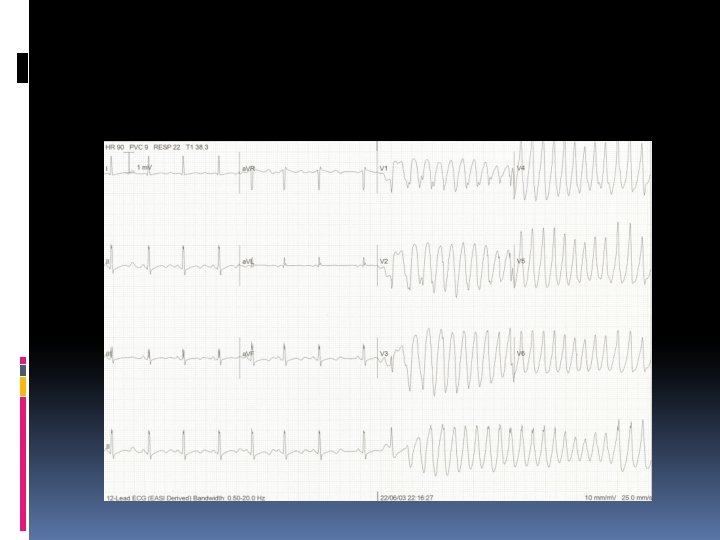
Hyperkalemia characterized by Muscle weakness, Paresthesias, and tetany; Ascending paralysis; Arrhythmias. Electrocardiographic changes associated with hyperkalemia include • Peaked T waves, • Widening of the QRS complex, • Arrhythmias such as sinus bradycardia or sinus arrest, atrioventricular block, nodal or idioventricular rhythms, and ventricular tachycardia or fibrillation.
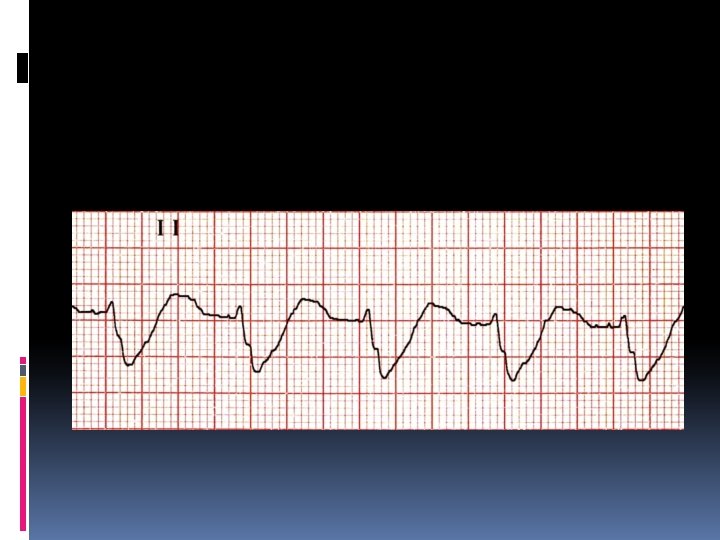
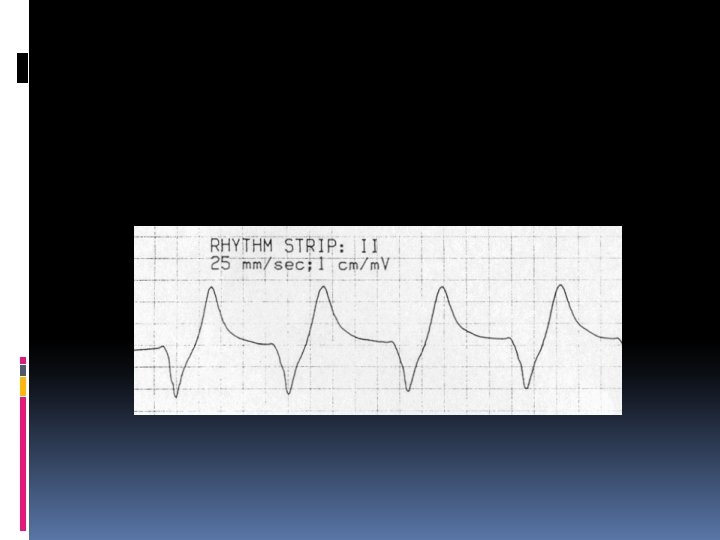
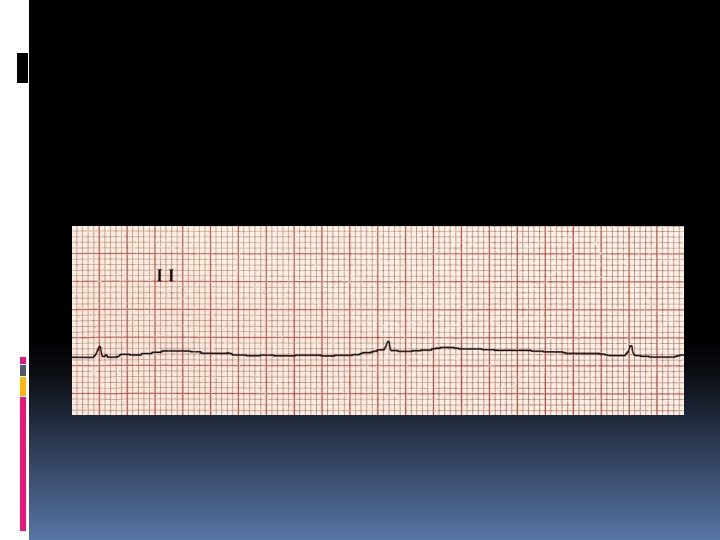

The severity of hyperkalemia depends on the ECG changes, the status of the other electrolytes, and the stability of the underlying disorder. A rhythm strip should be obtained when significant hyperkalemia is suspected. If the serum [K+] is < 6. 5 m. Eq/L, discontinuing K+ supplementation is usually sufficient. If the serum [K+] is > 7 m. Eq/L or if potentiating factors such as hyponatremia, digitalis toxicity, and renal failure are present, more aggressive therapy is needed. If ECG changes or arrhythmias are present, treatment must be initiated promptly.

IV 10% calcium gluconate (0. 2– 0. 5 m. L/kg over 2– 10 minutes) will rapidly ameliorate depolarization and may be repeated after 5 minutes if ECG changes persist. Calcium should be given only with a cardiac monitor in place and should be discontinued if bradycardia develops. The IV administration of a diuretic that acts in the loop of Henle, such as furosemide (1– 2 mg/kg), will augment renal K+ excretion and can be very helpful in lowering serum and total body [K+]. Administering Na+ and increasing systemic p. H with bicarbonate therapy (1– 2 m. Eq/kg) will shift K+ from the ECF to the ICF compartment, as will therapy with a β-agonist such as albuterol.

In nondiabetic patients, 0. 5 g/kg of glucose over 1 – 2 hours will enhance endogenous insulin secretion, lowering serum [K+] 1– 2 m. Eq/L. Administration of IV glucose and insulin may be needed as a simultaneous drip (0. 5– 1 g/kg glucose and 0. 3 units of regular insulin per gram of glucose) given over 2 hours with monitoring of the serum glucose level every 15 minutes.

The therapies outlined above provide transient benefits. Ultimately, K+ must be reduced to normal levels by: reestablishing adequate renal excretion using diuretics optimizing urinary flow, using ion exchange resins such as sodium polystyrene sulfonate orally or as a retention enema (0. 2– 0. 5 g/kg orally or 1 g/kg as an enema), or by dialysis.

- Slides: 74