Fluid Electrolyte Balance Seattle University College of Nursing

Fluid & Electrolyte Balance Seattle University College of Nursing NURS 3205: Pathophysiology Maria Pettinato, RN, Ph. D Therry Eparwa, DNP, RN, FNP-

Fluids • Total Body Water (TBW) = sum of all fluids within all body compartments – About 45 -80% of body weight is water • Fat cells (less water) • Muscle cells (more water) • 1 Liter water = 2. 2 lbs (1 kg) Total Body Water (%) in Relation to Body Weight Body Build Adult Male Adult Female Infant Normal 60 50 70 Lean 70 60 80 Obese 50 42 60

Fluids Intracellula r Fluid (2/3) Total Body Water Interstitial Fluid (~2/3) Extracellul ar Fluid (1/3) Intravascul ar Fluid (~1/3) Miscellane ous Body Fluids

The Importance of Fluids • Homeostasis • Maintaining volume balance • Maintaining solute/electrolyte concentrations (osmolarity) balance • Maintaining p. H (concentration of H+ ions)

Definitions • Solute: a substance dissolved in a solution (e. g. , a salt) • Solvent: a liquid, solid, or gas that dissolves another solid, liquid, or gaseous solute, resulting in a solution • Solution: a homogeneous mixture composed of 2+ substances dissolved in a solvent

Solute Concentration • The amount of solute dissolved in solvent • Solute concentrations are being compared between two solutions • Three solute states: – Hypertonic = Concentrated; a solution with a higher concentration of solutes – Hypotonic = Diluted; a solution with a lower concentration of solutes – Isotonic = Balanced ; a solution that is equal in concentration to another solution

Volume States • Volume usually refers to vascular volume – Too little = dehydration – Too much = overhydration • Retaining fluids – Balanced = normovolemic • Volume is regulated to within about 10% by renin-angiotensin-aldosterone system (RAAS) and by natriuretic factors
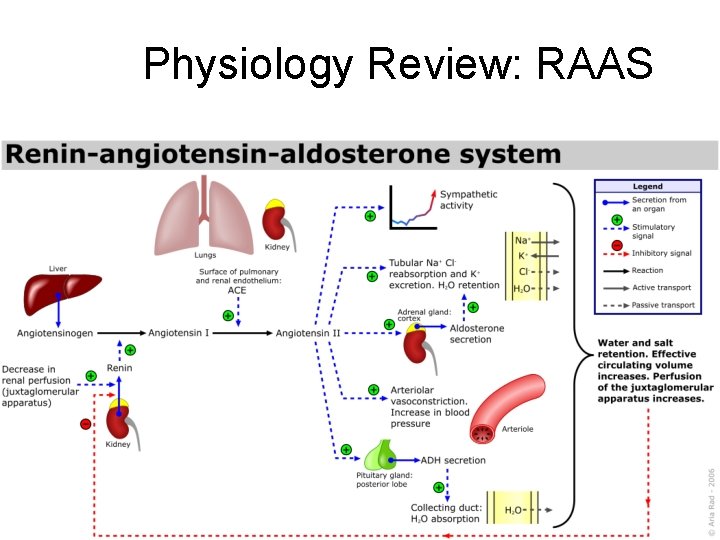
Physiology Review: RAAS

Solute/Volume States • Solute: Hypertonic, hypotonic, isotonic • Volume: Dehydration, overhydration, normovolemic – Hypertonic dehydration; hypotonic dehydration, isotonic dehydration – hypertonic overhydration, hypotonic overhydration; isotonic overhydration – Hypertonic normovolemic, hypotonic normovolemic – Isotonic normovolemic = Normal

Principles of Regulation • Blood (intravascular compartment) is the only thing we can affect or measure • The protein and ion composition of the different compartments is different while their osmolality (concentration) is the same – Intracellular: high protein, high K+, high PO 43 - (phosphate ions), low Na+ – Extracellular: high Na+, high Cl– Interstitial: low protein – Intravascular (blood): high protein

Capillary Walls • Capillary walls control the movement of fluid between intravascular space (the fluid in blood vessels) and interstitial fluid (the fluid in between cells) • Walls are made of single layer flat endothelial cells, often with fenestrations that allow ions and small molecules out • Usually proteins and cells stay in • All capillaries permeable to O 2, CO 2, H 2 O and glucose • Permeability varies by location – Example: the brain and the blood-brain barrier • Permeability varies during inflammatory response – Permeability is increased

Water Movement • The principal force determining the movement of water is Osmotic Force – a. k. a. osmotic pressure – Water moves toward the compartment where the number of particles is greatest (i. e. , water moves to the area with higher concentrations) • Distribution of water and other elements occurs as a result of hydrostatic pressure and osmotic pressure • Osmotic pressure (a. k. a. oncotic pressure) is the attractive pressure of a colloid or particle in solution – The pressure that pulls water across a membrane • Hydrostatic pressure is the pressure exerted by a

Osmoreceptors • Sensors in the hypothalamus that can detect changes in osmotic pressure and plasma osmolality – Plasma Osmolality = the measure of concentration of substances in blood (sodium, chloride, potassium, etc. ) • Regulates water balance • Regulates concentrations to within about 2% • Releases antidiuretic hormone (ADH) – ADH increases the permeability of renal
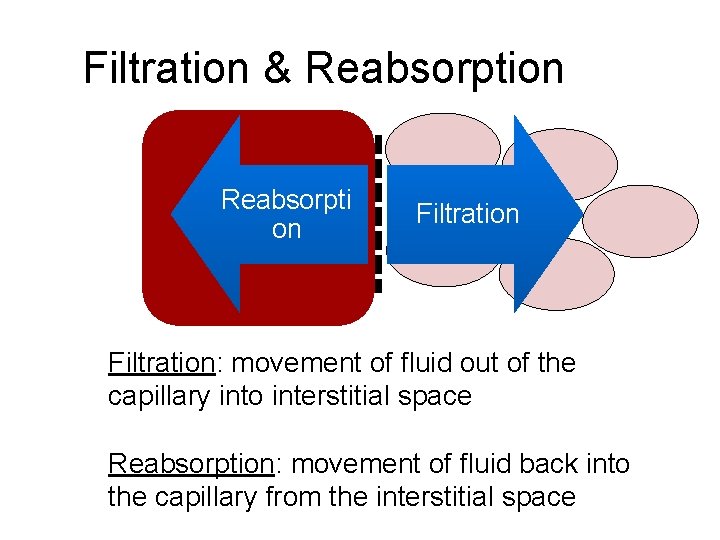
Filtration & Reabsorption Reabsorpti on Filtration: movement of fluid out of the capillary into interstitial space Reabsorption: movement of fluid back into the capillary from the interstitial space

Filtration & Reabsorption • Four Forces: – Capillary hydrostatic pressure (blood pressure) – Capillary oncotic pressure – Interstitial hydrostatic pressure – Interstitial oncotic pressure

Starling’s Hypothesis • In 1892 Starling concluded that fluid balance across the capillary must be a balance of hydrostatic and osmotic forces • The movement of fluid back and forth across the capillary wall is its net filtration • Starling’s Forces – Net filtration = (forces favoring filtration) – (forces opposing filtration) – Forces favoring filtration = capillary hydrostatic pressure and interstitial oncotic pressure – Forces opposing filtration =capillary oncotic pressure and interstitial hydrostatic pressure

Edema • Excessive accumulation of fluid within the interstitial spaces • Mechanisms of Edema • Lymphatic obstruction (ex: post-mastectomy scarring) • Inflammation (ex: sepsis, anaphylaxis) • Increased hydrostatic pressure in vessels (ex: CHF with chronic renin activation, nonjudicious use of IV therapy) • Decreased osmotic concentration of the blood (ex: liver failure, starvation)

Edema

Alterations of Fluid Balance • If capillaries allow protein to leak out – Fluid shifts to interstitial compartment, causes swelling and edema – Patient will be hypovolemic because we measure the fluid volume in the vascular compartment • Liver Failure – Liver is no longer making plasma proteins to go into the blood vasculature – Fluid shifts from intravascular space to interstitial spaces (edema, ascites) • Starvation – Not eating so not making enough plasma proteins, causes edema, ascites -- Kwashiorkor in children • Increased Blood Pressure – Fluid shifts fluid out of capillary because of

Isotonic Alterations • Occur when TBW changes are accompanied by proportional changes in electrolytes • Osmolality remains in normal range – Osmolality = the measure of concentration of substances in a compartment (sodium, chloride, potassium, etc. )

Isotonic Alterations: Examples • Isotonic Dehydration: loss of isotonic fluid in vasculature – Patient is losing both electrolytes and fluid – Bleeding, vomiting or diarrhea, “third spacing” (body fluids accumulate in the third space) – Symptoms: orthostatic hypotension, sudden weight loss*, flat neck veins, dizziness, concentrated urine • Isotonic Overhydration: too much isotonic fluid in vasculature – Patient has too much electrolytes and fluid – Either from organ failure or too much administration • Congestive heart failure (CHF), renal failure,

Hypertonic Alterations • Develops when osmolality (concentration) of the ECF is above normal • Most common causes: – Sodium excess in ECF – Water deficit in ECF

Hypertonic Alteration: Hypernatremia • • Na+ > 145 m. Eq/L of serum Occurs with acute gain in sodium or net loss of water Results in intracellular dehydration Causes: inappropriate admin. of hypertonic saline, oversecretion of aldosterone, tube feeding, neardrowning in salt water, loss of more water than salt (diabetes insipidus), osmotic diuresis (diabetic ketoacidosis) – Hypertonic overhydration – drinking sea water • Symptoms: thirst, fever, dry mucous membranes; CNS dysfunction – confusion, lethargy, seizures, coma, death

Hypertonic Alteration: Water Deficit (Dehydration) • Pure water deficits are rare – Dehydration is usually with both sodium and water loss • Causes: hyperventilation caused by fever, insensible water loss, renal clearance of free water with inability to concentrate urine • Symptoms: thirst, dry skin and mucous membranes, elevated temperatures, tachycardia, weak pulses

Hypotonic Alterations • Develops when osmolality (concentration) of the ECF is less than normal • Most common causes: – Sodium deficit in ECF – Water excess in ECF

Hypotonic Alteration: Hyponatremia • Na+ < 135 m. Eq/L of serum • Occurs with sodium loss, inadequate sodium intake, or dilution of sodium level by water excess • Pure Sodium Deficit: vomiting, diarrhea, GI suctioning, burns, diuretic use, inadequate sodium intake (rare) • Dilutional Hyponatremia: fluid • Hypoosmolar Hyponatremia: both TBW and sodium levels are increased, but TBW exceeds the increase in sodium • Hypertonic Hyponatremia: hyperlipidemia, hyperproteinemia, hyperglycemia and low levels of sodium • Symptoms: lethargy, confusion,

Hypotonic Alteration: Water Excess • Almost impossible to produce TBW excess when body is functioning normally • Causes: Acute renal failure, CHF, cirrhosis, compulsive water drinking, decreased urine formation • Symptoms: – acute excess: confusion, convulsions – long-term excess: weakness, nausea, muscle twitching, headache, weight gain
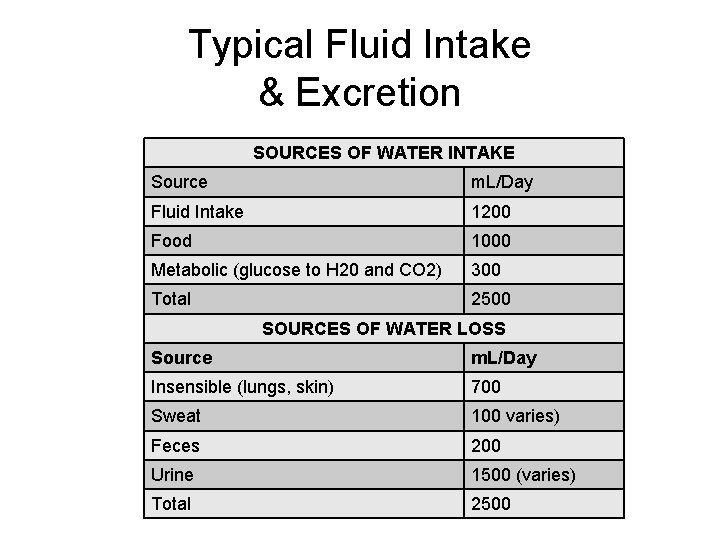
Typical Fluid Intake & Excretion SOURCES OF WATER INTAKE Source m. L/Day Fluid Intake 1200 Food 1000 Metabolic (glucose to H 20 and CO 2) 300 Total 2500 SOURCES OF WATER LOSS Source m. L/Day Insensible (lungs, skin) 700 Sweat 100 varies) Feces 200 Urine 1500 (varies) Total 2500

Potassium (K+) • Cells are rich in K+ • Distribution of K+ across the cell membrane involves electrochemical equilibrium • Alterations in intracellular and extracellular H 2 O or in extracellular K+ will affect the distribution of K+ and the membrane potential – This has important effects on electrically active cells (nerves and muscles)

Potassium (K+) • ECF: K+ concentration 3. 5 - 5. 5 m. Eq/L (commit to memory) • ICF: 150 - 160 m. Eq/L • There is a greater K+ concentration inside cells. – Differences in concentration maintained by sodium-potassium adenosine triphosphatase pump. – K+ can be actively secreted by distal tubule of healthy kidneys and by the

Potassium Hypokalemia Hyperkalemia • K+ <3. 5 m. Eq/L in serum • Causes: low intake, K+ shifting into cells (metabolic or respiratory alkalosis), renal excretion, diuretics, laxative abuse, diarrhea • Symptoms: (mild losses) asymptomatic; (at about 3. 0 m. Eq/L) skeletal muscle • K+ > 5. 5 m. Eq/L in serum • Causes: increased intake, leak or shift of intracellular K+ to ECF (crushing injury, cytotoxic drugs), decreased renal excretion (renal failure, K+ sparing diuretics) • Symptoms: (mild) diarrhea, neuromuscular

Fluid and Electrolyte Therapies • Isotonic – 0. 9% Na. Cl (normal saline) – 5% Dextrose (D 5 W) – 5% Dextrose in 0. 225% Na. Cl (D 5 -1/4 NS) – Lactated Ringers • Hypertonic – 5% Dextrose in 0. 9% Na. Cl (D 5 -NS) – 5% Dextrose in 0. 45% Na. Cl (D 5 -1/2 NS) – 10% Dextrose (D 10 W) • Hypotonic

Discussion Questions… • You’re not thirsty, but you drink a liter of pure water. What kind of fluid shifts happen in the short term? • You run 10 miles on an 80°F day. What happens? • A 19 -year-old college student takes a dare in the dining hall to swallow the contents of a whole saltshaker. What happens? • You donate a pint of blood. What have you lost? What do you need to replace?
- Slides: 33