Fluid and Electrolytes Balance and Disturbance Copyright 2010

Fluid and Electrolytes: Balance and Disturbance Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins

Fluid and Electrolyte Balance • Necessary for life, homeostasis • Nursing role: help prevent, treat fluid, electrolyte disturbances Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins

Fluid • Approximately 60% of typical adult is fluid – Varies with age, body size, gender • Intracellular fluid • Extracellular fluid – Intravascular: the fluid within the blood vessels e. g. plazma – Interstitial: fluid surrounds the cell (e. g. lymph – Transcellular: cerebrospinal, pericardial, synovial, intraocular, pleural, sweat and digestive secretion • “Third spacing”: loss of ECF into space that does not contribute to equilibrium Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins

Electrolytes • Active chemicals that carry positive (cations), negative (anions) electrical charges – Major cations: sodium, potassium, calcium, magnesium, hydrogen ions – Major anions: chloride, bicarbonate, phosphate, sulfate, and proteinate ions • Electrolyte concentrations differ in fluid compartments Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins

Regulation of Fluid • Movement of fluid through capillary walls depends on – Hydrostatic pressure: exerted on walls of blood vessels – Osmotic pressure: exerted by protein in plasma • Direction of fluid movement depends on differences of hydrostatic, osmotic pressure Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins

Regulation of Fluid • Osmosis: area of low solute concentration to area of high solute concentration • Diffusion: solutes move from area of higher concentration to one of lower concentration • Filtration: movement of water, solutes occurs from area of high hydrostatic pressure to area of low hydrostatic pressure • Active transport: physiologic pump that moves fluid from area of lower concentration of one of higher concentration Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins

Active Transport • Physiologic pump that moves fluid from area of lower concentration to one of higher concentration • Movement against concentration gradient • Sodium-potassium pump: maintains higher concentration of extracellular sodium, intracellular potassium • Requires adenosine (ATP) for energy Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins

Question • Tell whether the following statement is true or false: • Osmosis is the movement of a substance from an area of higher concentration to one of lower concentration. Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins

Answer • False. • Rationale: Diffusion is the movement of a substance from an area of higher concentration to one of lower concentration. The concentration of dissolved substances draws fluid in that direction. Osmosis is the movement of fluid, through a semipermeable membrane, from an area of low solute concentration to an area of high solute concentration until the solutions are of equal concentration. Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins

Routes of Gains and Losses • Gain – Dietary intake of fluid, food or enteral feeding Ingested fluid (60%) and solid food (30%) – Metabolic water or water of oxidation (10%) – Parenteral fluids Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins

Routes of Gains and Losses (cont’d) • Loss – Kidney: urine output – Skin loss: sensible, insensible losses – Lungs – GI tract – Other – Urine (60%) and feces (4%) – Insensible losses (28%), sweat (8%) Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins
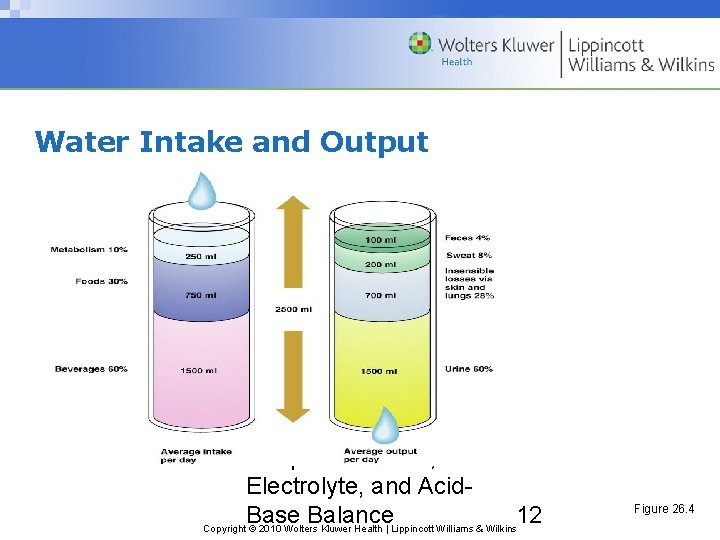
Water Intake and Output Chapter 26: Fluid, Electrolyte, and Acid. Base Balance 12 Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins Figure 26. 4

Question • What is the average daily urinary output in an adult? • 0. 5 L • 1. 0 L • 1. 5 L • 2. 5 L Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins

Answer • C. 1. 5 L • Rationale: Vital to the regulation of fluid and electrolyte balance, the kidneys normal filter 170 L of plasma every day in the adult, while excreting only 1. 5 L of urine. Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins

Regulation of Water Intake • The hypothalamic thirst center is stimulated: – By a decline in plasma volume of 10%– 15% – By increases in plasma osmolality of 1– 2% – Via baroreceptor input, angiotensin II, and other stimuli Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins

• Thirst is quenched as soon as we begin to drink water • Feedback signals that inhibit the thirst centers include: – Moistening of the mucosa of the mouth and throat – Activation of stomach and intestinal stretch receptors Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins

Regulation of Water Output • Obligatory water losses include: – Insensible water losses from lungs and skin – Water that accompanies undigested food residues in feces • Obligatory water loss reflects the fact that: – Kidneys excrete 900 -1200 m. Osm of solutes to maintain blood homeostasis – Urine solutes must be flushed out of the body in water 17 Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins

Influence and Regulation of ADH • Water reabsorption in collecting ducts is proportional to ADH release • Low ADH levels produce dilute urine and reduced volume of body fluids • High ADH levels produce concentrated urine • Hypothalamic osmoreceptors trigger or inhibit ADH release • Factors that specifically trigger ADH release include prolonged fever; excessive sweating, vomiting, or diarrhea; severe blood loss; and traumatic burns 18 Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins
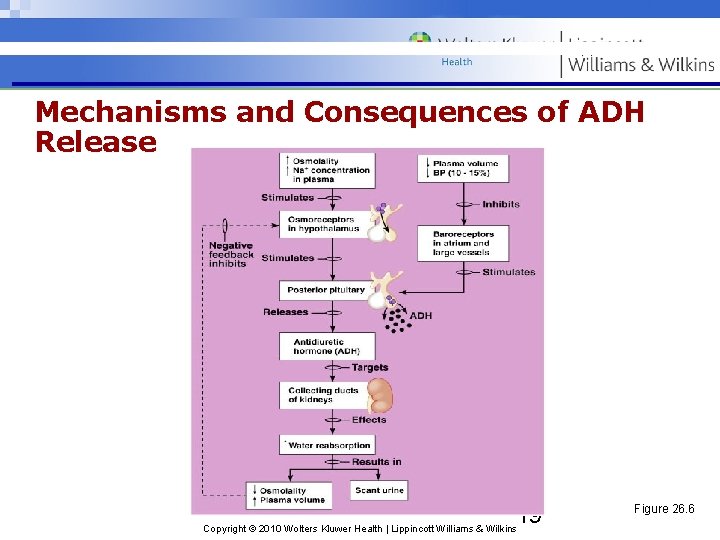
Mechanisms and Consequences of ADH Release 19 Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins Figure 26. 6
Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins

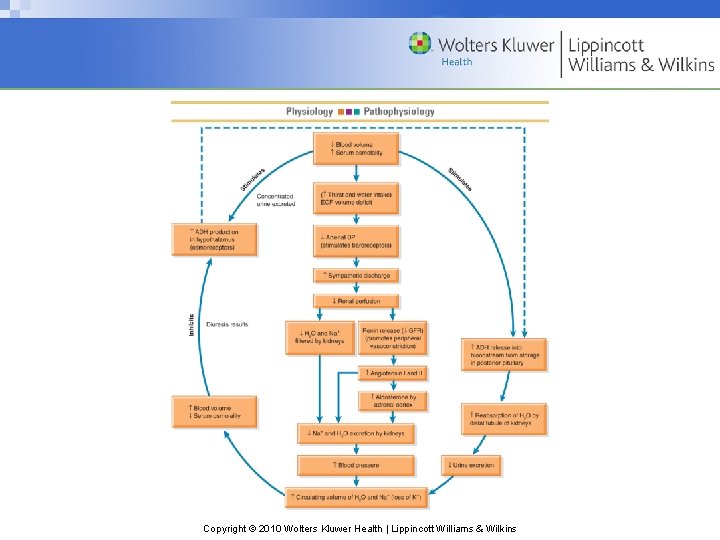
Fluid Volume Imbalances • Fluid volume deficit (FVD): hypovolemia • Fluid volume excess (FVE): hypervolemia Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins

Fluid Volume Deficit • Loss of extracellular fluid exceeds intake ratio of water – Electrolytes lost in same proportion as they exist in normal body fluids • Dehydration: loss of water along with increased serum sodium level – May occur in combination with other imbalances Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins

Fluid Volume Deficit (cont’d) • Dehydration – Causes: fluid loss from vomiting, diarrhea, GI suctioning, sweating, decreased intake, inability to gain access to fluid – Risk factors: diabetes insipidus, adrenal insufficiency, osmotic diuresis, hemorrhage, coma, Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins

Fluid Volume Deficit (cont’d) • Manifestations: rapid weight loss, decreased skin turgor, oliguria, concentrated urine, postural hypotension, rapid weak pulse, increased temperature, cool clammy skin due to vasoconstriction, lassitude, thirst, nausea, muscle weakness, cramps • Laboratory data: elevated BUN in relation to serum creatinine, increased hematocrit – Serum electrolyte changes may occur Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins

Fluid Volume Deficit (cont’d) • Medical management: provide fluids to meet body needs – Oral fluids – IV solutions (isotonic electrolytes solution; e. g. ringer lactate solution or 0. 9 sodium chloride) Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins

Fluid Volume Deficit - Nursing Management • I&O, VS • Monitor for symptoms: skin and tongue turgor, mucosa, UO, mental status • Measures to minimize fluid loss • Oral care • Administration of oral fluids • Administration of parenteral fluids Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins

Question • What is a major indicator of extracellular FVD? • Full and bounding pulse • Drop in postural blood pressure • Elevated temperature • Pitting edema of lower extremities Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins
Answer • B. Drop in postural blood pressure • Rationale: FVD signs and symptoms include acute weight loss; decreased skin turgor; oliguria; concentrated urine; orthostatic hypotension due to volume depletion; a weak, rapid heart rate; flattened neck veins; increased temperature; thirst; decreased or delayed capillary refill; decreased central venous pressure; cool, clammy, pale skin related to peripheral vasoconstriction; anorexia; nausea; lassitude; muscle weakness; and cramps. Clinical manifestations of FVE result from expansion of the ECF and include edema, distended neck veins, and crackles (abnormal lung sounds). Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins

Fluid Volume Excess • Due to fluid overload or diminished homeostatic mechanisms • Risk factors: heart failure, renal failure, cirrhosis of liver • Contributing factors: excessive dietary sodium or sodiumcontaining IV solutions • Manifestations: edema, distended neck veins, abnormal lung sounds (crackles), tachycardia, increased BP, pulse pressure and CVP, increased weight, increased UO, shortness of breath and wheezing • Medical management: directed at cause, restriction of fluids and sodium, administration of diuretics Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins

Disorders of Water Balance: Edema • Atypical accumulation of fluid in the interstitial space, leading to tissue swelling • Caused by anything that increases flow of fluids out of the bloodstream or hinders their return • Factors that accelerate fluid loss include: – Increased blood pressure, capillary permeability – Incompetent venous valves, localized blood vessel blockage – Congestive heart failure, hypertension, high blood volume Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins

Edema • Hindered fluid return usually reflects an imbalance in colloid osmotic pressures • Hypoproteinemia – low levels of plasma proteins – Forces fluids out of capillary beds at the arterial ends – Fluids fail to return at the venous ends – Results from protein malnutrition, liver disease, or glomerulonephritis Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins

Edema • Blocked (or surgically removed) lymph vessels: – Cause leaked proteins to accumulate in interstitial fluid – Exert increasing colloid osmotic pressure, which draws fluid from the blood • Interstitial fluid accumulation results in low blood pressure and severely impaired circulation Chapter 26: Fluid, Electrolyte, and Acid. Base Balance 32 Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins

Fluid Volume Excess - Nursing Management • I&O and daily weights; assess lung sounds, edema, other symptoms; monitor responses to medicationsdiuretics • Promote adherence to fluid restrictions, patient teaching related to sodium and fluid restrictions • Monitor, avoid sources of excessive sodium, including medications • Promote rest • Semi-Fowler’s position for orthopnea • Skin care, positioning/turning Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins

Electrolyte Imbalances • Sodium: hyponatremia, hypernatremia • Potassium: hypokalemia, hyperkalemia • Calcium: hypocalcemia, hypercalcemia • Magnesium: hypomagnesemia, hypermagnesemia • Phosphorus: hypophosphatemia, hyperphosphatemia • Chloride: hypochloremia, hyperchloremia Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins

Hyponatremia • Serum sodium less than 135 m. Eq/L • Causes: adrenal insufficiency, water intoxication, SIADH (Syndrome of inappropriate ADH secretion) or losses by vomiting, diarrhea, sweating, diuretics • Manifestations: poor skin turgor, dry mucosa, headache, decreased salivation, decreased BP, nausea, abdominal cramping, neurologic changes • Medical management: water restriction, sodium replacement • Nursing management: assessment and prevention, dietary sodium and fluid intake, identify and monitor atrisk patients, effects of medications (diuretics, lithium) Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins

Hypernatremia • Serum sodium greater than 145 m. Eq/L • Causes: excess water loss, excess sodium administration, diabetes insipidus, heat stroke, hypertonic IV solutions • Manifestations: thirst; elevated temperature; dry, swollen tongue; sticky mucosa; neurologic symptoms; restlessness; weakness • Note: thirst may be impaired in elderly or the ill • Medical management: hypotonic electrolyte solution or D 5 W • Nursing management: assessment and prevention, assess for OTC (Over the counter drugs) sources of sodium, offer and encourage fluids to meet patient needs, provide sufficient water with tube feedings Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins

Hypokalemia • Below-normal serum potassium (<3. 5 m. Eq/L), may occur with normal potassium levels with alkalosis due to shift of serum potassium into cells • Causes: GI losses, medications, alterations of acid-base balance, hyperaldosterism, poor dietary intake • Manifestations: fatigue, anorexia, nausea, vomiting, dysrhythmias, muscle weakness and cramps, paresthesias, glucose intolerance, decreased muscle strength and DTRs • Medical management: increased dietary potassium, potassium replacement, IV for severe deficit • Nursing management: assessment, severe hypokalemia is life-threatening, monitor ECG and ABGs, dietary potassium, nursing care related to IV potassium administration Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins

Hyperkalemia • Serum potassium greater than 5. 0 m. Eq/L • Causes: usually treatment related, impaired renal function, hypoaldosteronism, tissue trauma, acidosis • Manifestations: cardiac changes and dysrhythmias, muscle weakness with potential respiratory impairment, paresthesias, anxiety, GI manifestations • Medical management: monitor ECG, limitation of dietary potassium, cation-exchange resin (Kayexalate), IV sodium bicarbonate , IV calcium gluconate, regular insulin and hypertonic dextrose IV, -2 agonists, dialysis Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins

Hyperkalemia (cont’d) • Nursing management: assessment of serum potassium levels, mix IVs containing K+ well, monitor medication affects, dietary potassium restriction/dietary teaching for patients at risk • Hemolysis of blood specimen or drawing of blood above IV site may result in false laboratory result • Salt substitutes, medications may contain potassium • Potassium-sparing diuretics may cause elevation of potassium – Should not be used in patients with renal dysfunction Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins

Question • Tell whether the following statement is true or false: • The ECG change that is specific to hyperkalemia is a peaked T wave. Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins

Answer • True. • Rationale: The ECG changes that are specific to hyperkalemia are peaked T wave; wide, flat P wave; and wide QRS complex. The ECG changes that are specific to hypokalemia are flatted T wave and the appearance of a U wave. Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins

Hypocalcemia • Serum level less than 8. 5 mg/d. L, must be considered in conjunction with serum albumin level • Causes: hypoparathyroidism, malabsorption, pancreatitis, alkalosis, massive transfusion of citrated blood, renal failure, medications, other • Manifestations: tetany, circumoral numbness, paresthesias, hyperactive DTRs, Trousseau’s sign, Chovstek's sign, seizures, respiratory symptoms of dyspnea and laryngospasm, abnormal clotting, anxiety Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins

Hypocalcemia (cont’d) • Medical management: IV of calcium gluconate, calcium and vitamin D supplements; diet • Nursing management: assessment, severe hypocalcemia is life-threatening, weight-bearing exercises to decrease bone calcium loss, patient teaching related to diet and medications, and nursing care related to IV calcium administration Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins

Trousseau’s Sign Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins

Hypercalcemia • Serum level above 10. 5 mg/d. L • Causes: malignancy and hyperparathyroidism, bone loss related to immobility • Manifestations: muscle weakness, incoordination, anorexia, constipation, nausea and vomiting, abdominal and bone pain, polyuria, thirst, ECG changes, dysrhythmias • Medical management: treat underlying cause, fluids, furosemide, phosphates, calcitonin, biphosphonates • Nursing management: assessment, hypercalcemic crisis has high mortality, encourage ambulation, fluids of 3 to 4 L/d, provide fluids containing sodium unless contraindicated, fiber for constipation, ensure safety Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins

Hypomagnesemia • Serum level less than 1. 8 mg/d. L, evaluate in conjunction with serum albumin • Causes: alcoholism, GI losses, enteral or parenteral feeding deficient in magnesium, medications, rapid administration of citrated blood; contributing causes include diabetic ketoacidosis, sepsis, burns, hypothermia • Manifestations: neuromuscular irritability, muscle weakness, tremors, athetoid movements, ECG changes and dysrhythmias, alterations in mood and level of consciousness • Medical management: diet, oral magnesium, magnesium sulfate IV Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins

Hypomagnesemia (cont’d) • Nursing management: assessment, ensure safety, patient teaching related to diet, medications, alcohol use, and nursing care related to IV magnesium sulfate • Hypomagnesemia often accompanied by hypocalcemia – Need to monitor, treat potential hypocalcemia • Dysphasia common in magnesium-depleted patients – Assess ability to swallow with water before administering food or medications Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins

Hypermagnesemia • Serum level more than 2. 7 mg/d. L • Causes: renal failure, diabetic ketoacidosis, excessive administration of magnesium • Manifestations: flushing, lowered BP, nausea, vomiting, hypoactive reflexes, drowsiness, muscle weakness, depressed respirations, ECG changes, dysrhythmias • Medical management: IV calcium gluconate, loop diuretics, IV NS of RL, hemodialysis • Nursing management: assessment, do not administer medications containing magnesium, patient teaching regarding magnesium containing OTC medications Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins

Hypophosphatemia • Serum level below 2. 5 mg/DL • Causes: alcoholism, refeeding of patients after starvation, pain, heat stroke, respiratory alkalosis, hyperventilation, diabetic ketoacidosis, hepatic encephalopathy, major burns, hyperparathyroidism, low magnesium, low potassium, diarrhea, vitamin D deficiency, use of diuretic and antacids • Manifestations: neurologic symptoms, confusion, muscle weakness, tissue hypoxia, muscle and bone pain, increased susceptibility to infection • Medical management: oral or IV phosphorus replacement • Nursing management: assessment, encourage foods high in phosphorus, gradually introduce calories for malnourished patients receiving parenteral nutrition Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins

Hyperphosphatemia • Serum level above 4. 5 mg/DL • Causes: renal failure, excess phosphorus, excess vitamin D, acidosis, hypoparathyroidism, chemotherapy • Manifestations: few symptoms; soft-tissue calcifications, symptoms occur due to associated hypocalcemia • Medical management: treat underlying disorder, vitamin. D preparations, calcium-binding antacids, phosphatebinding gels or antacids, loop diuretics, NS IV, dialysis • Nursing management: assessment, avoid highphosphorus foods; patient teaching related to diet, phosphate-containing substances, signs of hypocalcemia Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins

Hypochloremia • Serum level less than 96 m. Eq/L • Causes: Addison’s disease, reduced chloride intake, GI loss, diabetic ketoacidosis, excessive sweating, fever, burns, medications, metabolic alkalosis • Loss of chloride occurs with loss of other electrolytes, potassium, sodium • Manifestations: agitation, irritability, weakness, hyperexcitability of muscles, dysrhythmias, seizures, coma • Medical management: replace chloride-IV NS or 0. 45% NS • Nursing management: assessment, avoid free water, encourage high-chloride foods, patient teaching related to high-chloride foods Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins

Hyperchloremia • Serum level more than 108 m. Eq/L • Causes: excess sodium chloride infusions with water loss, head injury, hypernatremia, dehydration, severe diarrhea, respiratory alkalosis, metabolic acidosis, hyperparathyroidism, medications • Manifestations: tachypnea, lethargy, weakness, rapid, deep respirations, hypertension, cognitive changes • Normal serum anion gap • Medical management: restore electrolyte and fluid balance, LR, sodium bicarbonate, diuretics • Nursing management: assessment, patient teaching related to diet and hydration Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins

Maintaining Acid-Base Balance • Normal plasma p. H 7 -35 -7. 45: hydrogen ion concentration • Major extracellular fluid buffer system; bicarbonate-carbonic acid buffer system • Kidneys regulate bicarbonate in ECF • Lungs under control of medulla regulate CO 2, carbonic acid in ECF Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins

Maintaining Acid-Base Balance (cont’d) • Other buffer systems – ECF: inorganic phosphates, plasma proteins – ICF: proteins, organic, inorganic phosphates – Hemoglobin Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins

Question • What is the most common buffer system in the body? • Plasma protein • Hemoglobin • Phosphate • Bicarbonate-carbonic acid Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins

Answer • D. Bicarbonate-carbonic acid • Rationale: The body’s major extracellular buffer system is the bicarbonate–carbonic acid buffer system, which is assessed when arterial blood gases are measured. Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins

Metabolic Acidosis • Low p. H <7. 35 • Low bicarbonate <22 m. Eq/L • Most commonly due to renal failure • Manifestations: headache, confusion, drowsiness, increased respiratory rate and depth, decreased blood pressure, decreased cardiac output, dysrhythmias, shock; if decrease is slow, patient may be asymptomatic until bicarbonate is 15 m. Eq/L or less • Correct underlying problem, correct imbalance – Bicarbonate may be administered Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins

Metabolic Acidosis (cont’d) • With acidosis, hyperkalemia may occur as potassium shifts out of cell • As acidosis is corrected, potassium shifts back into cell, potassium levels decrease • Monitor potassium levels • Serum calcium levels may be low with chronic metabolic acidosis – Must be corrected before treating acidosis Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins

Metabolic Alkalosis • High p. H >7. 45 • High bicarbonate >26 m. Eq/L • Most commonly due to vomiting or gastric suction – May also be due to medications, especially long-term diuretic use • Hypokalemia will produce alkalosis • Manifestations: symptoms related to decreased calcium, respiratory depression, tachycardia, symptoms of hypokalemia Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins

Metabolic Alkalosis (cont’d) • Correct underlying disorder, supply chloride to allow excretion of excess bicarbonate, restore fluid volume with sodium chloride solutions Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins

Respiratory Acidosis • Low p. H <7. 35 • Pa. CO 2 >42 mm Hg • Always due to respiratory problem with inadequate excretion of CO 2 • With chronic respiratory acidosis, body may compensate, may be asymptomatic – Symptoms may be suddenly increased pulse, respiratory rate and BP, mental changes, feeling of fullness in head Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins

Respiratory Acidosis (cont’d) • Potential increased intracranial pressure • Treatment aimed at improving ventilation Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins

Respiratory Alkalosis • High p. H >7. 45 • Pa. CO 2 <35 mm Hg • Always due to hyperventilation • Manifestations: lightheadedness, inability to concentrate, numbness and tingling, sometimes loss of consciousness • Correct cause of hyperventilation Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins

Arterial Blood Gases • p. H 7. 35 - (7. 4) - 7. 45 • Pa. CO 2 35 - (40) - 45 mm Hg • HCO 3ˉ 22 - (24) - 26 m. Eq/L – Assumed average values for ABG interpretation • Pa. O 2 80 to 100 mm Hg • Oxygen saturation >94% • Base excess/deficit ± 2 m. Eq/L Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins

Parenteral fluid therapy • Purpose: 1 - to provide water, electrolytes, and nutrition to meet daily requirements. 2 - to replace water and correct electrolytes imbalance 3 - to administer medication and blood products Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins

Types • Isotonic fluids: which have a total osmolality close to that of the ECF and does not effect on RBC. 1 lit of these solutions expand the ECF by 1 lit and the plasma by 0. 25 lit 1 - D 5 W: used for supply water and increase serum osmolality; don't administer it during fluid resuscitation bcz of hyperglycemia - About 1 lit of D 5 W provides fewer than 200 kcal 2 - normal saline: (0. 9%): used to correct the EC volume defect, within blood administration, to replace sodium losses in case of burn. - Not used for HF, PE, Renal impairment, sodium retention Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins

Types (cont…) 3 - other solutions: Lactated Ringers; contains potassium, calcium, and sodium chloride. - Used to correct dehydration, sodium depletion, and replace GI losses • Hypotonic fluids: used to replace cellular fluid, provide free water to excrete the body wastes, treat hypernatremia and other hyperosmolar conditions. • 1 - half strength saline (0. 45% sodium chloride) • SE of the excessive use of hypotonic solutions leads to intravascular fluids depletion, decrease BP, cellular edema, and cell damage Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins

• Hypertonic fluids: when normal saline and Lactated ringer’s contains 5% dextrose, the total osmolality exceed that the ECF • Other IV substances: When the patient is unable to tolerate food, nutritional requirements are often met using IV route - e. g. 50% dextrose in water. Protein, or fat, Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins

hypertonic • A solution that exerts a higher osmotic pressure than that of blood plasma. Administration of this fluid increases the solute concentration of plasma, drawing water out of the cells and into the extracellular compartment to restore osmotic equilibrium; cells will then shrink. • 1. D 5 W in normal saline solution. • 2. D 5 W in half normal saline solution (only slightly hypertonic • because dextrose is rapidly metabolized and renders only temporary osmotic pressure). Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins

• 3. Dextrose 10% in water. • 4. Dextrose 20% in water. • 5. 3% or 5% sodium chloride solution. • 6. Hyperalimentation solutions. • 7. D 5 W in lactated Ringer’s solution. • 8. Albumin 25%. Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins
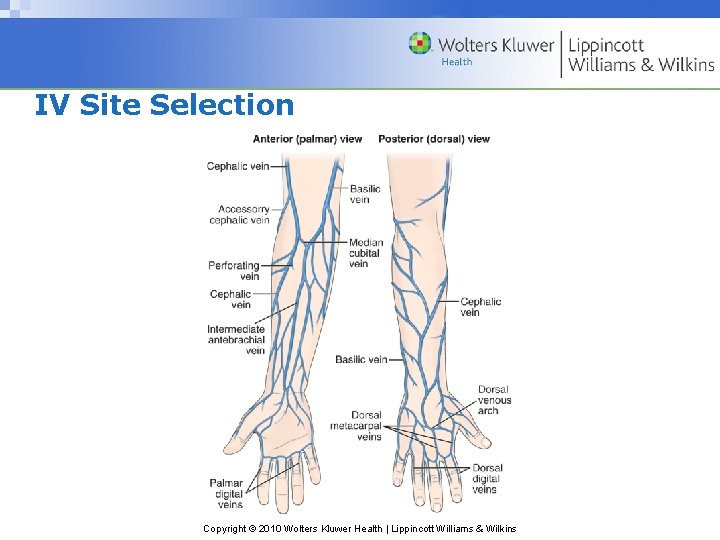
IV Site Selection Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins

Complications of IV Therapy • Fluid overload, Air embolism • Septicemia, other infections • Infiltration: is the unintentional administration of nonvesicant solutions or medication into surrounding tissue as a result of dislodges or perforates the wall of the veins , • extravasation: similar to Infiltration with an inadvertent administration of vesicant or irritant solution • Phlebitis • Thrombophlebitis • Hematoma • Clotting, obstruction Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins

Composition of Fluids • 1. Saline solutions—water and electrolytes (Na+, Cl−). • 2. Dextrose solutions—water or saline and calories. • 3. Lactated Ringer’s solution—water and electrolytes (Na+, K+, Cl−, Ca++, lactate). • 4. Balanced isotonic solution—varies; water, some calories, electrolytes(Na+, K+, Mg++, Cl−, HCO 3−, gluconate). Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins

• 5. Whole blood and blood components. • 6. Plasma expanders—albumin, mannitol, dextran, plasma protein fraction 5%, hetastarch; exert increased oncotic pressure, pulling fluid from interstitium into the circulation and temporarily increasing blood volume. • 7. Parenteral hyperalimentation—fluid, electrolytes, amino acids, and calories. Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins

Uses and Precautions with Common Types of Infusions • 1. D 5 W • a. Used to replace water (hypotonic fluid) losses, supply • some caloric intake, or administer as carrying solution for numerous medications. • b. Should be used cautiously in patients with water intoxication (hyponatremia, syndrome of inappropriate antidiuretic hormone release). Should not be used as concurrent solution infusion with blood or blood components. Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins

• 2. Normal saline solution • a. Used to replace saline (isotonic fluid) losses, administer with blood components, or treat patients in hemodynamic shock. • b. Should be used cautiously in patients with isotonic volume excess (heart failure, renal failure). • 3. Lactated Ringer’s solution • a. Used to replace isotonic fluid losses, replenish specific electrolyte losses, and moderate metabolic acidosis. Use cautiously in patients with liver failure. Copyright © 2010 Wolters Kluwer Health | Lippincott Williams & Wilkins
- Slides: 76