FLUID AND ELECTROLYTE BALANCE Prof ZCAN GKE MD

FLUID AND ELECTROLYTE BALANCE Prof. ÖZCAN GÖKÇE, MD Director of the Department of General Surgery Yeditepe University Hospital

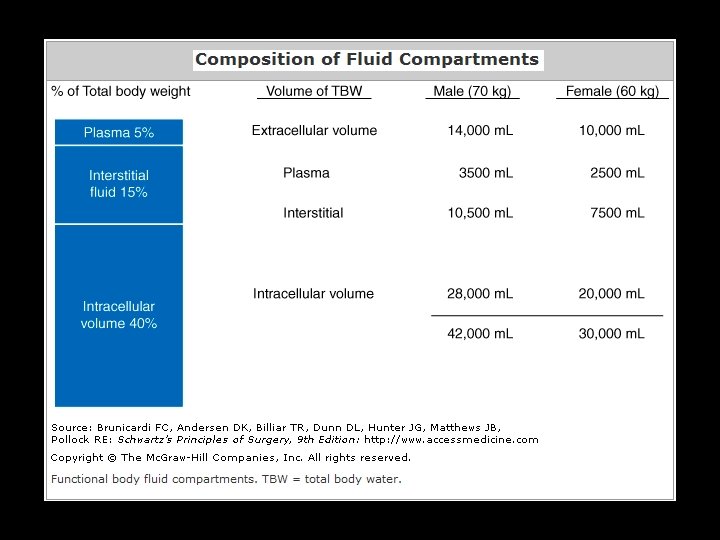
Chemical and biologic activities of fluid 1) Total particle count in per unit volume of solution (mmol/L) 2) Electrically active particles in per unit volume of solution (m. Eql/L) 3) Osmotically active particles in per unit volume of solution (m. O/L)

Osmolality is a measure of the osmoles of solute per kilogram of solvent (osmol/kg) The osmole (Osm or osmol) is a non-SI unit of measurement that defines the number of moles of a chemical compound that contribute to a solution's osmotic pressure. Osmolarity is the measure of solute concentration (osmol/L)
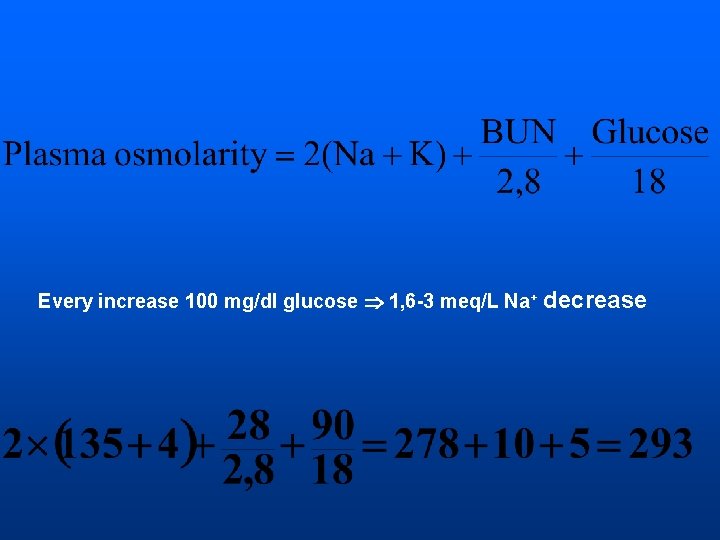
Every increase 100 mg/dl glucose 1, 6 -3 meq/L Na+ decrease

Henderson-Hasselbach equation
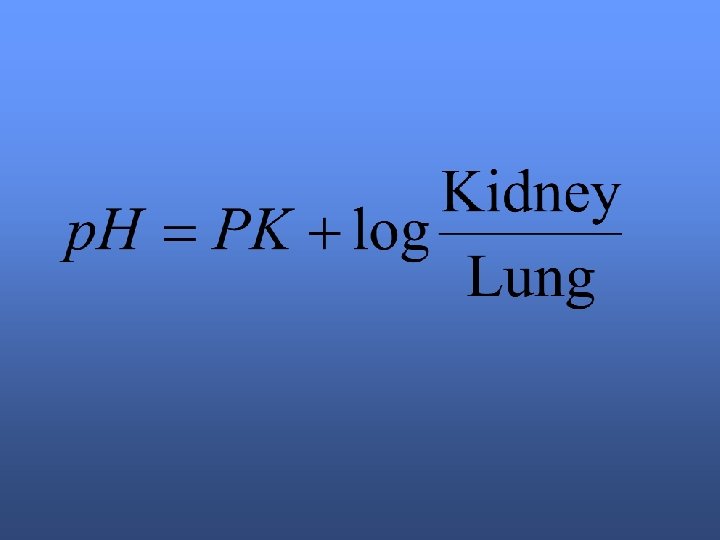

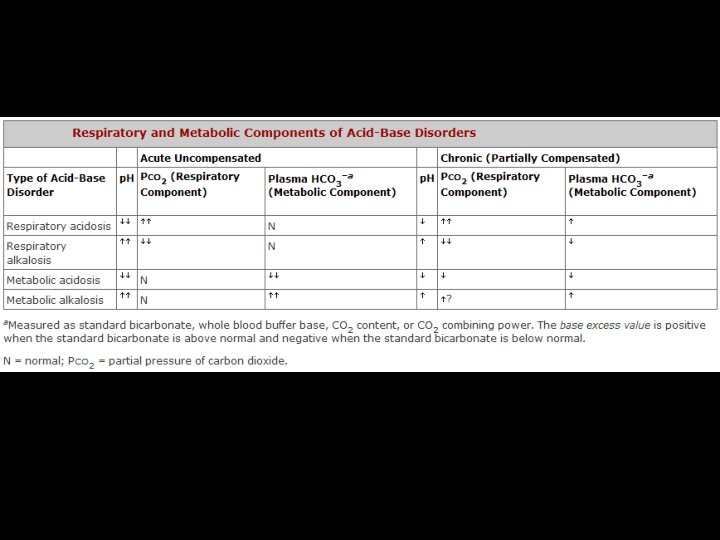
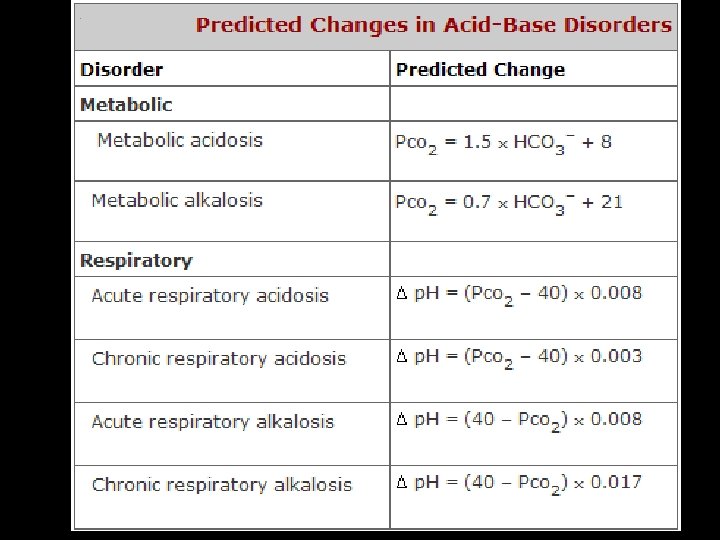
p. CO 2= 37 -42 mm Hg HCO 3 - = 22 -28 meq/L p. H=7. 34 -7, 44
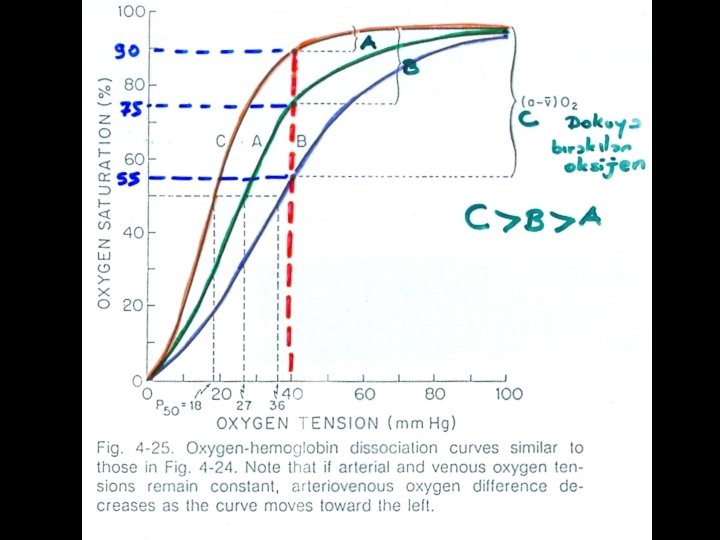

P 50 • it is common to use the concept of P 50 to describe the affinity of a given hemoglobin for oxygen. • The P 50 is the PO 2 at which the hemoglobin becomes 50% saturated with oxygen. • As the P 50 decreases, oxygen affinity increases and visa versa. • Normal adult Hemoglobin A has a P 50 of 26. 5 mm Hg while • Fetal Hemoglobin F has a P 50 of 20 mm Hg and • sickle cell anemia Hemoglobin S has a P 50 of 34 mm Hg.

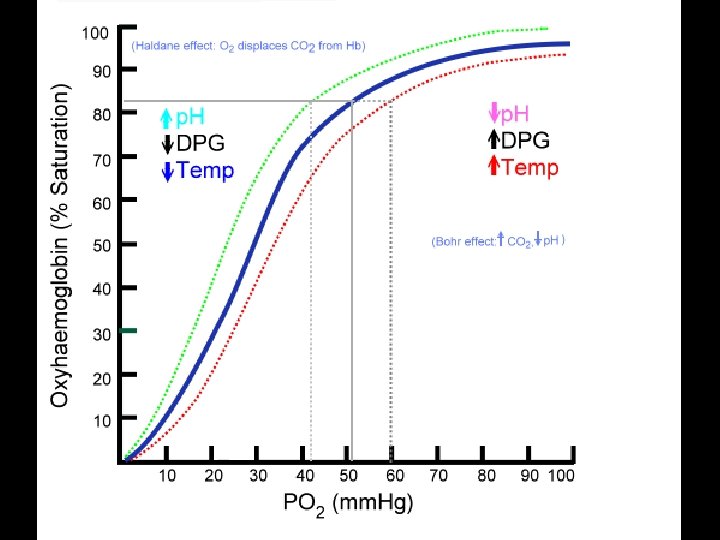
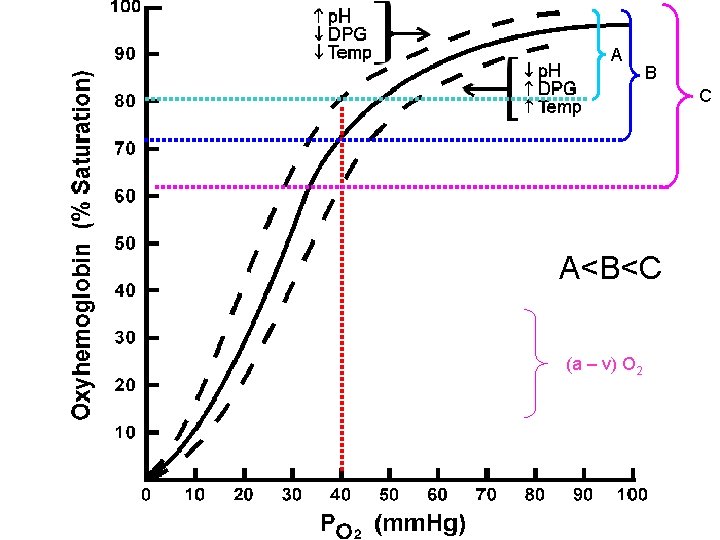
A B C A<B<C (a – v) O 2


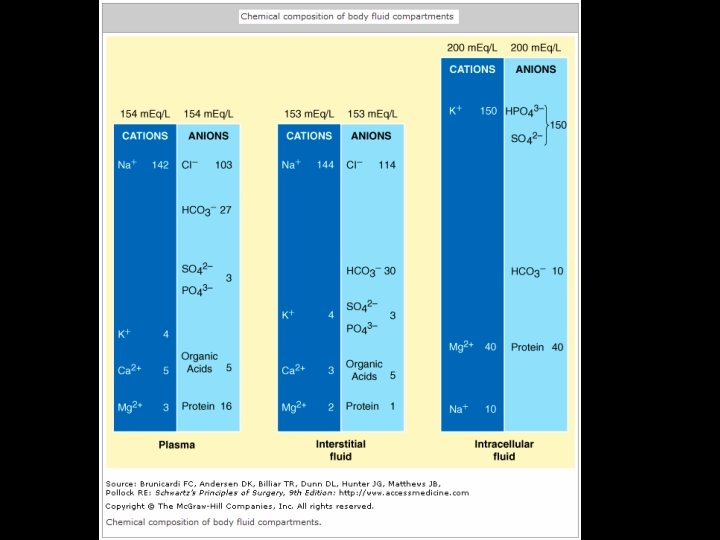
Anionic gap Measured cathions = Na, K Measured anions= HCO 3 - , Cl-

Anionic gap = Unmeasured anions-Unmeasured cathions Na+K + Unmeasured cathions = HCO 3 - + Cl- + Unmeasured anions Anionic gap = Na+K - ( HCO 3 - + Cl- ) Anionic gap = 140+4 - ( 28+ 104 ) Anionic gap = 12 Meq/L Correcred anionic gap = Actual anionic gap - {2, 5(4, 5 -Albumin)}
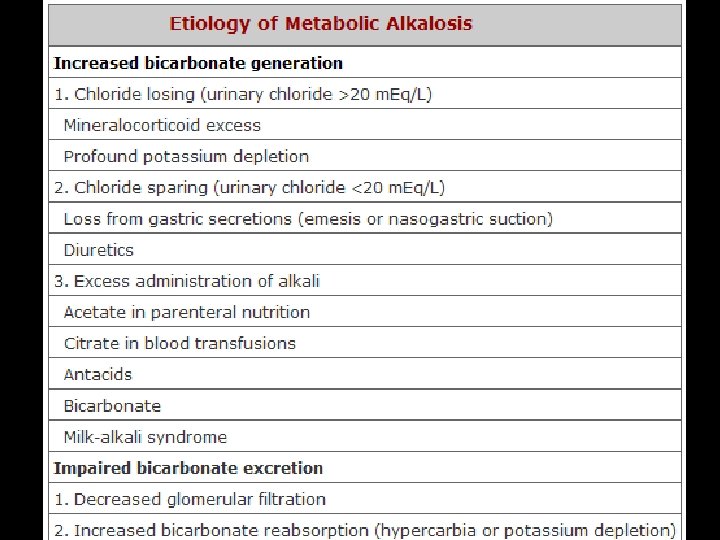
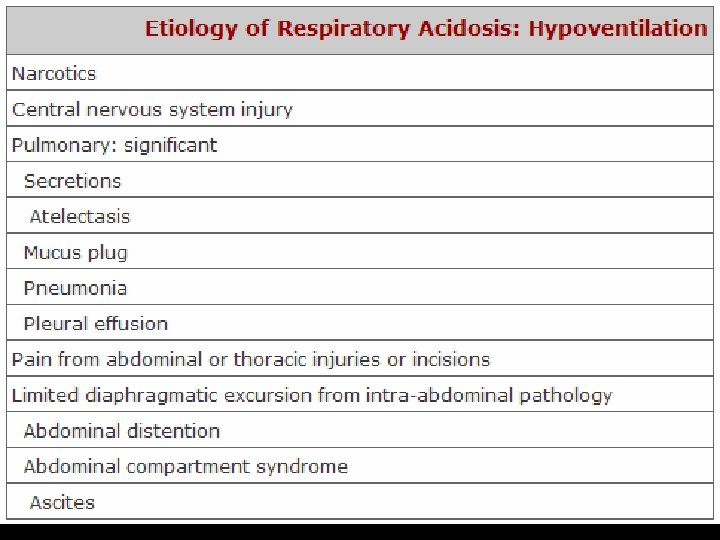
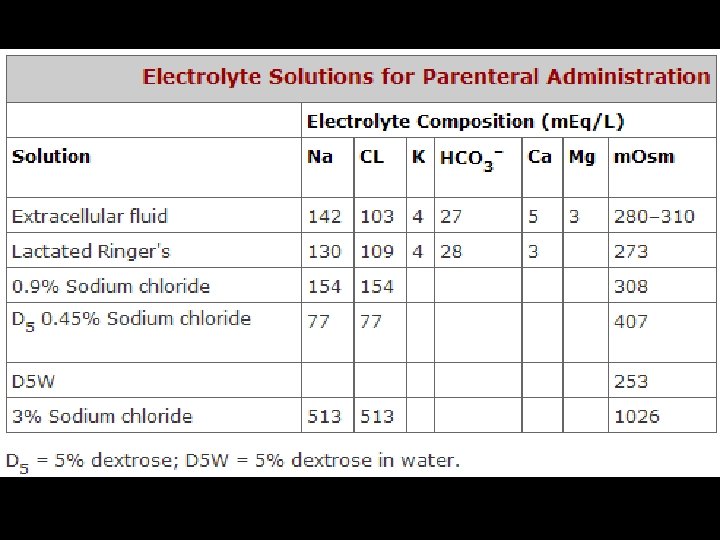
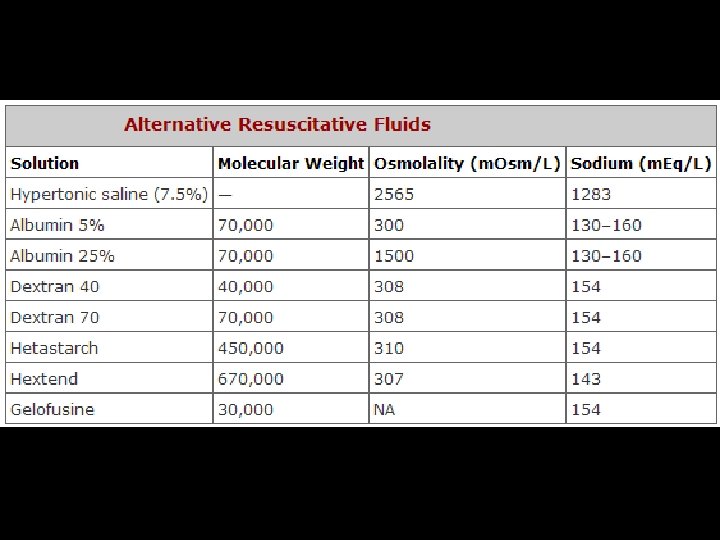

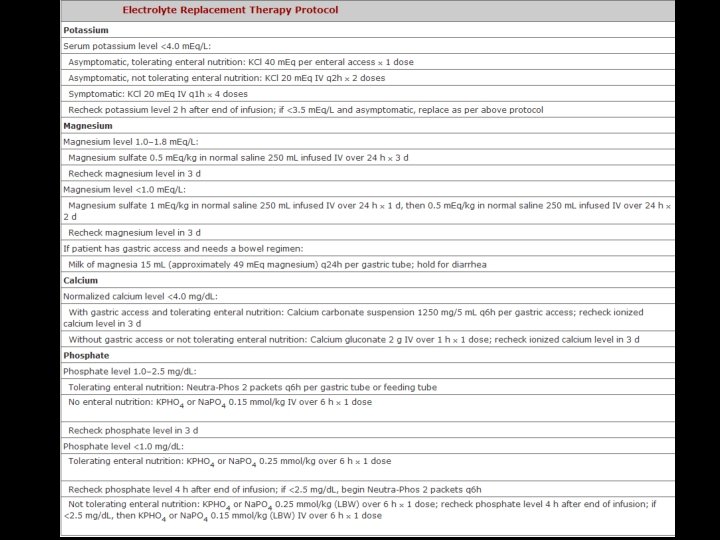

FLUID REPLACEMENT

PRACTICAL ISSUES 1 • Protein intake increases urea(Urine) and causes osmotic diuresis. • 1 gr protein needs 7 ml water

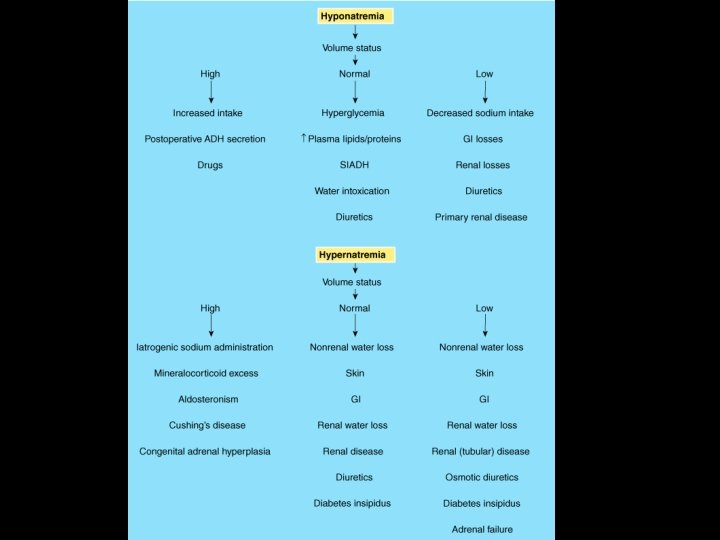
PRACTICAL ISSUES 2 • Treatment of Hypernatremia %5 dextroz, CVP and control of electrolytes

PRACTICAL ISSUES 3 • Treatment of Hyponatremia 135 - Na m. Eq/lt = A A*0, 5*Body weight = B %3 Na. Cl…… 518 m. Eq/lt B*1000 = C 518 C/2…. in 2 hours C/2…. in other 6 hours

PRACTICAL ISSUES 4 • Treatment of Acidosis 15 -CO 2 m. Eq/lt = A A*0, 3*Body weight = B B*1000 = C (1/6 M Lactate) 167 B*1000 = C (1/6 M Na. HCO 3) 178 C/2…. in 2 hours C/2…. in other 6 hours

PRACTICAL ISSUES 5 • Treatment of Hypopotasemia 3, 5 - K m. Eq/lt = A A*0, 2*Body weight = B B is not given more than 40 m. Eq/hour 40 m. Eq/liter Blood K should be measured every 2 hs K solutions contains 1 m. Eq/cc

PRACTICAL ISSUES 6 • Daily Na requirement is 2 -4 m. Eq/kg • Daily K requirement is Na/2 m. Eq/kg

THE END
- Slides: 41