FLUID AND ELECTROLYTE BALANCE AND DISORDERS LECTURER DR

FLUID AND ELECTROLYTE BALANCE AND DISORDERS LECTURER: DR. E. I. UGWUJA 1

Fluid and Electrolyte Balance §Water constitutes about 70% of total body weight of an adult male and about 60% in adult female. § Two-third (2/3) of TBW is inside the cells as intracellular fluid (ICF) §While the remaining one-third (1/3) is outside the cell and represents the extracellular fluid (ECF). §Of the ECF, ¾ is within the tissue and is referred to a interstitial fluid. §While the remaining ¼ represents the aqueous portion of the plasma. 2

• Currently, extracellular compartment has been recognised as heterogeneous and subdivided into four namely: 1. Plasma: Fluid within the heart and blood vessels. 2. Interstitial and lymph fluid: approximately fluid outside the cells. 3. Fluid of dense connective tissue, cartilage and bones: Fluid present in these tissues does not exchange fluid and electrolytes readily with remainder of body water. 4. Trancellular fluid: Fluid collections formed by the transport or secretory activities of cells; e. g. fluids in salivary gland, pancreas, liver, aqueous humour, CSF, etc. 3
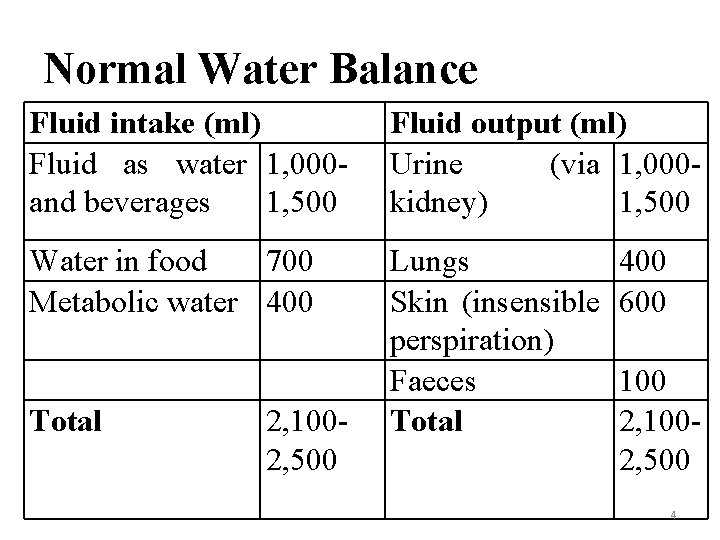
Normal Water Balance Fluid intake (ml) Fluid as water 1, 000 and beverages 1, 500 Fluid output (ml) Urine (via 1, 000 kidney) 1, 500 Water in food 700 Metabolic water 400 Lungs Skin (insensible perspiration) Faeces Total 2, 1002, 500 400 600 100 2, 1002, 500 4

OBLIGATORY LOSSES These are water losses from the skin, lungs, urine and GIT by Individuals over 24 hrs period under temperate condition to remain in water balance. Skin 600 ml Lungs 400 ml Gut 100 ml Kidneys 500 ml TOTAL 1600 ml 5

Minimum Excretory Volume. • Minimum amount of urine excretion required to rid the body of metabolic wastes in 24 hours. • If the intake of water is low or excessive water is lost through extra-renal route, the excretion of urine is diminished until only this amount is excreted. • It is usually between 500 -600 ml/24 hrs. • Minimum excretory volume is dependent on the concentrating power of the kidneys and the quantity of waste to be eliminated. 6

Regulation of water balance In health, the volume and composition of various body fluid compartments are maintained within physiological limits even in the face of wide variation in intake of water and solutes. 7

• Water balance in the body is controlled by the same mechanisms that are responsible for the constancy and tonicity of the ECF. These mechanisms include: • • • The effect of rennin-angiotesin-aldosterone. The effect of ADH on the collecting tubules of the kidneys. The thirst centre. Kinins. Atrial Natriuretic Peptide (ANP). 8

Renin-Angiotesin-Aldosterone Effect • In response to decreased RAP or plasma sodium, the JGA secretes into the blood increased amount of rennin. • Renin will convert angiotesinogen to angiotensin I. • Angiotensin I is thereafter converted to angiotensin II by angiotensin converting enzyme. • Angiotensin II is a very potent vaso-constrictor. • It acts on the adrenal cortex causing it to release aldosterone, which is responsible for absorption of water and sodium. • prolonged action of angiotensin II is halted by angiotesinestrase. 9

Antidiuretic Hormone (ADH) Effect • Further reabsorption of water is effected by ADH or Vasopressin at the collecting tubules of the kidneys. • The ADH is made in the hypothalamus and stored in the posterior pituitary. • The amount of the circulating hormone is regulated by osmoreceptors located in the hypothalamus. • These osmoreceptors are stimulated by increased plasma osmolarlity (concentration of solute in a particular solution) and low plasma volume. • ADH plays this role by increasing the diameter of the pores of the collecting tubules, thereby allowing increased amount of water to be osmotically reabsorbed into renal medulla. 10

The Thirst Centre • The thirst centre is located in the hypothalamus. • It is stimulated by either an increased osmotic pressure in the interstitial fluid or a decreased ECF volume. • Its stimulation prompt increased water consumption. 11

Role of Kinins • The main kinins that control blood pressure, ECF volume and metabolism of salt and water are Bradykinin and Kallidin. • Kinins are formed from inactive pre-enzyme called pre-kallikreins in response to increased ECF volume and damaged RBC. • Kinins have been found to cause increased excretion of salt (natriuresis) and water (dieresis). • Activities of kinins are modulated by prostaglandins. 12

Atrial Natriuretic Peptide (ANP) Action §It has been found that if the GFR and aldosterone secretion rate are kept constant, an increase in intravascular volume (IVV) will result in natriuresis. §This effect has been attributed to a polypeptide isolated from cardiac atrium ANP. §ANP has the following effect in vivo: • Increase in GFR • Increase in GFF • Causes natriuresis, dieresis and kaliuresis • Decreases rennin and aldosterone secretion • Decreases blood pressure. 13

DISORDERS OF FLUID METABOLISM q. Abnormalities of fluid metabolism are of two types viz: 1. Dehydration: Disturbance of water balance where output exceeds the intake, causing a reduction of body water. However, the clinical state to which it refers is more than this because, characteristically there is an accompanying disturbance of electrolytes. Hence dehydration may be the result of: (a). Pure water depletion (b). Pure salt depletion or (c). Mixed type in which both water and salt are depleted. 2. Water intoxication or overload 14

Pure Water Depletion • This is also called primary dehydration. • It occurs when water intake is stopped or water intake is inadequate without parallel loss of salt from the body. • Causes of primary dehydration include: (i). When patient is too weak or too ill to satisfy his/her water needs (ii). In mental patients who refuse to drink (iii). In comatose patient of patient with dysphagia (difficulty in swallowing) 15

Pathophysiology • Depletion occurs primarily due to inadequate intake and not as a result of water loss. • Obligatory water loss which continues even in the face of inadequate water intake and the body water becomes depleted. • Electrolyte concentration in the ECF to rise and the ECF becomes hypertonic or hyperosmolar. • Water then flows from ICC to the ECC along the osmotic gradient to correct the imbalance and to maintain uniform osmotic pressure throughout the body. • Thus the volume of ECF is maintained almost to normal at the expense of the ICF, which is grossly reduced in volume 16

• This causes intracellular dehydration characterized by: i). Thirst ii). Oliguria iii). Normal or slighthly increased blood urea iv). Normal of slightly reduced plasma volume v). Lack of circulatory collapse or fall in blood pressure vi). High urinary chloride vii). Death will occur when water loss is 15% of body weight (about 22% of total body water); about 7 -10 days of complete water deprivation. 17

Pure Salt Deprivation • Pure salt depletion occurs when fluids of high Na+ and Cl- are lost from the body and are replaced with salt-deficient fluids, such as water by mouth intra-venous glucose solution. • The term ‘pure salt depletion’ is now replace with a more appropriate term; ‘sodium depletion’ to reflect the importance of Na+ in the maintenance of ECF volume. 18

Causes of sodium depletion 1. Excessive sweating. 2. Loss of GIT fluid as in vomiting, diarrhea, fistulaes etc. 3. Increased urinary loss of Na+ as in Addison’s disease, DKA, chronic renal disease. 4. Vigorous diuretic use. 19
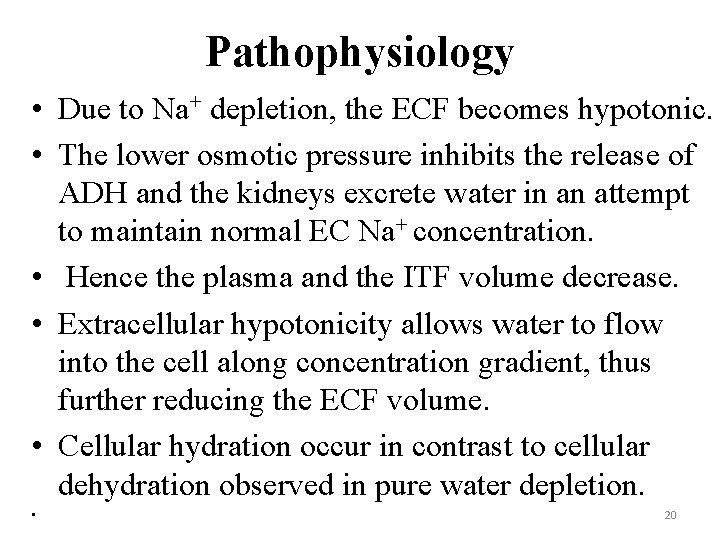
Pathophysiology • Due to Na+ depletion, the ECF becomes hypotonic. • The lower osmotic pressure inhibits the release of ADH and the kidneys excrete water in an attempt to maintain normal EC Na+ concentration. • Hence the plasma and the ITF volume decrease. • Extracellular hypotonicity allows water to flow into the cell along concentration gradient, thus further reducing the ECF volume. • Cellular hydration occur in contrast to cellular dehydration observed in pure water depletion. • 20

• Clinical and biochemical features of sodium depletion are mainly due to reduction in ECF volume and sodium deficiency and these include: • Lack of thirst • Apathy • Hallucinations • Anorxia/Nausea • Vomiting • Cramps • Sunken eyes and inelastic skin (due to loss of intestinal fluid) • Haemoconcentration • Low Bp • Increased plasma urea • Absence of urinary Na+ (Except in Addison’s disease) • Death is due to oligaemic shock. 21

Water Intoxication or Overload As with water depletion, water overload can be conveniently subdivided into: • Pure water overload and • isotonic water overload (water + plus an equivalent amount of Na ). 22

PURE WATER OVERLOAD • Water intoxication from excess intake is rare, since the capacity of a healthy individual to excrete a water load is considerable. • Once plasma tonicity falls by a few mosmol/kg. , ADH secretion ceases, and a large volume of dilute urine is excreted, until the excess water has been eliminated. • This process obviously requires intact renal function. • Failure to excrete a water load results in immediate dilutional hyponatraemia, and may lead to cerebral oedema, particularly when it develops acutely. 23

Causes of pure water overload 1. Water intake at a rate more than the kidney’s ability to excrete; generally >1 litre/hour. This occasionally arises as a mental disorder (psychogenic polydipsia) or in dedicated beer drinkers. 2. Water intake in patients with salt-losing forms of renal disease (‘salt-losing nephritis’) or those on diuretics that impair tubular salt reabsorption. 3. Syndrome of inappropriate ADH (SIADH), also known as Schwartz-Bartter syndrome, due to the failure of a low plasma osmolality to suppress ADH. 24

SIADH may be due to: • Intracranial pathology (head injury, haemorrhage, meningitis, encephalitis, or brain tumor), where there is direct stimulation of hypothalamic ADH release. • Pulmonary pathology (pneumonia, TB, assisted ventilation), where volume receptors in the pulmonary vascular bed falsely report a message of vascular depletion to the hypothalamus. • Ectopic production of ADH by tumors, particularly bronchial carcinomas. • Cortisol deficiency – since these hormones antagonize ADH, a deficiency of either will result in unopposed ADH action. • Pain, from trauma or surgery, stimulates ADH release. • Drugs, including psychoactive drugs (antidepressants, narcotics, carbamazepine), sulphonylureas, oxytocin for labour induction, vincristine for chemotherapy. 25

Findings in SIADH • Although patients with SIADH are over-hydrated, they are not frankly oedematous. • Retained water is shared between the ECF and ICF, and ECF expansion is eventually limited by release of atrial natriuretic factor (ANF) from cardiac atria, which promotes a natriuresis. • Major problem is hyponatraemia, leading to cerebral oedema and consequent depressed level of consciousness. • The more rapid the onset of hyponatraemia, the more severe is the cerebral oedema, since slow onset allows the brain time to decrease its osmolyte content. • Urine is typically inappropriately concentrated despite low plasma osmolality, and its [Na+] is high in the face of hyponatraemia. 26

ISOTONIC FLUID OVERLOAD • In this condition, accumulation of water is accompanied by an equivalent retention of Na+. • Since there is no osmotic force to drive excess fluid into the cells, the ECF bears the entire brunt of the fluid overload. • It may manifest as hypertension, or peripheral and/or pulmonary oedema, but never with neurological manifestations from intra-cerebral water shifts, as seen in pure water overload. 27

Major causes of isotonic fluid 1. Administration of excess isotonic fluid, particularly to patients with impaired renal function. 2. Hyperaldosteronism, which is the inappropriate secretion of aldosterone despite an expanded ECF volume. It may be either primary or secondary. 28

ELECTROLYTES IN THE BODY • In the ECF, the major cations are Na+ and K+ and the major anions are Cl- and HCO 3 -. • All the metabolic events are regulated to some degree by the relative and absolute concentrations of these electrolytes. • The electrolytes are important determinant of osmolarlity, state of hydration of the body and the p. H of both the ICF and ECF. • In addition, membrane potentials and normal functioning of the neurons and muscle tissues are closely regulated by the concentration difference between the ECF and ICF electrolytes. • Traditional unit of measurement of these electrolytes is m. Eq/L, but it is now mmol/L. • For a monovalent electrolyte, 1 m. Eq/L is equal to 1 mmol/L. 29

DISTIBUTION OF ELECTROLYTES IN THE BODY SODIUM (Na+) • This is the major cation in the ECF. • Reference range between 135 -148 mmol/L. Normal dietary intake is in the range of 100 -350 mmol/day. • When plasma sodium is above 110 mmol/L, Na+ will be excreted in urine and Na+ is present in urine of every healthy individual. • If Na+ intake is less than 30 mmol/day, or when plasma/serum Na+ is below 110 mmol/L, Na+ will not be excreted in urine. • It has been found that the renal threshold for Na+ is 110 30. mmol/L

Hyponatraemia §Lower plasma sodum Causes include: • Diarrhea, • Nephritic syndrome • Addison disease (hypoadrenocorticism), • Metabolic acidosis with excretion of cations and increased anio • Diuretic therapy and in polyuric state. § Replacing water alone in water and Na+ loss. §When total body water is increasd; i. e. hypervolaemia. §In Schwatz-Bartter Syndrome. In that condition, hyponatraemia is due to water retention secondary to continuous secretion of ADH, which is not related to serum osmolarlity. 31

Hypernatraemia Elevated plasma Na. Causes include: • In conditions in which water is lost in excess of salt. Eg in profuse sweating, prolonged hyperpnia, diarrhea, renal disease and polyuric state. • Lack of sufficient water intake owing to lack of adequate thirst mechanism; e. g. coma and hypothalamic disease. • Cushing’s disease and in hyperaldosteronism. 32

Potassium (K+) §Potassium is the major IC cation. §Only about 2% of K+ is EC. § Reference range for potassium is 3. 0 -5. 5 mmol/L. §Daily intake ranged between 50 -150 mmol/day. §Daily urinary excretion is between 25 -120 mmol. §Unlike Na+, there is no renal threshold for K+ and it will be excreted in urine even in K+ depleted state. 33

Hypokalaemia Low plasma Potassium Hypokalaemia occurs in: • GIT fliud losses as in vomiting, diarrhea • Renal disease • Following diuretic therapy • In mineralocorticoid excess (aldosteronism) • Alkalaemia • The hypokalaemia associated with alkalaemia is due to exchange of K+ for IC H+ and increased exchange of K+ for Na+ in the DCT. 34

Hyperkalaemia Elevated plasma Potassium Hyperkalaemia is seen in: • Acute and sometimes chronic renal disease. • Renal tubular acidosis in which the exchange of Na+ for K+ and or H+ is impaired. • In extensive tissue injury. • In acidotic state. • Artifactual. • Prolonged application of tourniquet • Haemolysis 35

ØElevated and depressed serum/plasma K+ concentrations may have profound adverse effect on the neuromuscular system. Aparthy, weakness, paralysis are some of the consequences of hyper-hypo-kalaemia. Both hypo- and hyperkalaemia will also have adverse effect on the myocardium. Serious arrythmiasis may develop and death will follow. 36

Treatment of Hyperkalaemia • Parenteral or non-parenteral administration of K+. • Calcium infusion, which will then antagonize the effect of K+ on the cardiac tissues. • Na. HCO 3 infussion, which will cause movement of K+ into the cells. • Glucose infusion, which will stimulate insulin production resulting in IC sequestration of K+ and glucose. • Renal dialysis • Oral or rectal administration of resins, which will bind K+ thereby removing it from ECF. • The use of diuretics. 37

Chloride Ion (Cl-) §Cl- is the major EC anion. §Reference range is between 98 -110 mmo/L. §Daily urinary output is 110 -250 mmol. § Low serum value are encountered in the following conditions: • Prolonged vomiting. • Metabolic acidotic state. • Addison’s disease. • Salt loosing renal disease. 38

• High level of Cl- is found in the following: • Metabolic acidosis associated with prolonged diarrhea and loss of HCl. • In renal tubular disease in which there is decreased excretion of H+ and therefore a decreased reabsorption of HCO 3 -. • In some cases of hyperthyroid diseases 39

Bicarbonate Ion (HCO 3 ) • Bicarbonate ion is quantitatively the 2 nd most important anion in serum. Co 2 produced by cellular metabolism diffuse into the plasma. • Inside the plasma, it enters the RBC, where it combines with water in the presence of carbonic anhydrase to form carbonic acid (H 2 CO 3). • Carbonic acid dissociate under the catalysis of carbonic anhydrase to form H+ and HCO 3 - as shown below: 2 CO 2 + 2 H 2 O H 2 CO 3 CA CA 2 H 2 CO 3 H+ + HCO 340

• As the levels of HCO 3 - builds up, it will diffuse down the gradient into the plasma. • As it does so, Cl- will move into the RBC to maintain electrical neutrality. • The movement of Cl from the plasma into the cell is known as chloride shift. 41

Thanks for your attention 42
- Slides: 42