Flow Cell Batteries GROUP B MIRO KOSKINEN ANTTI

Flow Cell Batteries GROUP B: MIRO KOSKINEN ANTTI MYLLYNEN TIA LOHTANDER JOHANNA VALIO

Contents • Case study: Flow cell batteries • Active materials and durability of flow cell batteries • LCA of wind mill and recycling of vanadium

Case study: Flow cell batteries
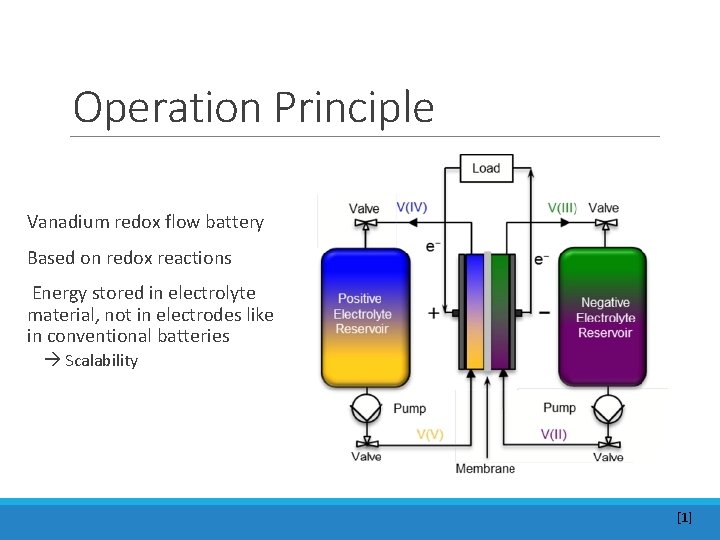
Operation Principle Vanadium redox flow battery Based on redox reactions Energy stored in electrolyte material, not in electrodes like in conventional batteries Scalability [1]

All Vanadium System In cell In stack ◦ Electrolyte ◦ compressing the cell stack ◦ active species V 2+/V 3+ & V 4+/V 5+ in sulphuric acid ◦ rubber gasket seals ◦ steel tie-bolts ◦ Membrane ◦ current conductors ◦ ion selective membrane, e. g. Nafion® ◦ Al, Cu plates ◦ Electrodes ◦ electrolyte compartments ◦ Carbon based: felt, composites etc. ◦ PTFE or other chemically resistant polymer In flow circuit ◦ storage tanks, pipes, pumps, control valves: ◦ materials the same as in chemical plants: acidious and corrosive environment [2, 3]

Material Restrictions Vanadium ◦ Scalability to terawatt level ◦ Low production volume Lithium Alternatives flow cells ◦ Zinc ◦ Bromine [4]

Material development Pros: + Scalability and flexibility +fast responsiveness + low environmental impact + only minor safety issues Cons: - low energy density - membrane fouling - cross contamination Alternatives: Zn-Br Zn-polyiodide All organic [5, 6, 7, 8]
![References [1] Peljo P. et al. All-Vanadium Dual Circuit Redox Flow Battery for Renewable References [1] Peljo P. et al. All-Vanadium Dual Circuit Redox Flow Battery for Renewable](http://slidetodoc.com/presentation_image_h2/7dd2aa41ea864bca8c284f0aef4ca5b8/image-8.jpg)
References [1] Peljo P. et al. All-Vanadium Dual Circuit Redox Flow Battery for Renewable Hydrogen Generation and Desulfurisation. Green Chemistry. 2016. [2] Tokuda N. et al. Development of a Redow Flow Battery System. Sei Technical Review. 2000. Vol. 50. 88 -94. [3] Leung P. et al. Progress in redox flow batteries, remaining challenges and their applications in energy storage. RSC Advances. 2012. Vol. 2. 10125 -10156. [4] Vesborg P. C. K. Addressing the terawatt challenge: scalability in the supply of chemical elements for renewable energy. RSC Advances. 2012. Vol. 2. P. 7933– 7947 [5] Alotto P. , Guarnieri M. , Moro F. Redox flow batteries for the storage of renewable energy: A review. Renewable and Sustainable Energy Reviews. 2014. Vol. 29. P. 325 -335. [6] Skyllas-Kazacos M. et at. Progress in Flow Battery Research and Development. Journal of The Electrochemical Society. 2011. Vol. 158: 8. P. R 55 -R 79. [7] Li B. et al. Ambipolar zinc-polyiodide electrolyte for a high-energy density aqueous redox flow battery. Nature. 2015. Vol. 6. P. 1 -8. [8] CHEM-E 4255 Electrochemical energy conversion. Lecture slides by David Lloyd. 2015.

Active materials and durability
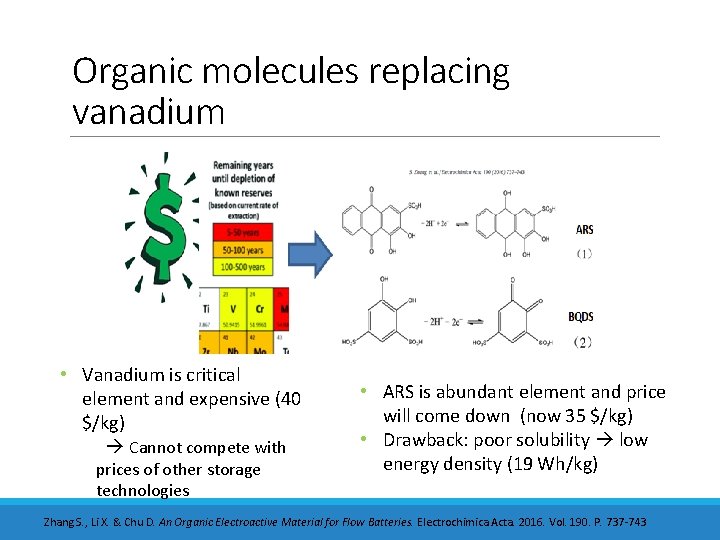
Organic molecules replacing vanadium • Vanadium is critical element and expensive (40 $/kg) Cannot compete with prices of other storage technologies • ARS is abundant element and price will come down (now 35 $/kg) • Drawback: poor solubility low energy density (19 Wh/kg) Zhang S. , Li X. & Chu D. An Organic Electroactive Material for Flow Batteries. Electrochimica Acta. 2016. Vol. 190. P. 737 -743
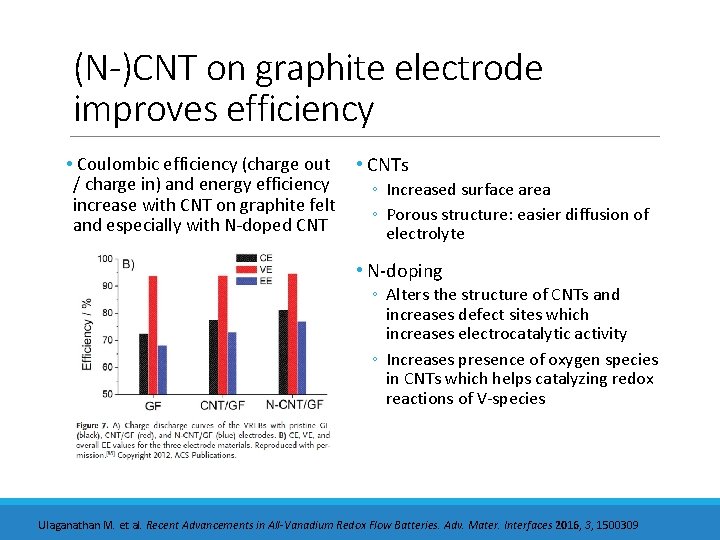
(N-)CNT on graphite electrode improves efficiency • Coulombic efficiency (charge out / charge in) and energy efficiency increase with CNT on graphite felt and especially with N-doped CNT • CNTs ◦ Increased surface area ◦ Porous structure: easier diffusion of electrolyte • N-doping ◦ Alters the structure of CNTs and increases defect sites which increases electrocatalytic activity ◦ Increases presence of oxygen species in CNTs which helps catalyzing redox reactions of V-species Ulaganathan M. et al. Recent Advancements in All-Vanadium Redox Flow Batteries. Adv. Mater. Interfaces 2016, 3, 1500309
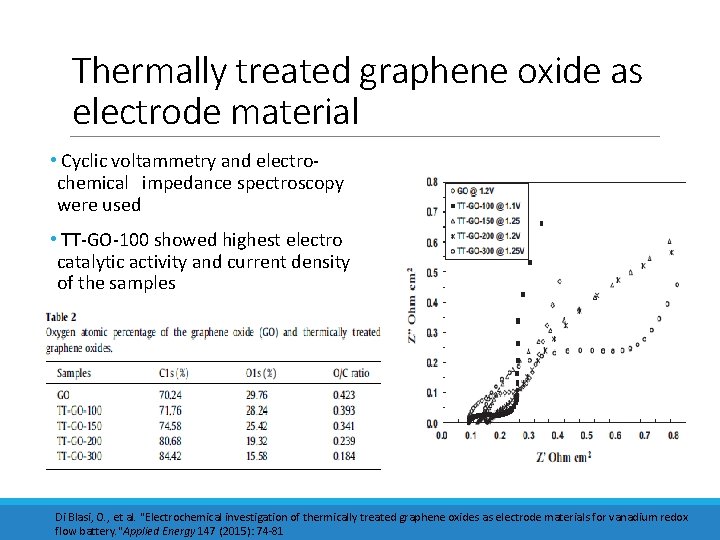
Thermally treated graphene oxide as electrode material • Cyclic voltammetry and electrochemical impedance spectroscopy were used • TT-GO-100 showed highest electro catalytic activity and current density of the samples Di Blasi, O. , et al. "Electrochemical investigation of thermically treated graphene oxides as electrode materials for vanadium redox flow battery. "Applied Energy 147 (2015): 74 -81
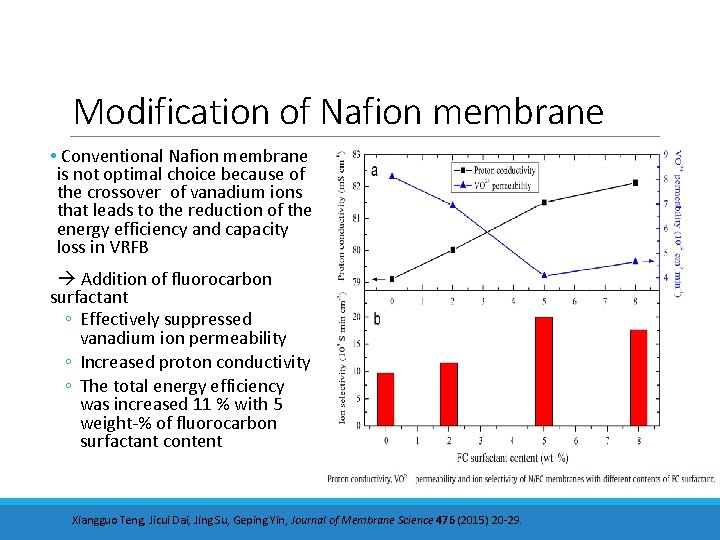
Modification of Nafion membrane • Conventional Nafion membrane is not optimal choice because of the crossover of vanadium ions that leads to the reduction of the energy efficiency and capacity loss in VRFB Addition of fluorocarbon surfactant ◦ Effectively suppressed vanadium ion permeability ◦ Increased proton conductivity ◦ The total energy efficiency was increased 11 % with 5 weight-% of fluorocarbon surfactant content Xiangguo Teng, Jicui Dai, Jing Su, Geping Yin, Journal of Membrane Science 476 (2015) 20 -29.
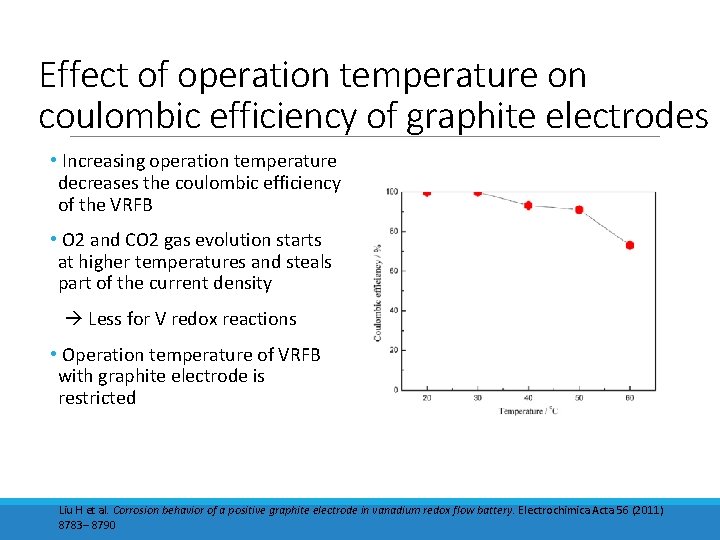
Effect of operation temperature on coulombic efficiency of graphite electrodes • Increasing operation temperature decreases the coulombic efficiency of the VRFB • O 2 and CO 2 gas evolution starts at higher temperatures and steals part of the current density Less for V redox reactions • Operation temperature of VRFB with graphite electrode is restricted Liu H et al. Corrosion behavior of a positive graphite electrode in vanadium redox flow battery. Electrochimica Acta 56 (2011) 8783– 8790
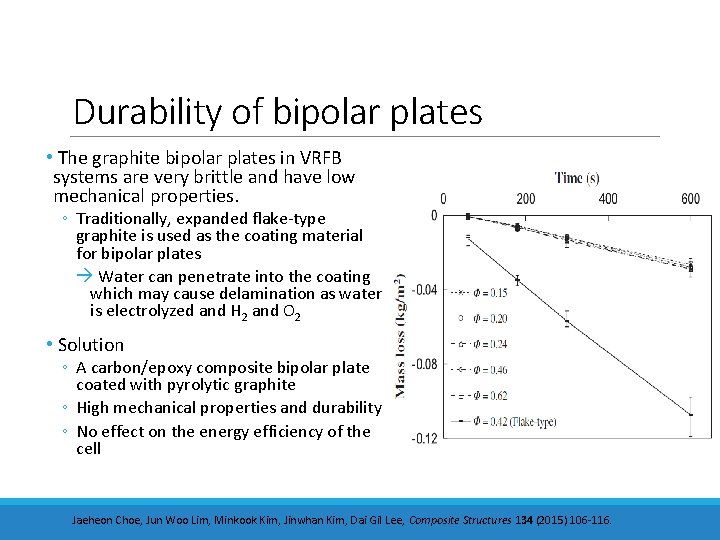
Durability of bipolar plates • The graphite bipolar plates in VRFB systems are very brittle and have low mechanical properties. ◦ Traditionally, expanded flake-type graphite is used as the coating material for bipolar plates Water can penetrate into the coating which may cause delamination as water is electrolyzed and H 2 and O 2 • Solution ◦ A carbon/epoxy composite bipolar plate coated with pyrolytic graphite ◦ High mechanical properties and durability ◦ No effect on the energy efficiency of the cell Jaeheon Choe, Jun Woo Lim, Minkook Kim, Jinwhan Kim, Dai Gil Lee, Composite Structures 134 (2015) 106 -116.
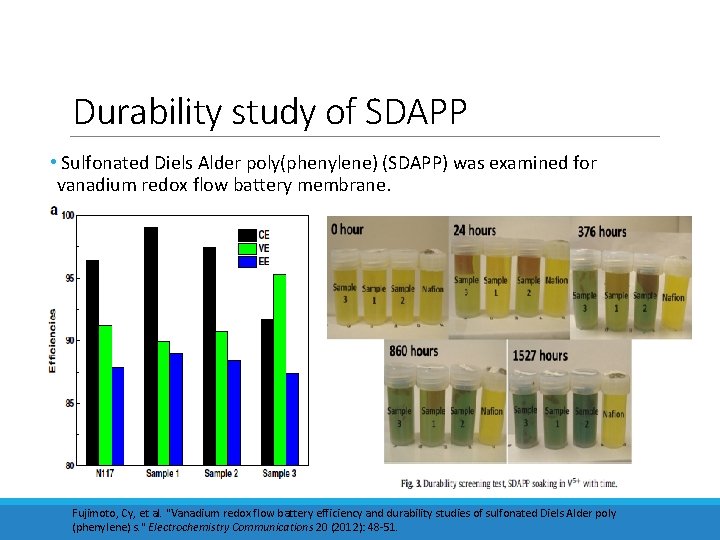
Durability study of SDAPP • Sulfonated Diels Alder poly(phenylene) (SDAPP) was examined for vanadium redox flow battery membrane. Fujimoto, Cy, et al. "Vanadium redox flow battery efficiency and durability studies of sulfonated Diels Alder poly (phenylene) s. " Electrochemistry Communications 20 (2012): 48 -51.

Increasing durability of Nafion membrane Solution Crossover process capacity losses • Layer-by-layer deposited PEI films improves durability of Nafion Crossover -70 %, oxygen permeation -13 % Improved coulombic efficiency +12 and energy efficiency +4 percentage points Increases life-time and efficiency of V/O₂ flow batteries Austing J. et al. Layer-by-layer modification of Nafion membranes for increased life-time and efficiency of vanadium/air redox flow batteries. Journal of Membrane Science. 2016.

LCA of wind mill and recycling of vanadium

LCA of Floating Wind Turbine • Increased amount of construction materials Higher environmental impacts • Mostly from steel production BUT better wind conditions, better capacity factor • Similar environmental impacts as using non-floating turbines • Except toxic emissions • Energy payback time 5, 2 months Weinzettel J. et al. Life cycle assessment of a floating offshore wind turbine. Renewable Energy. 2008. Vol 34. doi: 10. 1016/j. renene. 2008. 04. 004

Recycling of Vanadium • Steel industry uses 93 % of all vanadium production ◦ Not recycled • Pyrometallurgical methods economically not possible • Hydrometallurgical methods under development ◦ Ecological and economical E. Worrell and M. A. Reuter. Handbook of recycling. Chapter 2 & 10. ISBN: 9780123964595
- Slides: 20