FLAVONOIDS BABY PART I CONTENTS Definition Properties Classification

FLAVONOIDS BABY, PART I.

CONTENTS Definition Properties Classification Isolation Uses

FLAVONOIDS • The flavonoids are polyphenolic compounds possessing 15 carbon atoms; two benzene rings joined by a linear three carbon chain having the carbon skeleton C 6 - C 3 - C 6 and they are the plant pigments and they are having polar nature and is solouble in methanol and water. • Flavonoids constitute one of the most characteristic classes of compounds in higher plants. Many flavonoids are easily recognised as flower pigments in most angiosperm families (flowering plants).
• • However, their occurence is not restricted to flowers but include all parts of the plant. They are secondary metabolite and effective in CNS disorders.

PROPERTIES OF FLAVONES Most flavones are yellow solids Most flavones are soluble in water, ethanol and dilute acids and alkalis. Flavones are precipitated by lead salt. With ferric chloride, flavones give either a dull green or a red brown colour.

In acidic medium, flavones are usually more highly coloured than the bases from which they are derived. In acidic medium flavones form oxonium salts which impart this colour. However these oxonium salts are very unstable in presence of water. The flavones differ in this respect from the anthocyanidins which give strong oxonium salts and are found as such in plants. Cl O - + OH Cl H + O + O OH}Cl Different structures of oxonium salts of flavones. Flavones exhibit two absorption bands: Band I 330 -350 nm and Band II, 250 -270
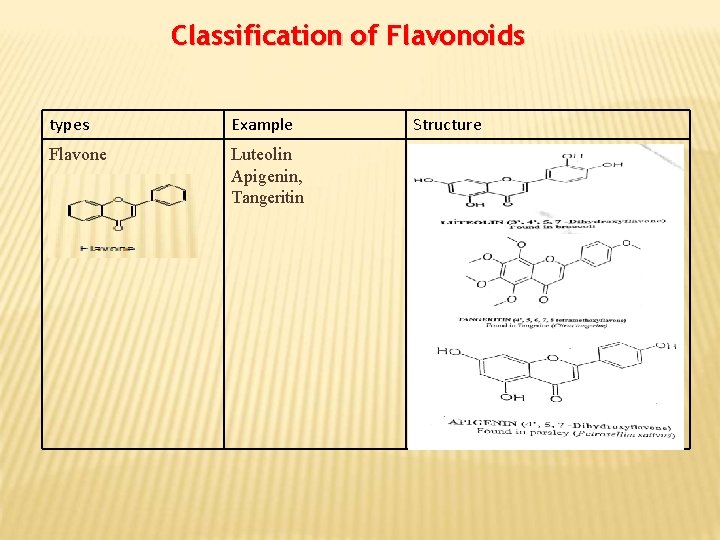
Classification of Flavonoids types Example Flavone Luteolin Apigenin, Tangeritin Structure
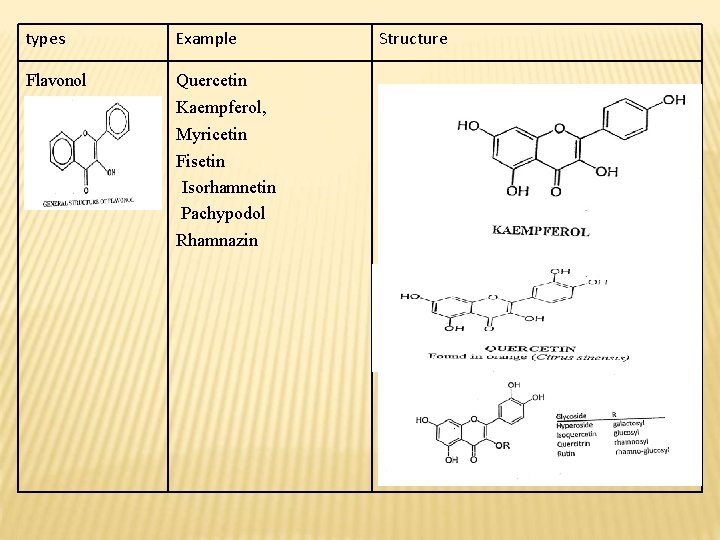
types Example Flavonol Quercetin Kaempferol, Myricetin Fisetin Isorhamnetin Pachypodol Rhamnazin Structure
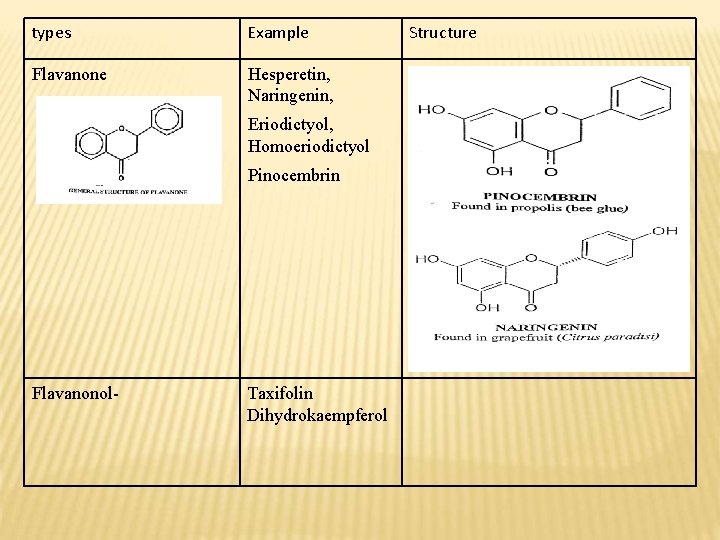
types Example Flavanone Hesperetin, Naringenin, Eriodictyol, Homoeriodictyol Pinocembrin Flavanonol- Taxifolin Dihydrokaempferol Structure
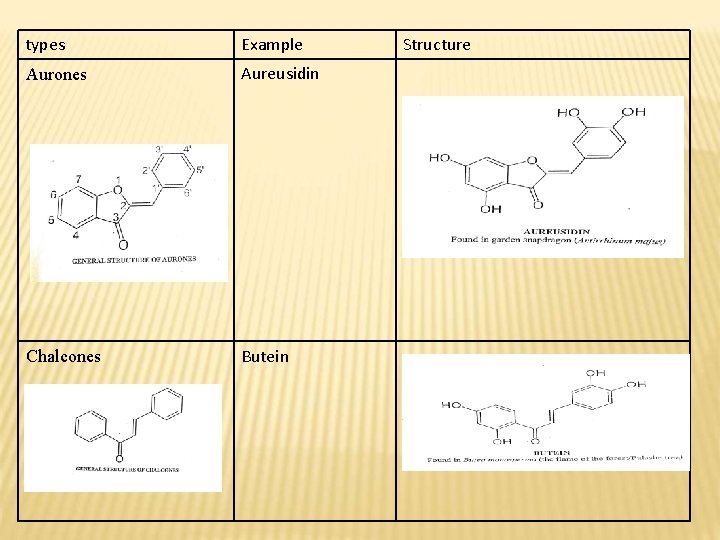
types Example Aurones Aureusidin Chalcones Butein Structure
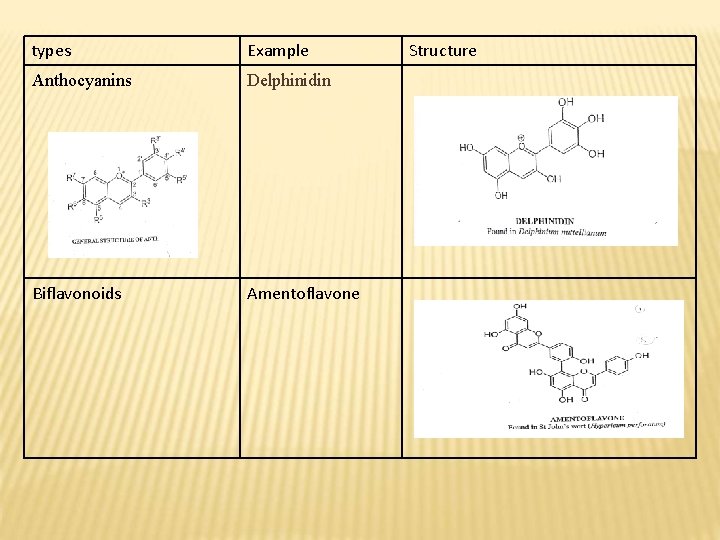
types Example Anthocyanins Delphinidin Biflavonoids Amentoflavone Structure

TESTS FOR FLAVONOIDS The extracts were dissolved in ethanol, filtered and subjected to following tests. Shinoda test: The dried extracts were dissolved in 95% ethanol (5 ml) and few drops of concentrated hydrochloric acid (HCL) were added. Then the magnesium turnings were put into the solution and observed for appearance of pink color. Lead acetate solution test: To small quantity of above residue, lead acetate solution was added and observed for appearance of formation of yellow colored precipitates.
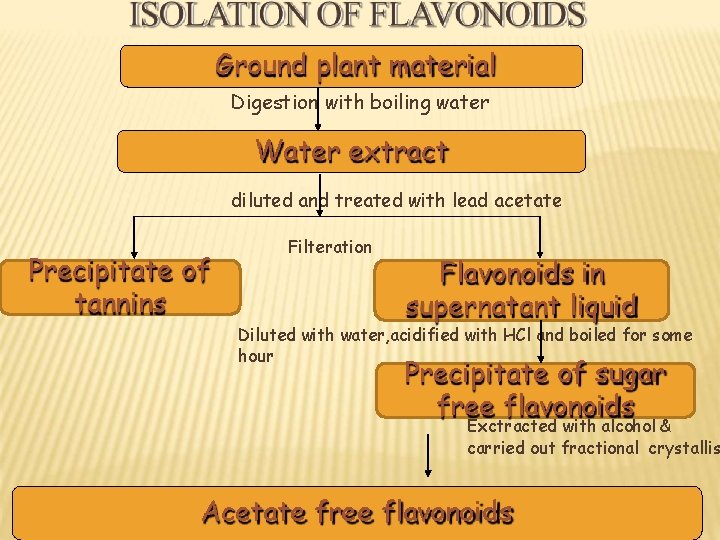
Ground plant material Digestion with boiling water Water extract diluted and treated with lead acetate Precipitate of tannins Filteration Flavonoids in supernatant liquid Diluted with water, acidified with HCl and boiled for some hour Precipitate of sugar free flavonoids Exctracted with alcohol & carried out fractional crystallis Acetate free flavonoids

SEPARATION & PURIFICATION OF FLAVONES PAPER CHROMATOGRAPHY Convenient means of separating and purifying flavones on milligram scale Dried plant material is extracted with either 70 % or 80 % methanol. The aqueous extract is then concentrated to a small volume in vacuo and refiltered if necessary. An aliquot of this concentrate should be applied on Whatman No. 3 filter paper.

Separation of the flavones present in concentrate is generally carried out in the solvent mixture BAW(n-butanol-acetic acidwater), 4: 1: 5 Individual bands are eluted and concentrated. Further fractioned in water, 5 % acetic acid. Purified in n-butanol-ethanol-water(4: 1: 2. 2)

THIN LAYER CHROMATOGRAPHY More sensitive method than paper chromatography. Layers of microcrystalline cellulose is employed. Solvent system same as that of PC. Removal of lipid impurities is essential otherwise considerable streacking may occur. Visualisation of plates may be done by viewing the plate in UV light(336 nm) either in the presence or absence of ammonia vapour. It is often assisted by the use of layers which contain a UV-Fluoroscent indicator. Flavonoids appear as dark spots against a fluoroscent green background. Another useful method of detection is brief exposure of the plate to iodine vapours which produce yellow-brown spots against white background with most flavonoids. Both methods are non destructive.

COLUMN CHROMATOGRAPHY Used for large scale separations. Adsorbents used include cellulose, celite, magnesolcelite, sililic acid, polyamide and sephadex. Polyamide is the widely used separation of the different flavone glycosides being achieved by gradient elution with water-methanol mixture. Recently separation of flavanol glycosides as their molybdate complexes on columns of Sephadex G-25 or LH-20 is employed. Elution with water followed by 1 M molybdate will separate mixtures of the common flavone glycosides on G-25. Alternatively simple mixtures of flavanol glycosides and aglycons can be separated on the 250 mg scale by adsorption on Sephadex I, H-20 and subsequent elution with methanol.

GAS LIQUID CHROMATOGRAPHY Not used extensively for the analysis & isolation of flavanoids. It is an acceptable method provided the flavanoid is derivatized to increase it volatility. Trimethylsilyl ether derivatives have been found most effective for this purpose, although methyl ether and acetate derivatives have also been used. The stationary phases, SE-30 and OV-1 are most commonly used for the separation of flavonoids.

Flavonoids Act like antioxidants. How effective they are depends on their molecular structural characteristics Some flavonoids in hops and beer have been found to have better antioxidant effects than tea or red wine; most flavonoids are found in fruits, vegetables, teas, and other drinks. Flavonoids have been known to have antiviral, antiallergic, antiplatelet, anti-inflammatory, antitumor and antioxidant activities

QUESTIONS LONG ESSAYS (10 M) üDefine flavonoids. Classify with examples. Discuss the general test for flavonoids. SHORT ESSAY (5 M) üDefine and classify flavonoids. Give the chemical tests for flavonoids. SHORT ANSWERS (2 M) üFlavonoids

- Slides: 21