FLASHBACK How many electrons do all atoms want

FLASHBACK • How many electrons do all atoms want in their outer shell? • How do these atoms get those e-’s in the outer shell? • How can you tell how many valence e-’s an atom has? • If atoms gain e-’s, they become ______ charged. • If atoms lose e-’s, they become ______ charged.

Unit 4 CHEMICAL REACTIONS!! Ch. 20/21

CHEMICAL VOCAB!!

WHY DO ATOMS BOND? To increase stability of the atoms Group 1 and 2 metals transfer electrons to Group 16 and 17 nonmetals to create ionic bonds. Metals lose electrons & nonmetals gain electrons to achieve noble gas structure!

BONDING A bond forms when… two atoms gain, lose, or share electrons in their outer energy levels. -1 Ca. Cl 2 Cl Cl -1 +2 Ca Calcium ion will give up its outer electrons to both chlorine atoms
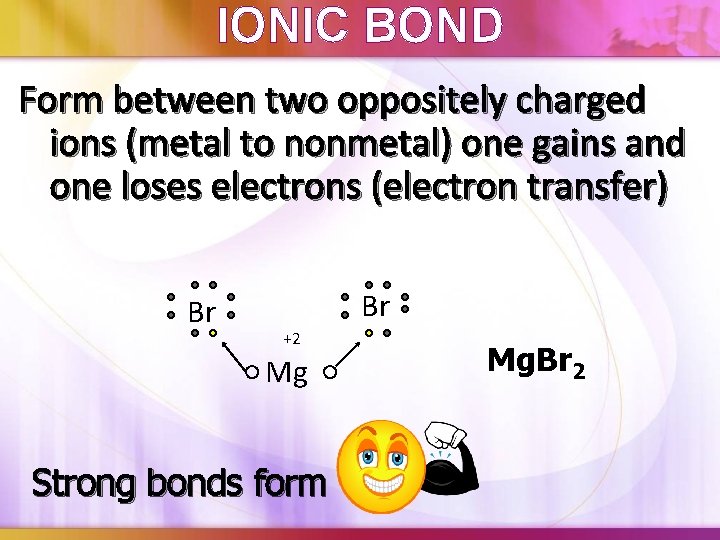
IONIC BOND Form between two oppositely charged ions (metal to nonmetal) one gains and one loses electrons (electron transfer) Br Br +2 Mg Strong bonds form Mg. Br 2

COVALENT BOND Form when atoms share one or more pairs of electrons = nonmetals to nonmetals Oxygen only needs two H 2 O H electrons so hydrogen shares its one valence electron so that both atoms fill their outer shells. Weaker bonds form Ionic vs. Covalent Video

ELECTRON DOT DIAGRAMS Shows only valence electrons of atom with dots around the element's symbol What’s the point?
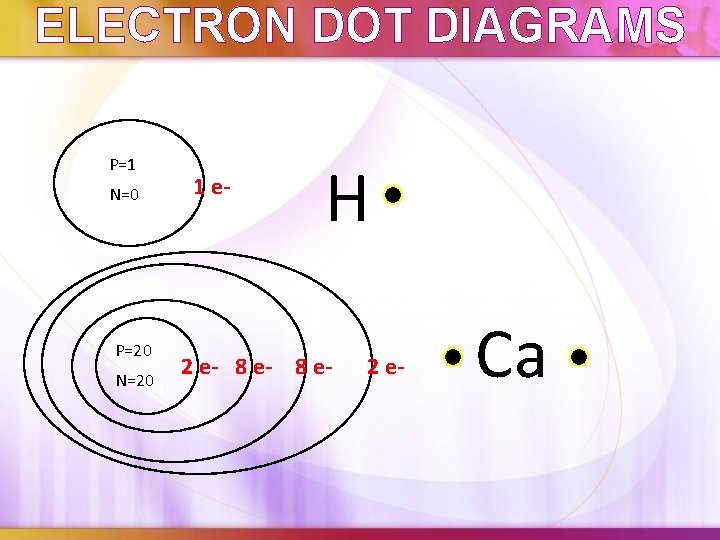
ELECTRON DOT DIAGRAMS P=1 N=0 P=20 N=20 1 e- H 2 e- 8 e- 2 e- Ca

ELECTRON DOT DIAGRAMS 1. 2. Determine # of valence electrons Correctly place the electrons around symbol Nitrogen: Strontium: Water: H 2 O N Sr

FLASHBACK 1. What types of elements occupy space in groups 1 and 2? 2. How many valence e-’s do these groups have, respectively? 3. What will their ionic charges be, respectively, if they lose their valence e-’s? 4. Metals will _______ e-’s. 5. Non-metals will ________ e-’s. 6. What e-’s do the bonding? 7. Explain difference between ionic and covalent bonds.

Bonding Worksheet: 5. Cl 4. Na 7. Be 11. 10. Ne Mg Covalent or ionic bond? 12. HCl_____________ 14. Al. O 3____________ Using electron dot diagrams, draw the ionic bonds… 17. Na. Cl

LAB TIME! Ionic vs. covalent. . . Which will win? ? ? Investigating the difference between sugar and salt! Sugar! Salt!

Bonding Rap… Who knew Chemical Bonding could be so cool? ? Want to give it a try? ? ? Extra Credit by the end of the 9 weeks (March 23 th) for a Chemistry Rap Video!

BONDS Covalent Structures: make molecules and SHARE ELECTRONS Example: H 20, CO 2, C 12 H 22 O 11 same charges, so no strong attraction Ionic Structures: make ionic crystals and TRANSFER ELECTRONS Example: Na. Cl, KI, Ca. F 2 + and – charges make a STRONG attraction!

IONIC STRUCTURE A. When positive and negative ions surround each other, they form tightly packed structures called ionic crystals or crystal lattices Substances with network (ionic) structures are usually strong solids with high melting and boiling points Substances made of molecules have lower melting and boiling points

ELECTRON DOT DIAGRAMS What happens if it is not the neutral element. . But the ion? ? e- diagram Na Cl Mg lose or gain e-? new (ion) e- diagram

SECTION 1 REVIEW Pg. 606 # 4, 5 CHAPTER REVIEW Pg. 626 # 4 -6, 11, 14, 17, 18

1. Which of the substances has the lower melting point (which melted faster)? Was this what you expected? Why or why not? 2. Relate your results to ionic and covalent bonding. 3. On a molecular level, how do the bonds in sugar and salt differ? 4. Why do atoms bond? 5. Water (H 2 O) contains a(n) _________ bond. Calcium oxide (Ca. O) contains a(n) ________ bond.

FLASHBACK EOC WORKBOOK!!! Pg. 38 (all) Pg. 39 (all) Pg. 36 (all) Pg. 37 (all)

FLASHBACK 1. Table salt (Na. Cl) has what kind of bond? 2. Table sugar (sucrose, C 12 H 22 O 11) has what kind of bond? 3. When heated, what happened to the bonds in sugar? 4. When heated, what happened to the bonds in salt? 5. Provide the electron dot structure for Barium (Ba), Iodine (I), and Cesium (Cs).

Review of Terms! 1. Cation: positively charged ion - Non metals or metals? 2. Anion: negatively charged ion - Non metals or metals?

Chemical Formula Terms 1. Symbol: element being used 2. Subscript: shows how many of the individual atoms are present ex. O 2 CO 2 HBr 3. Coefficient Number: placed before the element or compound, distributed to entire compound ex. 2 HBr or 2 H 2 + O 2 2 H 2 O 4. Oxidation Number: same as “charge”, how many electrons atoms need to gain or lose (to become stable) **get off of periodic table!! **write them on your P. T. (Al = +3)

Chemical Formulas The chemical symbols and numbers indicating the number of atoms contained in the basic unit of a substance C 6 H 12 O 6 Carbon = 6 atoms Hydrogen = 12 atoms Oxygen = 6 atoms How many atoms of each element are present in 3 molecules of glucose? 3 C 6 H 12 O 6 C=18 H=36 O=18

Writing Ionic Formulas The elements overall charges have to equal zero Compounds have no net charge!! Write the cation first with its charge, then the anion and its charge. Al+3 F-1 Write the chemical formula, using subscripts to indicate how many of each ion are needed to make a neutral compound. criss-cross method: Al+3 F-1 = Al. F 3

MORE COMPOUNDS!! § Balance the formula so the compound formed has a neutral charge Examples: § Ca+2 + Br – 1 § Na+1 + S – 2 § Al +3 + Cl-1


NAMING IONIC COMPOUNDS Ionic compounds are formed by the strong attractions between cations and anions. Both ions are important to the compound’s structure, so it makes sense that both ions are included in the name 1. Name cation first (metal) 2. Name anion (nonmetal) 3. Drop the end of the anion & add suffix –ide EXAMPLE: Na. Cl = sodium chloride
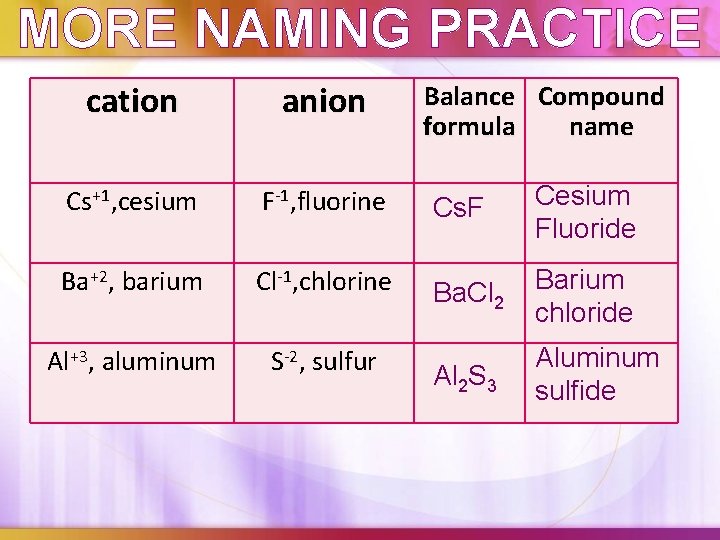
MORE NAMING PRACTICE cation anion Cs+1, cesium F-1, fluorine Cs. F Cesium Fluoride Ba+2, barium Cl-1, chlorine Ba. Cl 2 Barium chloride Al+3, aluminum S-2, sulfur Al 2 S 3 Aluminum sulfide Balance Compound formula name

NAMING COVALENT COMPOUNDS § Uses prefixes!!!! If there is only one atom of the first element, it does not get a prefix EX: BF 3= boron trifluoride Dihydrogen monoxide = ? ? Number of atoms 1 2 3 4 5 6 7 8 9 10 prefix Mono. Di. Tri. Tetra. Penta. Hexa. Hepta. Octa. Nona. Deca-
- Slides: 30