FLAME TEST INTRODUCTION A number of common metal

FLAME TEST

INTRODUCTION • A number of common metal ions (Li+, Na+, K+, Ca 2+, Ba 2+, Sr 2+, and Cu 2+) give a distinct color to a flame. Therefore, a flame test is often used as a confirmatory test in identifying an unknown metal. • Compounds of these ions provide the beautiful colors in a fireworks display. When glass is melted in a Bunsen burner flame, sodium ions color the flame. A copper wire inserted into the flame often results in a striking flame color. While the light emitted from a few excited metal ions is beautiful, in the laboratory a simple flame test is often very helpful in identifying an unknown metal ion. • In this experiment you will observe and record the flame colors of several metals.

PURPOSE • To experiment with flame tests on different salts. • To predict the identity of an unknown metal ion from a flame test.

SAFETY • Goggles • Hair tied back • Remain with bunsen burner at all times • Perform only authorized tests

MATERIALS • Salt solutions • Goggles • Bunsen burner • QTip • 11 test tubes/test tube rack

PROCEDURES 1 st Period 6 th Period • Clean 11 test tubes for your table and place them in a test tube rack • Place 1 m. L of each solution into each of the test tubes • CAUTION: Before you use the burner in the next procedures, check to see that long hair and loose clothing have been confined. • Dip the tip of the clean QTip into one of the solutions, and then hold it just above the inner flame of the Bunsen burner. • Observe the color of the flame just above the QTip. Heat only the tip of the QTip (burning the wooden piece may cause you to misinterpret your flame color!). • Record the color of the flame in the Data Table on the next page. • Test each of the remaining solutions and record the observed color in your data table. • Clean up test tubes (remove tape and wash) • Use a new QTip each trial to make sure you do not to contaminate your samples.

BARIUM

CALCIUM

COPPER

LEAD

LITHIUM

MAGNESIUM • If you are lucky you may see it! (it makes a white flame)

POTASSIUM
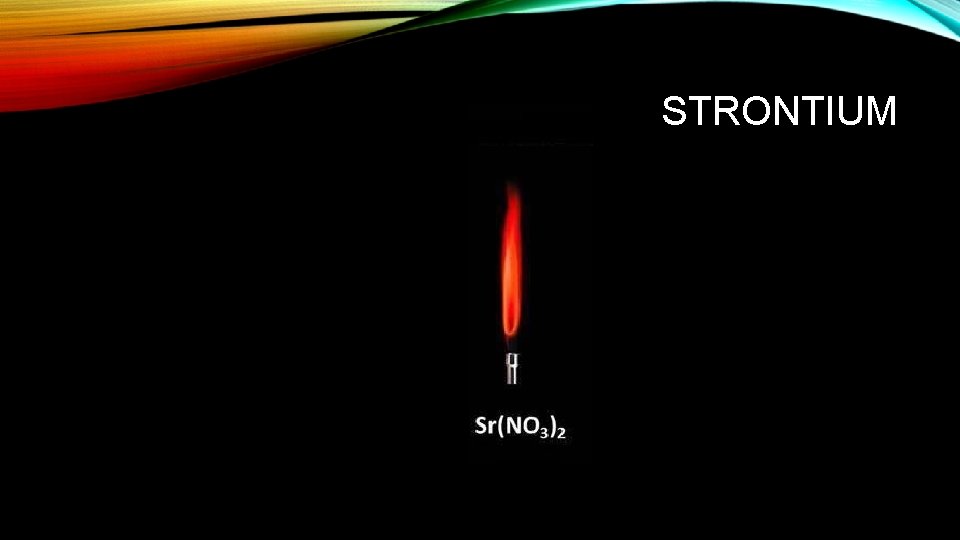
STRONTIUM

SODIUM
- Slides: 15