Flame Test Electron Configuration Warm Up What is
















- Slides: 42

Flame Test, Electron Configuration

Warm - Up • • What is a photon? How can we identify elements based on their emission spectrum? What is the Photoelectric Effect? Why do we say that light has a dual nature?

Objective • Today I will be able to: – Analyze the emission of energy in metals by completing a flame test – Calculate the electron configuration for elements on the periodic table

Homework • Electron Configuration Practice

Agenda • • • Warm – Up Flame Test Lab Electron Configuration Notes Electron Configuration Practice Exit Ticket

Electron Configuration

Electron Configuration • Electron configuration – arrangement of electrons in an atom • Electrons try to populate the lowest energy levels available – “ground state”

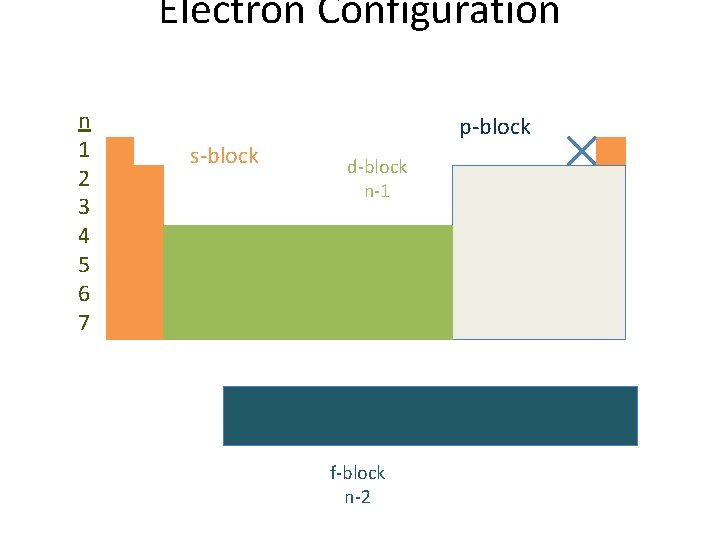
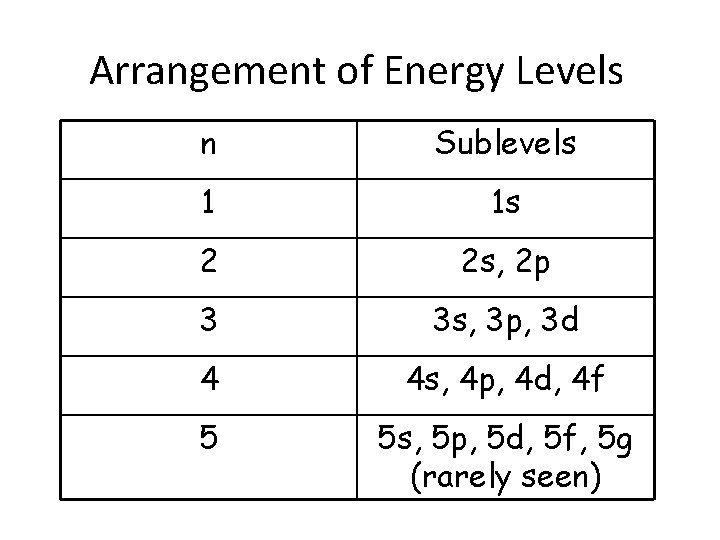
Arrangement of Energy Levels • Principal energy levels – represented by n • As atoms get bigger, there are more electrons, therefore more energy levels • Within each energy level, we have one or more sublevels • These sublevels are made up of orbitals

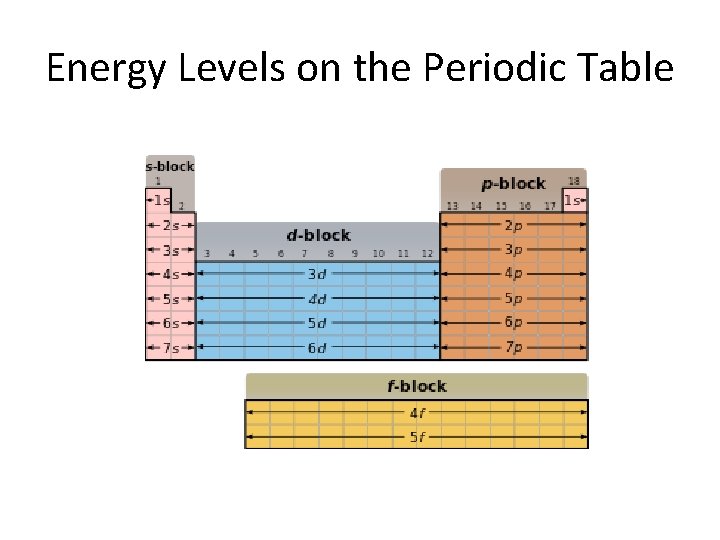
Energy Levels on the Periodic Table

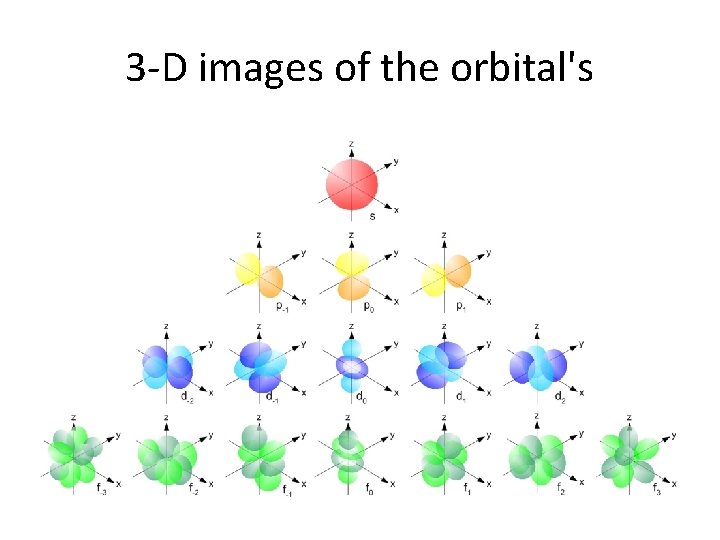
Orbitals • According to modern atomic theory electrons are found in orbitals • Orbital – 3 -D region around the nucleus indicating the probable location of the electron • There are 4 types of orbitals where the electrons can be found – s, p, d, f • Order of energy from lowest to highest: – s, p, d, f

3 -D images of the orbital's

Energy Sublevels • Each sublevel occupies a specific location on the periodic table:

Electron Configuration n 1 2 3 4 5 6 7 s-block p-block d-block n-1 f-block n-2

Arrangement of Energy Levels n Sublevels 1 1 s 2 2 s, 2 p 3 3 s, 3 p, 3 d 4 4 s, 4 p, 4 d, 4 f 5 5 s, 5 p, 5 d, 5 f, 5 g (rarely seen)

Orbitals cont. • Each sublevel can hold a specific amount of electrons – S=2 – P=6 – D=10 – F=14

Electron Configurations • We can write the electron configurations for each element (assuming the atom has a neutral charge) representing the location of the elements electrons in the energy levels

Examples • Sodium (Na) – Has 11 electrons (when neutral) – Electron Configuration: • 1 s 22 p 63 s 1 • Cobalt (Co) – Has 27 electrons (when neutral) – Electron Configuration • 1 s 22 p 63 s 23 p 64 s 23 d 7
Why does 4 s come before 3 d? • 3 d is of slightly higher energy than 4 s, but 4 s is further from the nucleus

z y The 3 axes represent 3 -dimensional space x

z y The nucleus of the atom is at the center of the three axes x

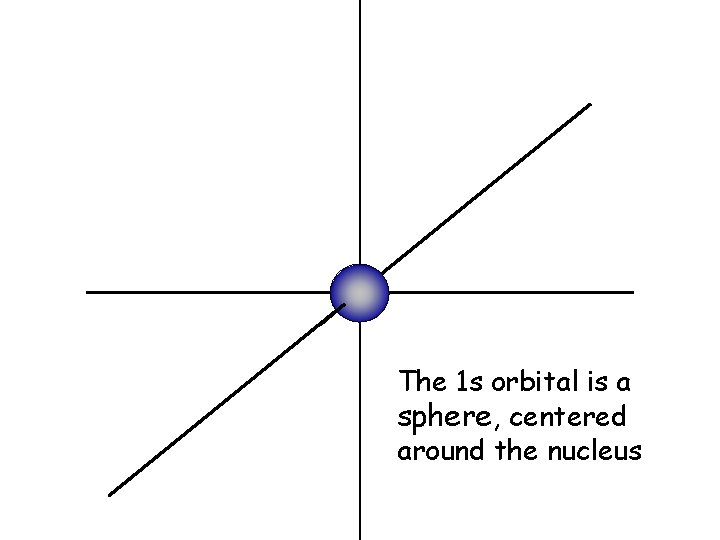
The 1 s orbital is a sphere, centered around the nucleus

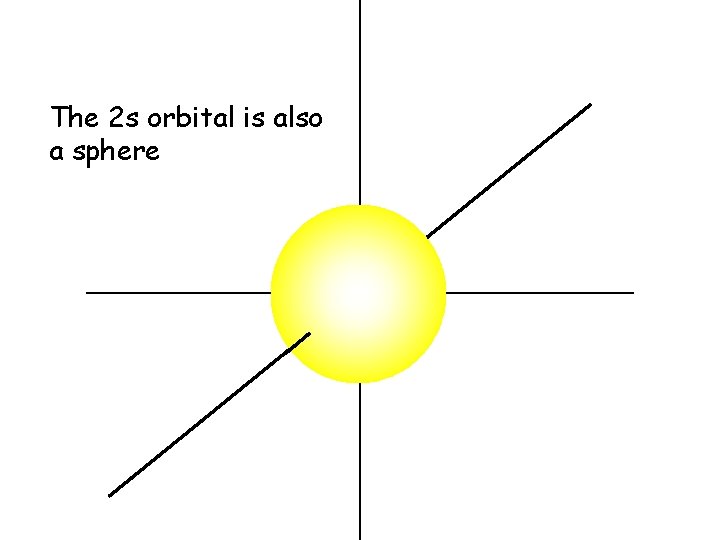
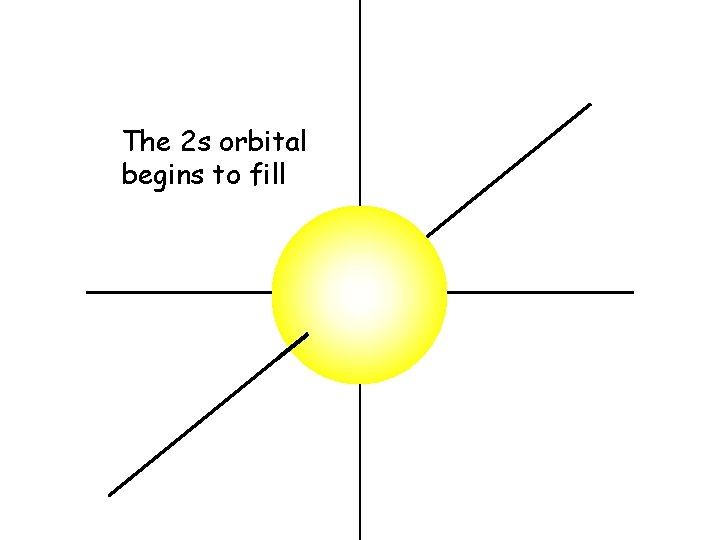
The 2 s orbital is also a sphere

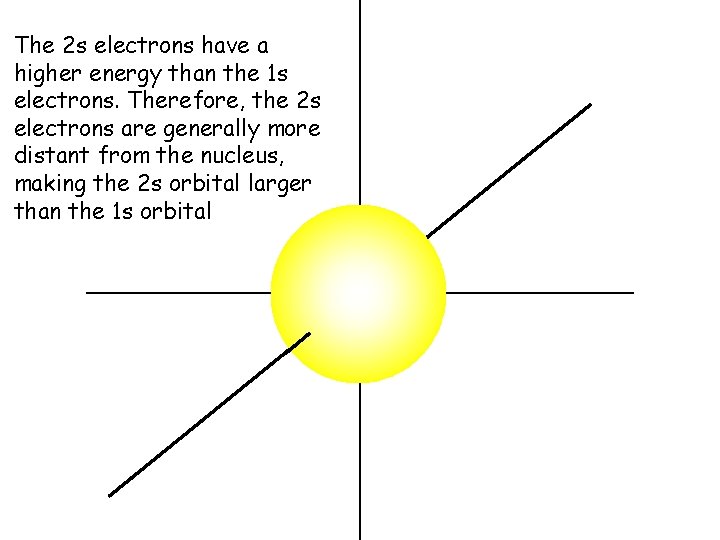
The 2 s electrons have a higher energy than the 1 s electrons. Therefore, the 2 s electrons are generally more distant from the nucleus, making the 2 s orbital larger than the 1 s orbital

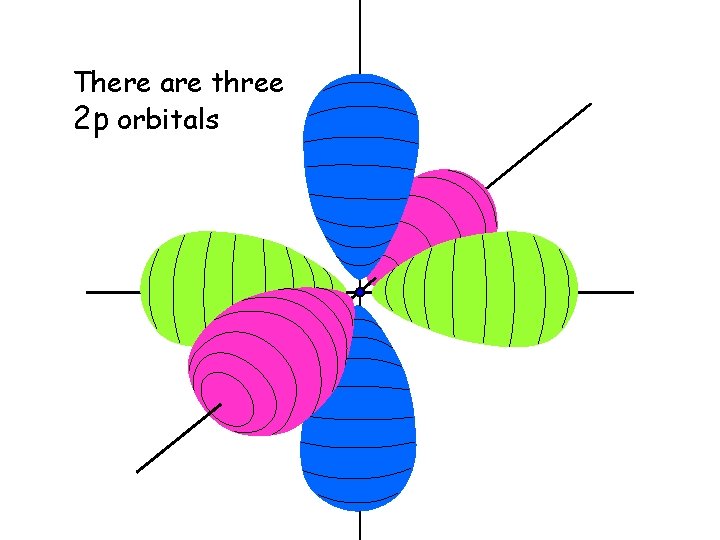
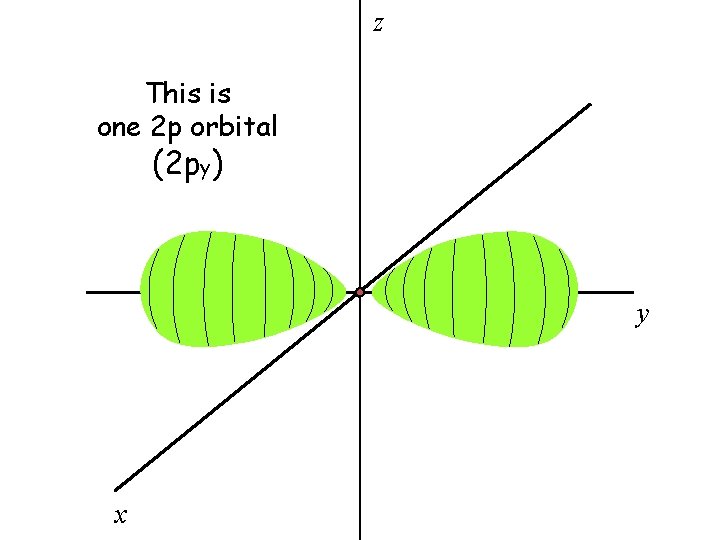
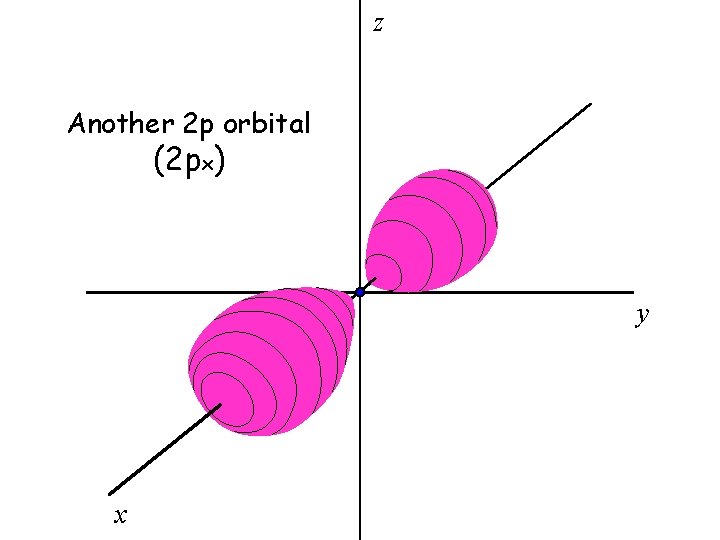
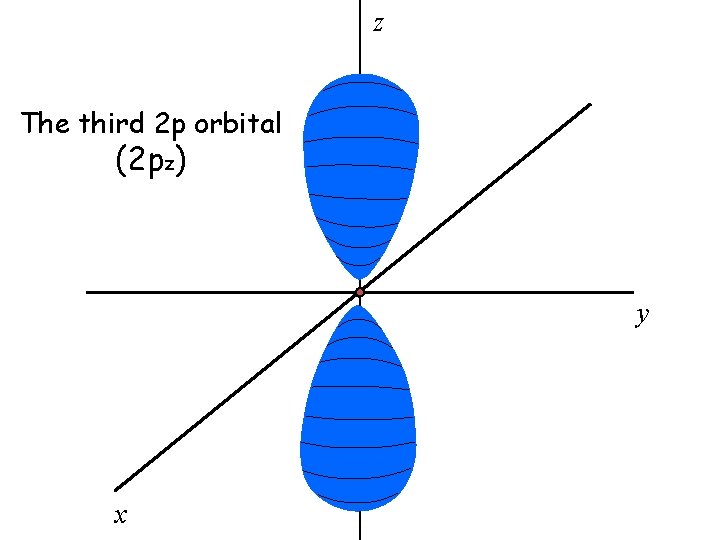
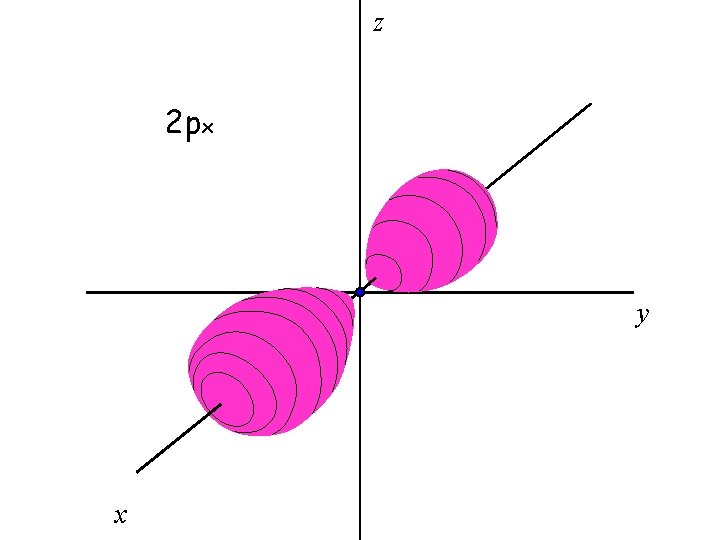
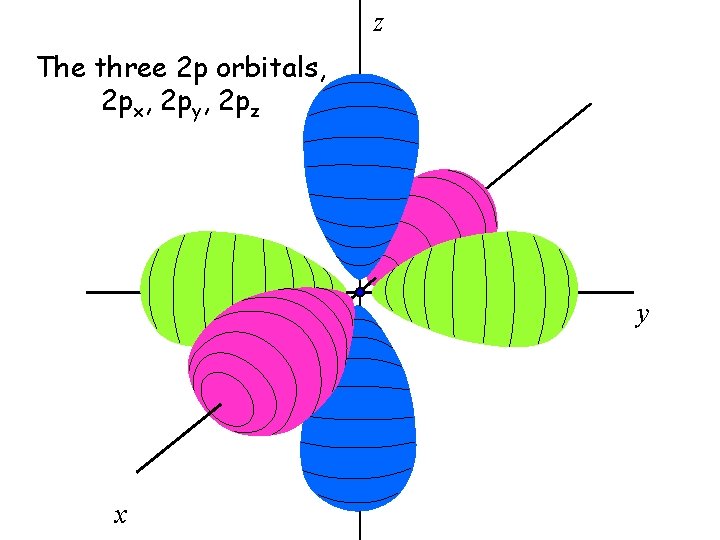
There are three 2 p orbitals

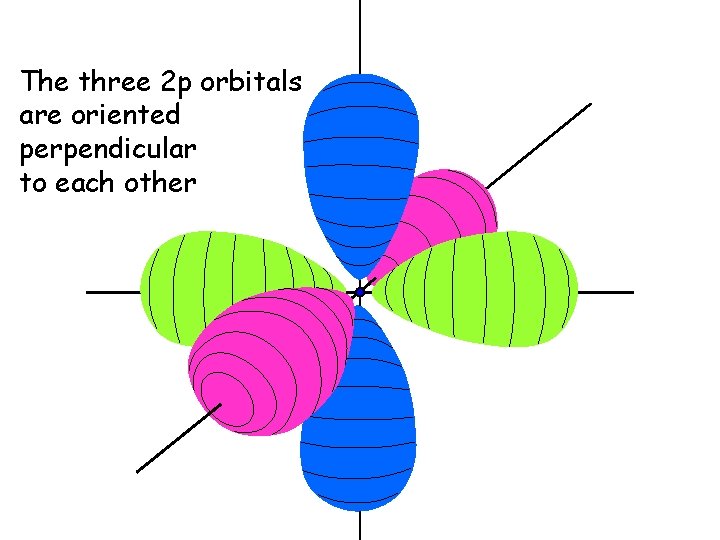
The three 2 p orbitals are oriented perpendicular to each other

z This is one 2 p orbital (2 py) y x

z Another 2 p orbital (2 px) y x

z The third 2 p orbital (2 pz) y x

z 2 px y x

z 2 px and 2 pz y x

z The three 2 p orbitals, 2 px, 2 py, 2 pz y x

Once the 1 s orbital is filled…

The 2 s orbital begins to fill

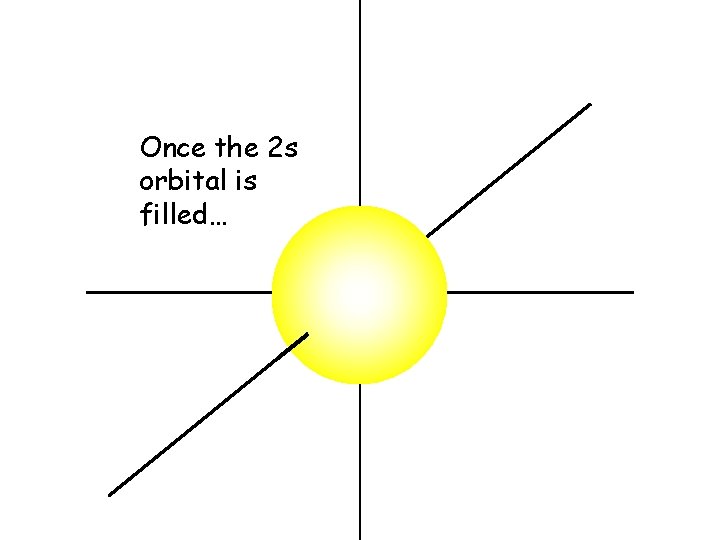
Once the 2 s orbital is filled…

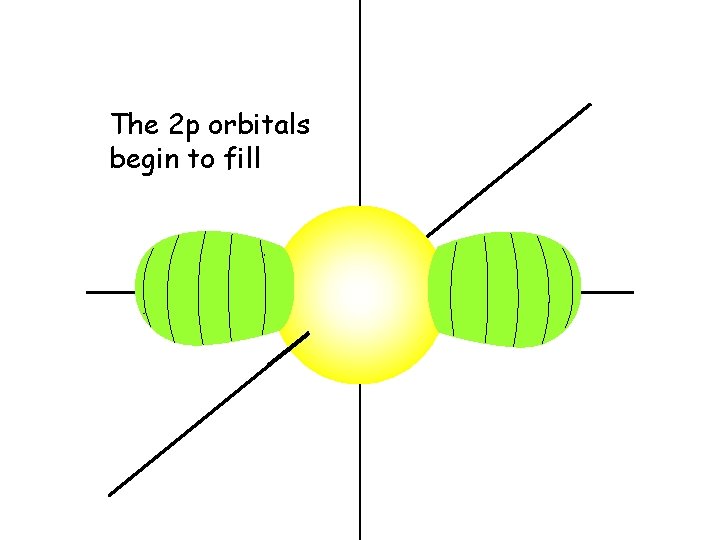
The 2 p orbitals begin to fill

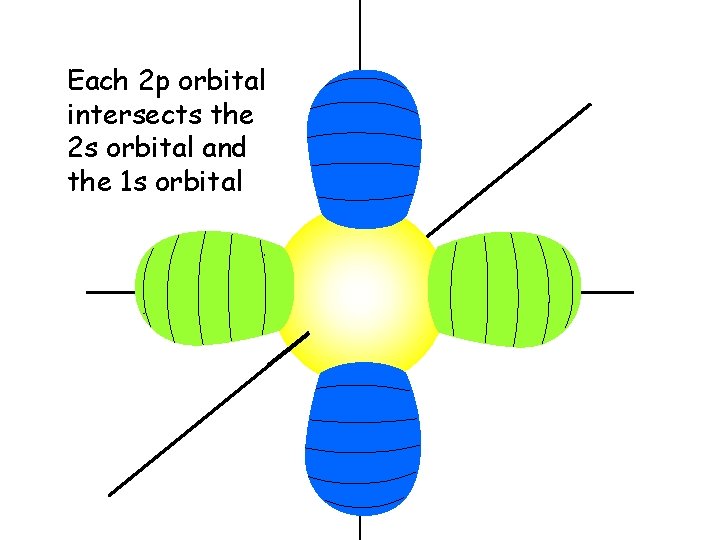
Each 2 p orbital intersects the 2 s orbital and the 1 s orbital

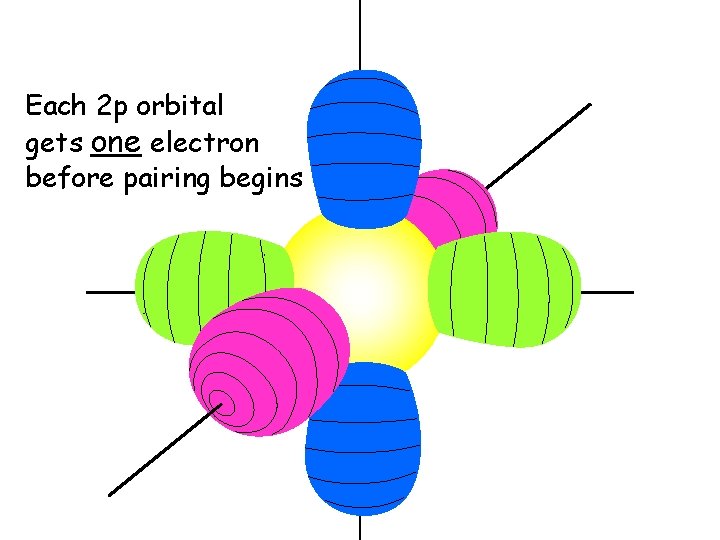
Each 2 p orbital gets one electron before pairing begins

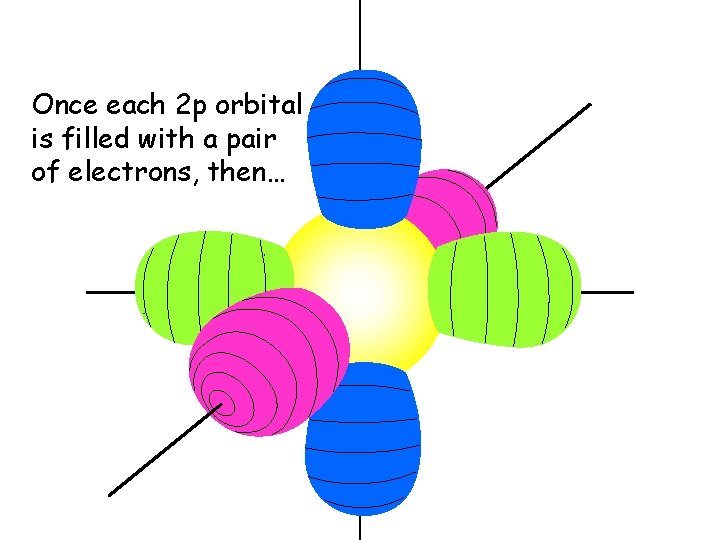
Once each 2 p orbital is filled with a pair of electrons, then…

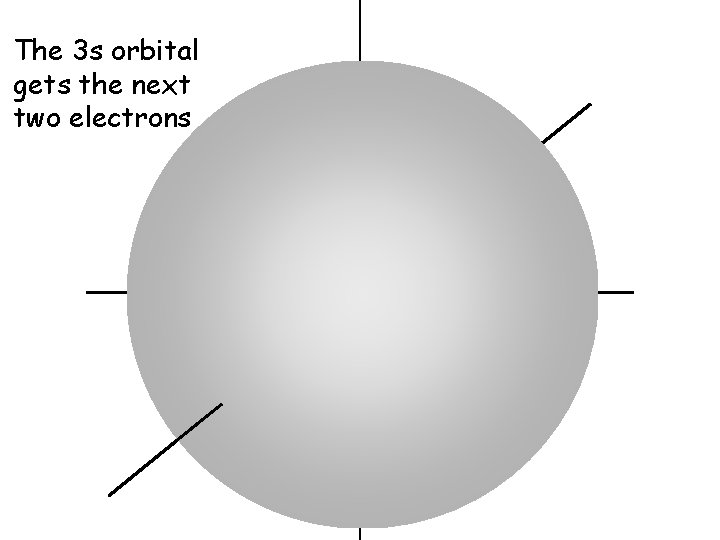
The 3 s orbital gets the next two electrons

The 3 s electrons have a higher energy than 1 s, 2 s, or 2 p electrons

3 s electrons are generally found further from the nucleus than 1 s, 2 s, or 2 p electrons

Exit Ticket • Explain the mechanism behind different colored fireworks