FixedDose Combination of DoravirineLamivudineTDF is NonInferior to EfavirenzEmtricitabineTDF

Fixed-Dose Combination of Doravirine/Lamivudine/TDF is Non-Inferior to Efavirenz/Emtricitabine/TDF in Treatment-Naïve Adults With HIV-1 Infection: Week 48 Results of the Phase 3 DRIVE-AHEAD Study IAS 2017 Abstract TUAB 0104 LB Kathleen E Squires 1, Jean-Michel Molina 2, Paul E Sax 3, Wing-Wai Wong 4, Chloe Orkin 5, Otto Sussmann 6, Richard Kaplan 7, Lisa Lupinacci 8, Anthony Rodgers 8, Xia Xu 8, Gina Lin 8, Sushma Kumar 8, Peter Sklar 8, Bach-Yen Nguyen 8, George J Hanna 8, Carey Hwang 8, Elizabeth Martin 8 on behalf of the DRIVE-AHEAD study team 1 Thomas Jefferson University, Philadelphia, PA, USA; 2 University of Paris Diderot, Hôpital Saint-Louis, Paris, France; 3 Brigham and Women's Hospital, Harvard Medical School, Boston, MA, USA; 4 Taipei Veterans General Hospital, Taipei, Taiwan; 5 Royal London Hospital, London, UK; 6 Asistencia Cientifica de Alta Complejidad SAS, Bogotá, Colombia; 7 Desmond Tutu HIV Foundation, Cape Town, South Africa; 8 Merck & Co. , Inc. , Kenilworth, NJ, USA

Acknowledgments We thank all the patients who participated in this study. The contributions of the investigators and their staff are also gratefully recognized. Primary investigators (by country): Australia – MT Bloch, RJ Finlayson, J Mc. Mahon; Belgium – S De Wit, I Derdelinckx, E Florence, L Vandekerckhove, B Vandercam; Canada – J De Wet, RP Le. Blanc, B Lebouche, D Longpre, B Trottier; Chile – C Beltran, CE Chahin, W Jensen, C Perez; Colombia – JMO Gutierrez, O Sussmann, JD Velez; Denmark – J Gerstoft, H Nielsen; Germany – K Arasteh, O Degen, S Esser, H-J Stellbrink, C Stephan, A Stoehr; Guatemala – EMR Alvarado, E Arathoon, RM Lopez, CR Mejia, LDG Patzan, R Pinzon; Honduras – C Parchment; Israel – M Chowers, I Levy, E Shahar, Z Sthoeger; Mexico – J Andrade, NPQ Perez, BEC Ramirez; Netherlands – JG den Hollander, B Rijnders; New Zealand – R Handy; Peru – N Bonifacio, P Campos, JA Hidalgo, RM Infante, ED Matos, Y Pinedo, ER Ticona; Portugal – JGS da Cunha, F Maltez, I Neves, P Pacheco, R Serrao; Puerto Rico – S Maldonado-Rivera, I Melendez-Rivera, RO Mendoza-Rodriguez, JO Morales-Ramirez, G Ortiz. Lasanta; Russia – S Kizhlo, VV Kulagin, VV Pokrovsky, TE Shimonova, AA Shuldyakov, OA Tsybakova, E Voronin, NG Zakharova; South Africa – E Baraldi, LF Fouche, F Hoosen, R Kaplan, JJ Lombaard, AA Mahomed, EA Mitha, L Mohapi, MS Rassool, N Siddique; Spain – J Berenguer, HK Freud, SM Guillen, FV Mendez, JS Moreno, DP Palter; Switzerland – A Calmy, M Cavassini, J Fehr; Taiwan – C-C Hung, W-C Ko, H-H Lin, Y-H Lin, H-C Tsai, W-W Wong; Thailand – A Avihingsanon, D Changpradub, P Chetchotisakd, S Kiertiburanakul, W Ratanasuwan, K Supparatpinyo; United Kingdom – A Clarke, A Fox, MA Johnson, MR Nelson, C Orkin, N Perry, A Ustianowski, IG Williams, A Winston; United States – DM Asmuth, NC Bellos, DS Berger, CJ Bettacchi, L Bhatti, T Campbell, RE Campo, KK Casey, JG Castro, GE Crofoot, D Cunningham, E De. Jesus, J Feinberg, FA Felizarta, C Fichtenbaum, TT Jefferson, M Johnson, PN Kumar, ST Lewis, RA Loftus, AE Luque, CK Mc. Donald, M Mogyoros, RM Novak, O Osiyemi, DM Parenti, DJ Prelutsky, DJ Riedel, WJ Robbins, PJ Ruane, M Scott, ASY Shon, J Sims, J Slim, L Sloan, DE Sweet, KT Tashima, P Tebas, MA Thompson, WJ Towner, KJ Vigil, BG Yangco, R Zane. Medical writing assistance was provided by Annette Smith, Ph. D, of Complete Medical Communications, Macclesfield, UK. This assistance was funded by Merck & Co. , Inc. , Kenilworth, NJ, USA. IAS 2017, Abstract TUAB 0104 LB. Copyright © 2017 Merck & Co. , Inc. All Rights Reserved

Disclosures • Dr. Kathleen Squires has received research grants awarded to her institution from Gilead Sciences, and has served on advisory boards for BMS, Gilead Sciences, Janssen, MSD, and Vii. V IAS 2017, Abstract TUAB 0104 LB. Copyright © 2017 Merck & Co. , Inc. All Rights Reserved

Background • Doravirine (DOR) is a novel, next-generation non-nucleoside reverse transcriptase inhibitor (NNRTI) Ø Unique resistance profile with in vitro activity against wild-type HIV-1 and the most prevalent NNRTI resistance mutations (RT: K 103 N, Y 181 C, G 190 A, K 103 N/Y 181 C, and E 138 K)1 Ø Dosed once daily (QD), without regard to food 2 Ø Low potential for drug–drug interactions, 3 including with acid-reducing agents 4 Ø Being developed as a single entity and fixed-dose combination with lamivudine (3 TC) and tenofovir disoproxil fumarate (TDF) • In the Phase 2 b clinical trial, DOR 100 mg demonstrated favorable efficacy and a superior neuropsychiatric profile vs efavirenz (EFV) 600 mg QD 5 • In the Phase 3 DRIVE-FORWARD clinical trial, DOR 100 mg showed non-inferior efficacy to ritonavir-boosted darunavir, and a more favorable lipid profile 6 1. Feng M, et al. Antimicrob Agents Chemother. 2016; 60: 2241 -2247; 2. Anderson MS, et al. Antivir Ther. 2015; 20: 397 -405; 3. Behm MO, et al. Clin Drug Investig. 2017; [Epub ahead of print]; 4. Khalilieh S, et al. IAS 2017. Poster MOPEB 0334; 5. Gatell JM, et al. J Int AIDS Soc. 2014; 17(4 Suppl 3): 19532; 6. Molina JM, et al. CROI 2017. Abstract 45 LB IAS 2017, Abstract TUAB 0104 LB. Copyright © 2017 Merck & Co. , Inc. All Rights Reserved
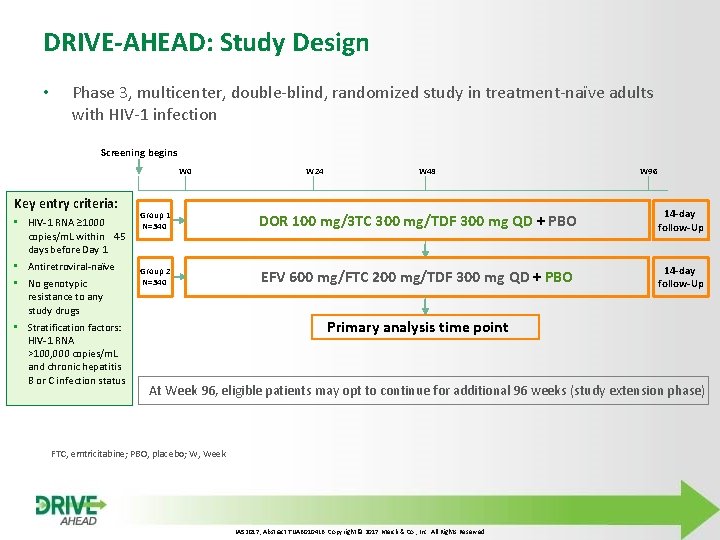
DRIVE-AHEAD: Study Design • Phase 3, multicenter, double-blind, randomized study in treatment-naïve adults with HIV-1 infection Screening begins W 0 Key entry criteria: • HIV-1 RNA ≥ 1000 copies/m. L within 45 days before Day 1 • Antiretroviral-naїve • No genotypic resistance to any study drugs • Stratification factors: HIV-1 RNA >100, 000 copies/m. L and chronic hepatitis B or C infection status W 24 W 48 W 96 Group 1 N=340 DOR 100 mg/3 TC 300 mg/TDF 300 mg QD + PBO 14 -day follow-Up Group 2 N=340 EFV 600 mg/FTC 200 mg/TDF 300 mg QD + PBO 14 -day follow-Up Primary analysis time point At Week 96, eligible patients may opt to continue for additional 96 weeks (study extension phase) FTC, emtricitabine; PBO, placebo; W, Week IAS 2017, Abstract TUAB 0104 LB. Copyright © 2017 Merck & Co. , Inc. All Rights Reserved

Study Hypotheses • Primary efficacy and safety hypotheses Ø DOR/3 TC/TDF QD is non-inferior to EFV/FTC/TDF QD, as assessed by the proportion of participants with HIV-1 RNA <50 copies/m. L at Week 48 § Considered non-inferior if lower bound of two-sided 95% CI for between-treatment difference is greater than -10 percentage points § 90% power with 340 participants in each treatment group assuming a response rate of 80% at Week 48 for both treatment groups (FDA Snapshot approach) Ø DOR/3 TC/TDF QD is superior to EFV/FTC/TDF QD, as assessed by the proportion of participants with neuropsychiatric adverse events (AEs) by Week 48 in the categories of dizziness, sleep disorders and disturbances, and altered sensorium (superiority to be tested sequentially within each category) • Secondary safety hypotheses Ø DOR/3 TC/TDF QD is superior to EFV/FTC/TDF QD, as assessed by the mean change from baseline in LDL-C and non–HDL-C at Week 48 LDL-C, low-density lipoprotein cholesterol; non–HDL-C, non–high-density lipoprotein cholesterol IAS 2017, Abstract TUAB 0104 LB. Copyright © 2017 Merck & Co. , Inc. All Rights Reserved

Disposition, Week 48 Randomized N=734 DOR/3 TC/TDF: 368 randomized 364 treated 51 (14%) discontinued 10 (3%) 18 (5%) 8 (2%) 6 (2%) 4 (1%) 2 (1%) 1 (<1%) EFV/FTC/TDF: 366 randomized 364 treated Adverse event Lack of efficacy Withdrew consent Lost to follow-up Protocol violation Physician decision Death Non-compliance Pregnancy 61 (17%) discontinued 23 (6%) 10 (3%) 11 (3%) 7 (2%) 2 (1%) 3 (1%) 2 (1%) 1 (<1%) 313 (86%) continuing 303 (83%) continuing IAS 2017, Abstract TUAB 0104 LB. Copyright © 2017 Merck & Co. , Inc. All Rights Reserved
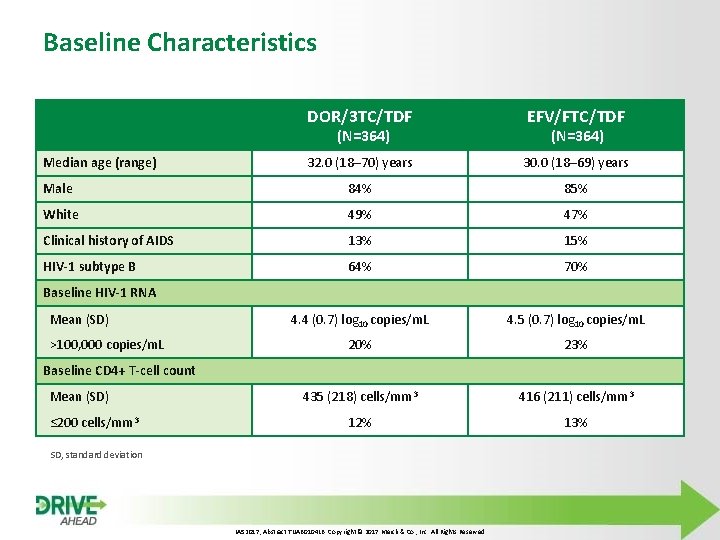
Baseline Characteristics DOR/3 TC/TDF EFV/FTC/TDF 32. 0 (18– 70) years 30. 0 (18– 69) years Male 84% 85% White 49% 47% Clinical history of AIDS 13% 15% HIV-1 subtype B 64% 70% 4. 4 (0. 7) log 10 copies/m. L 4. 5 (0. 7) log 10 copies/m. L 20% 23% 435 (218) cells/mm 3 416 (211) cells/mm 3 12% 13% (N=364) Median age (range) (N=364) Baseline HIV-1 RNA Mean (SD) >100, 000 copies/m. L Baseline CD 4+ T-cell count Mean (SD) ≤ 200 cells/mm 3 SD, standard deviation IAS 2017, Abstract TUAB 0104 LB. Copyright © 2017 Merck & Co. , Inc. All Rights Reserved
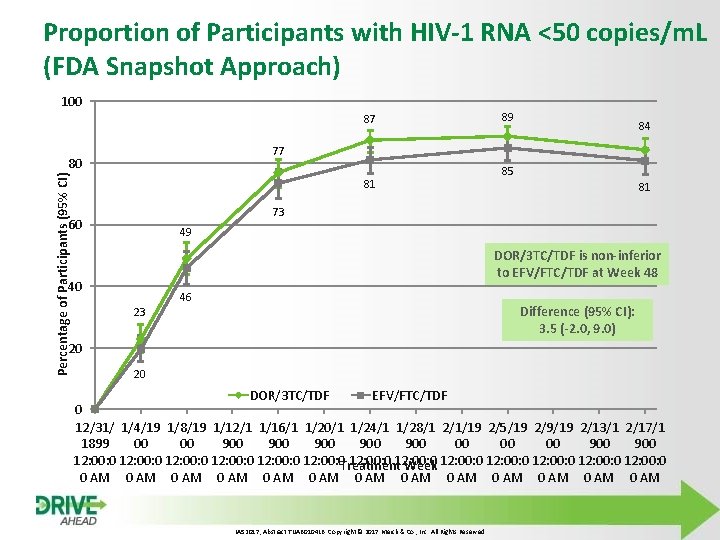
Proportion of Participants with HIV-1 RNA <50 copies/m. L (FDA Snapshot Approach) 100 87 84 77 80 Percentage of Participants (95% CI) 89 81 85 81 73 60 49 DOR/3 TC/TDF is non-inferior to EFV/FTC/TDF at Week 48 40 46 Difference (95% CI): 3. 5 (-2. 0, 9. 0) 23 20 20 DOR/3 TC/TDF EFV/FTC/TDF 0 12/31/ 1/4/19 1/8/19 1/12/1 1/16/1 1/20/1 1/24/1 1/28/1 2/1/19 2/5/19 2/9/19 2/13/1 2/17/1 1899 00 00 900 900 900 12: 00: 0 12: 00: 0 Treatment Week 12: 00: 0 12: 00: 0 0 AM 0 AM 0 AM 0 AM IAS 2017, Abstract TUAB 0104 LB. Copyright © 2017 Merck & Co. , Inc. All Rights Reserved
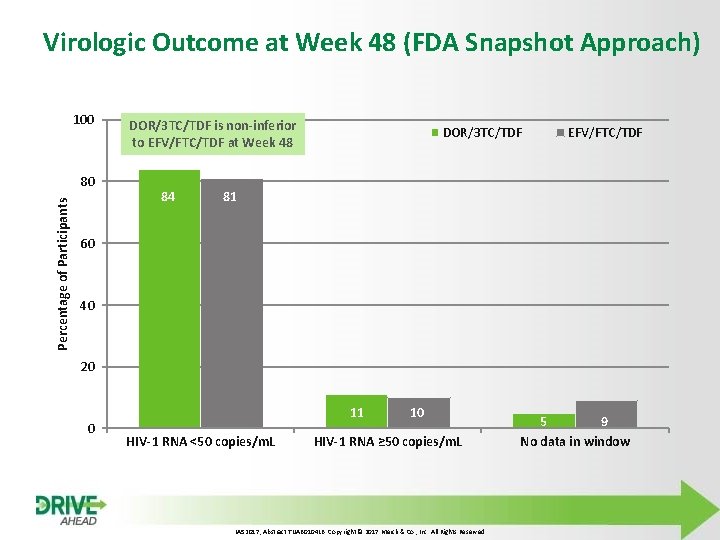
Virologic Outcome at Week 48 (FDA Snapshot Approach) 100 Percentage of Participants 80 DOR/3 TC/TDF is non-inferior to EFV/FTC/TDF at Week 48 84 DOR/3 TC/TDF EFV/FTC/TDF 81 60 40 20 0 11 HIV-1 RNA <50 copies/m. L 10 HIV-1 RNA ≥ 50 copies/m. L IAS 2017, Abstract TUAB 0104 LB. Copyright © 2017 Merck & Co. , Inc. All Rights Reserved 5 9 No data in window
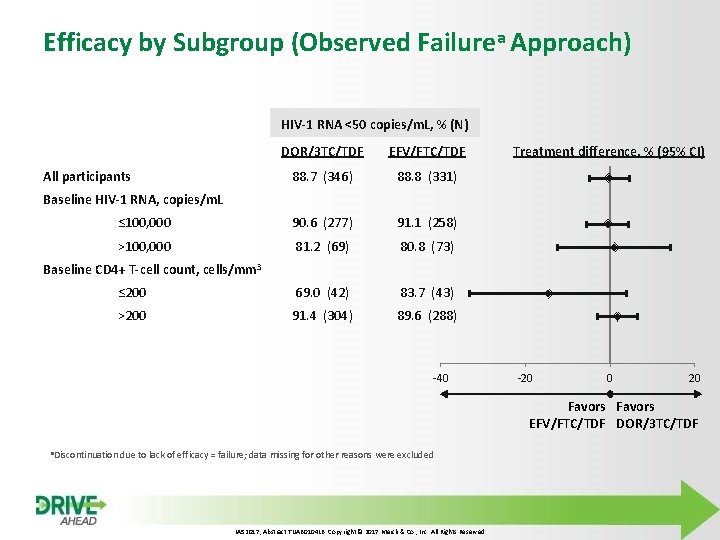
Efficacy by Subgroup (Observed Failurea Approach) HIV-1 RNA <50 copies/m. L, % (N) DOR/3 TC/TDF EFV/FTC/TDF 88. 7 (346) 88. 8 (331) ≤ 100, 000 90. 6 (277) 91. 1 (258) >100, 000 81. 2 (69) 80. 8 (73) ≤ 200 69. 0 (42) 83. 7 (43) >200 91. 4 (304) 89. 6 (288) All participants Treatment difference, % (95% CI) Baseline HIV-1 RNA, copies/m. L Baseline CD 4+ T-cell count, cells/mm 3 -40 -20 0 20 Favors EFV/FTC/TDF DOR/3 TC/TDF a. Discontinuation due to lack of efficacy = failure; data missing for other reasons were excluded IAS 2017, Abstract TUAB 0104 LB. Copyright © 2017 Merck & Co. , Inc. All Rights Reserved
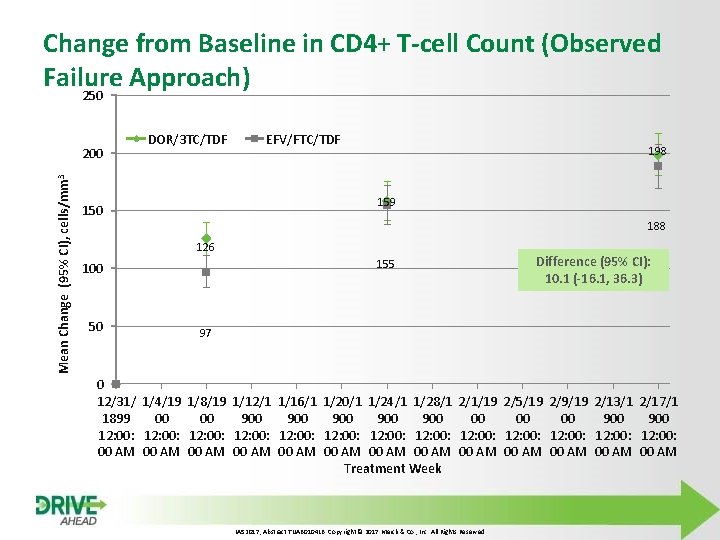
Change from Baseline in CD 4+ T-cell Count (Observed Failure Approach) 250 Mean Change (95% CI), cells/mm 3 200 DOR/3 TC/TDF EFV/FTC/TDF 198 159 150 188 126 155 100 50 Difference (95% CI): 10. 1 (-16. 1, 36. 3) 97 0 12/31/ 1/4/19 1/8/19 1/12/1 1/16/1 1/20/1 1/24/1 1/28/1 2/1/19 2/5/19 2/9/19 2/13/1 2/17/1 1899 00 00 900 900 900 12: 00: 12: 00: 12: 00: 12: 00 AM 00 AM 00 AM 00 AM Treatment Week IAS 2017, Abstract TUAB 0104 LB. Copyright © 2017 Merck & Co. , Inc. All Rights Reserved
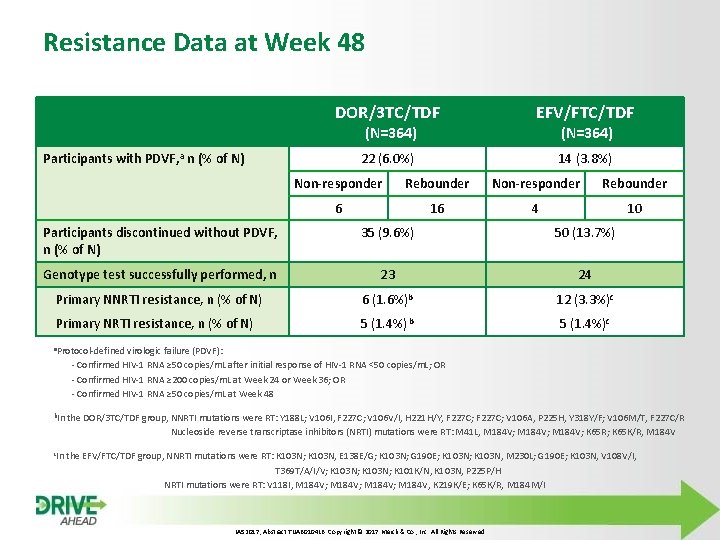
Resistance Data at Week 48 DOR/3 TC/TDF EFV/FTC/TDF 22 (6. 0%) 14 (3. 8%) (N=364) Participants with PDVF, a n (% of N) (N=364) Non-responder Rebounder 6 16 4 10 Participants discontinued without PDVF, n (% of N) 35 (9. 6%) 50 (13. 7%) Genotype test successfully performed, n 23 24 Primary NNRTI resistance, n (% of N) 6 (1. 6%)b 12 (3. 3%)c Primary NRTI resistance, n (% of N) 5 (1. 4%) b 5 (1. 4%)c a. Protocol-defined virologic failure (PDVF): - Confirmed HIV-1 RNA ≥ 50 copies/m. L after initial response of HIV-1 RNA <50 copies/m. L; OR - Confirmed HIV-1 RNA ≥ 200 copies/m. L at Week 24 or Week 36; OR - Confirmed HIV-1 RNA ≥ 50 copies/m. L at Week 48 b. In the DOR/3 TC/TDF group, NNRTI mutations were RT: Y 188 L; V 106 I, F 227 C; V 106 V/I, H 221 H/Y, F 227 C; V 106 A, P 225 H, Y 318 Y/F; V 106 M/T, F 227 C/R Nucleoside reverse transcriptase inhibitors (NRTI) mutations were RT: M 41 L, M 184 V; K 65 R; K 65 K/R, M 184 V c. In the EFV/FTC/TDF group, NNRTI mutations were RT: K 103 N; K 103 N, E 138 E/G; K 103 N; G 190 E; K 103 N, M 230 L; G 190 E; K 103 N, V 108 V/I, T 369 T/A/I/V; K 103 N; K 101 K/N, K 103 N, P 225 P/H NRTI mutations were RT: V 118 I, M 184 V; M 184 V, K 219 K/E; K 65 K/R, M 184 M/I IAS 2017, Abstract TUAB 0104 LB. Copyright © 2017 Merck & Co. , Inc. All Rights Reserved
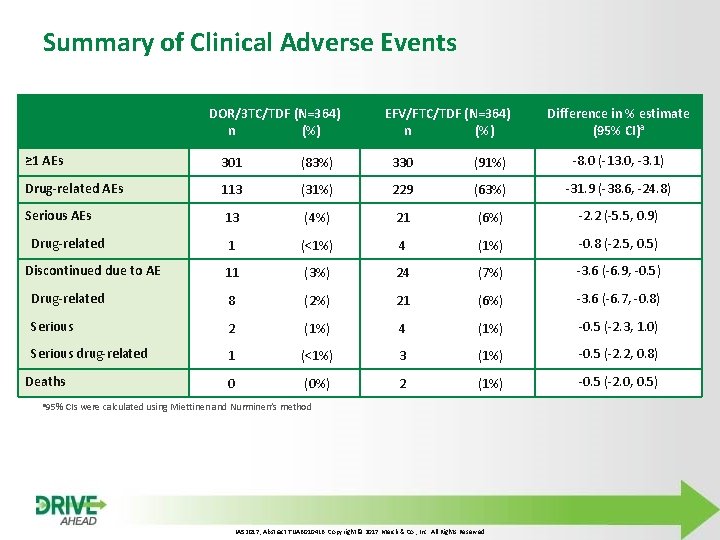
Summary of Clinical Adverse Events DOR/3 TC/TDF (N=364) n (%) EFV/FTC/TDF (N=364) n (%) Difference in % estimate (95% CI)a ≥ 1 AEs 301 (83%) 330 (91%) -8. 0 (-13. 0, -3. 1) Drug-related AEs 113 (31%) 229 (63%) -31. 9 (-38. 6, -24. 8) Serious AEs 13 (4%) 21 (6%) -2. 2 (-5. 5, 0. 9) Drug-related 1 (<1%) 4 (1%) -0. 8 (-2. 5, 0. 5) Discontinued due to AE 11 (3%) 24 (7%) -3. 6 (-6. 9, -0. 5) Drug-related 8 (2%) 21 (6%) -3. 6 (-6. 7, -0. 8) Serious 2 (1%) 4 (1%) -0. 5 (-2. 3, 1. 0) Serious drug-related 1 (<1%) 3 (1%) -0. 5 (-2. 2, 0. 8) Deaths 0 (0%) 2 (1%) -0. 5 (-2. 0, 0. 5) a 95% CIs were calculated using Miettinen and Nurminen’s method IAS 2017, Abstract TUAB 0104 LB. Copyright © 2017 Merck & Co. , Inc. All Rights Reserved
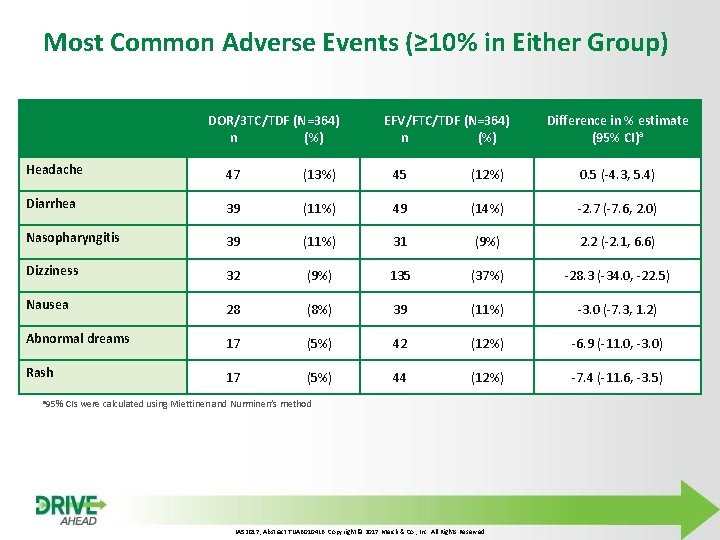
Most Common Adverse Events (≥ 10% in Either Group) DOR/3 TC/TDF (N=364) n (%) EFV/FTC/TDF (N=364) n (%) Difference in % estimate (95% CI)a Headache 47 (13%) 45 (12%) 0. 5 (-4. 3, 5. 4) Diarrhea 39 (11%) 49 (14%) -2. 7 (-7. 6, 2. 0) Nasopharyngitis 39 (11%) 31 (9%) 2. 2 (-2. 1, 6. 6) Dizziness 32 (9%) 135 (37%) -28. 3 (-34. 0, -22. 5) Nausea 28 (8%) 39 (11%) -3. 0 (-7. 3, 1. 2) Abnormal dreams 17 (5%) 42 (12%) -6. 9 (-11. 0, -3. 0) Rash 17 (5%) 44 (12%) -7. 4 (-11. 6, -3. 5) a 95% CIs were calculated using Miettinen and Nurminen’s method IAS 2017, Abstract TUAB 0104 LB. Copyright © 2017 Merck & Co. , Inc. All Rights Reserved
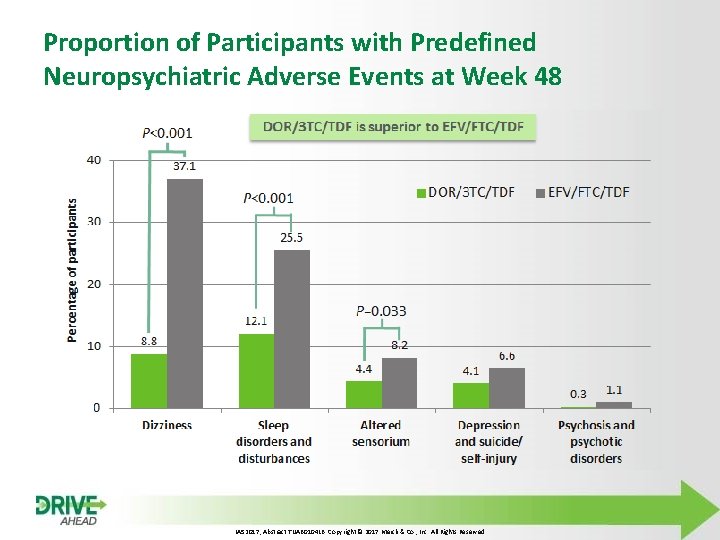
Proportion of Participants with Predefined Neuropsychiatric Adverse Events at Week 48 IAS 2017, Abstract TUAB 0104 LB. Copyright © 2017 Merck & Co. , Inc. All Rights Reserved
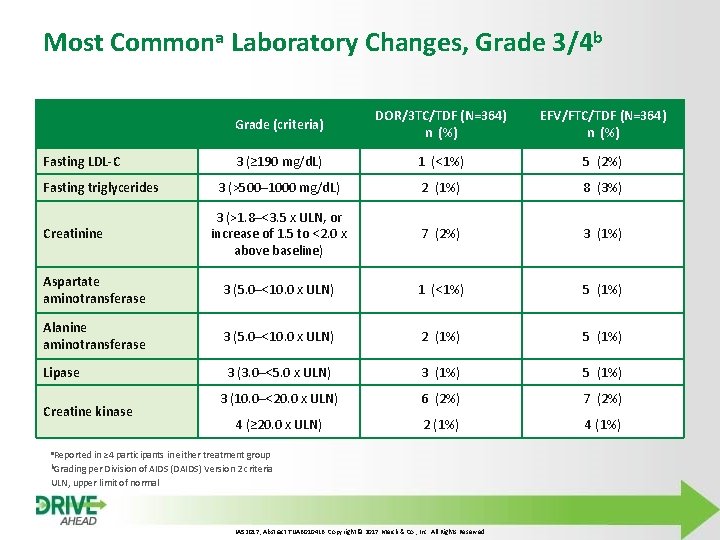
Most Commona Laboratory Changes, Grade 3/4 b Grade (criteria) DOR/3 TC/TDF (N=364) n (%) EFV/FTC/TDF (N=364) n (%) 3 (≥ 190 mg/d. L) 1 (<1%) 5 (2%) 3 (>500– 1000 mg/d. L) 2 (1%) 8 (3%) 3 (>1. 8–<3. 5 x ULN, or increase of 1. 5 to <2. 0 x above baseline) 7 (2%) 3 (1%) Aspartate aminotransferase 3 (5. 0–<10. 0 x ULN) 1 (<1%) 5 (1%) Alanine aminotransferase 3 (5. 0–<10. 0 x ULN) 2 (1%) 5 (1%) Lipase 3 (3. 0–<5. 0 x ULN) 3 (1%) 5 (1%) 3 (10. 0–<20. 0 x ULN) 6 (2%) 7 (2%) 4 (≥ 20. 0 x ULN) 2 (1%) 4 (1%) Fasting LDL-C Fasting triglycerides Creatinine Creatine kinase a. Reported in ≥ 4 participants in either treatment group b. Grading per Division of AIDS (DAIDS) Version 2 criteria ULN, upper limit of normal IAS 2017, Abstract TUAB 0104 LB. Copyright © 2017 Merck & Co. , Inc. All Rights Reserved

Fasting Lipids: Change from Baseline at Week 48 IAS 2017, Abstract TUAB 0104 LB. Copyright © 2017 Merck & Co. , Inc. All Rights Reserved

Conclusions • In treatment-naïve adults with HIV-1 infection, DOR/3 TC/TDF administered once daily demonstrated: Ø Antiviral potency with non-inferior efficacy to EFV/FTC/TDF regardless of baseline HIV-1 RNA Ø Low rate of resistance, with only 1. 6% of participants developing resistance to any study drug through Week 48 • DOR/3 TC/TDF was generally well tolerated and safe: Ø Neuropsychiatric profile superior to EFV/FTC/TDF, as measured by lower proportion of participants with neuropsychiatric AEs in categories of dizziness, sleep disorders and disturbances, and altered sensorium Ø Lipid profile superior to EFV/FTC/TDF, as assessed by difference from baseline in fasting LDL-C and non–HDL-C Doravirine is a novel, once-daily NNRTI for first-line treatment with consistent efficacy regardless of baseline viral load and favorable tolerability and safety profile in 2 Phase 3 clinical trials 1 1. Molina JM, et al. CROI 201. Abstract 45 LB IAS 2017, Abstract TUAB 0104 LB. Copyright © 2017 Merck & Co. , Inc. All Rights Reserved
- Slides: 19




































