Five slides about THE CBC METHOD OF ELECTRON

Five slides about… THE CBC METHOD OF ELECTRON COUNTING
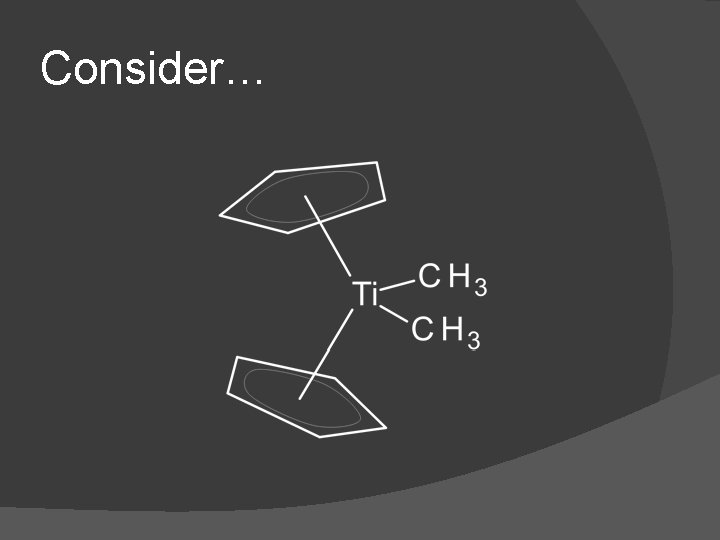
Consider…
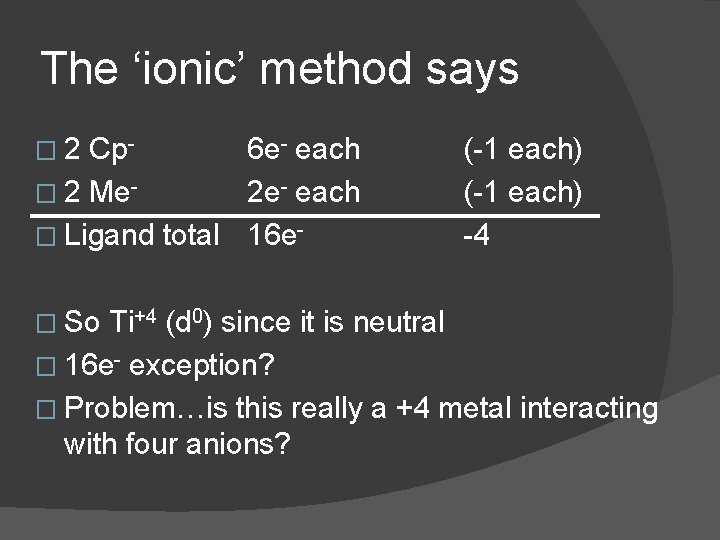
The ‘ionic’ method says � 2 Cp 6 e- each � 2 Me 2 e- each � Ligand total 16 e� So (-1 each) -4 Ti+4 (d 0) since it is neutral � 16 e- exception? � Problem…is this really a +4 metal interacting with four anions?

The ‘covalent’ method says � 2 Cp 5 e- each � 2 Me 1 e- each � Ligand total 12 e� So Ti 0 (d 4) since it is neutral � 16 e- exception? � Problem…assuming the d-electrons are nonbonding how is this diamagnetic?

CBC ligand classification �L �Ligand donates two electrons �CO, phosphines, alkenes �X �Ligand donates one electron �H, halides, alkyl �Z �Ligand donates zero electrons �boranes
![How to count? [MLl. Xx. Zz] � Total electron count �From ligands: 2 l How to count? [MLl. Xx. Zz] � Total electron count �From ligands: 2 l](http://slidetodoc.com/presentation_image_h2/5ac5867d9f28e339460785d24ef7daa2/image-6.jpg)
How to count? [MLl. Xx. Zz] � Total electron count �From ligands: 2 l + x �From metal: m �m + 2 l + x � Valence number (VN) for the metal = x + 2 z � Ligand bond number (LBN) for the metal = l + x + z � dn count for metal = m - VN
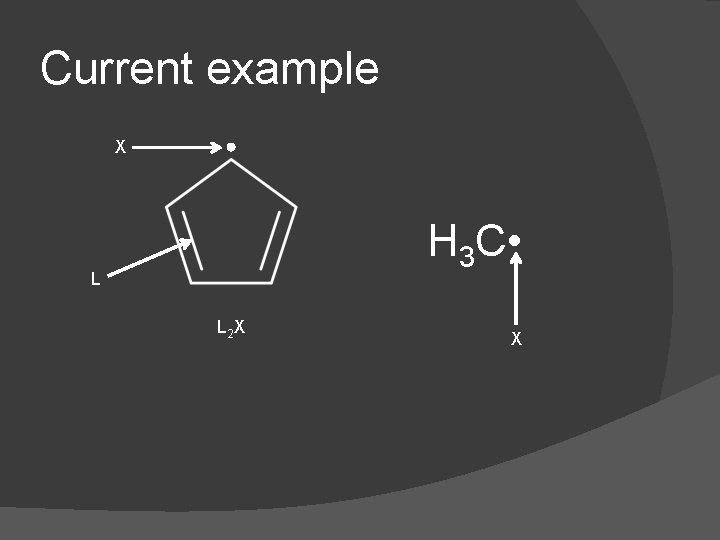
Current example X H 3 C • L L 2 X X

Counting � Compound is classified as ML 4 X 4 � Electron count: 4 + 10 + 2 = 16 � VN: 4 � LBN: 8 � d n: 0

What have we learned? � Electrons: 16 � VN: 4 � Classification: ML 4 X 4 Ti 0 Electron Count 8 ML 2 Valence 1 2 10 MLX 2 12 13 ML 4 ML 3 X ML 2 X 2 MLX 3 MX 4 6% 11 ML 3 ML 2 X 3 4 9 ML 4 X ML 2 X 3 <1% http: //www. columbia. edu/cu/chemistry/groups/parkin/mlxz. htm 16 ML 4 X 2 1% 18 ML 7 1% ML 6 X <1% ML 5 X 2 1% ML 4 X 3 5% ML 3 X 4 6% 17 ML 6 2% ML 5 X ML 3 X 3 <1% ML 2 X 4 7% 15 ML 3 X 2 MLX 4 1% 14 ML 6 X 2 4% ML 5 X 3 9% ML 4 X 4 49% ML 5 X 4 4%
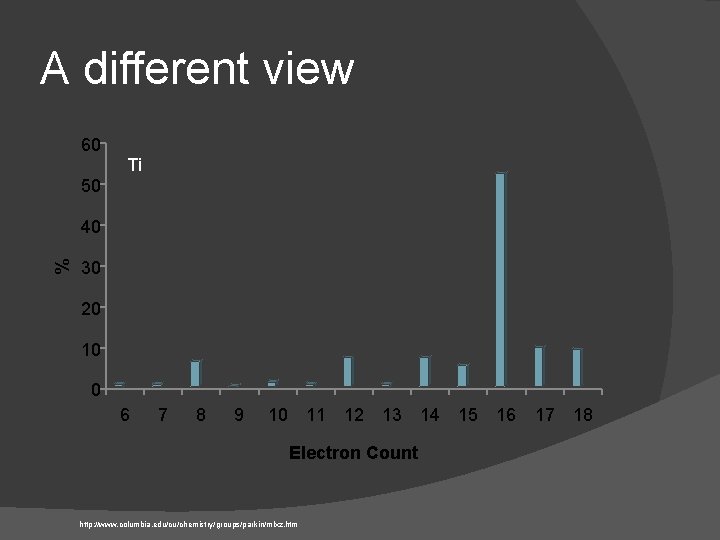
A different view 60 Ti 50 % 40 30 20 10 0 6 7 8 9 10 11 12 13 14 15 16 Electron Count http: //www. columbia. edu/cu/chemistry/groups/parkin/mlxz. htm 17 18
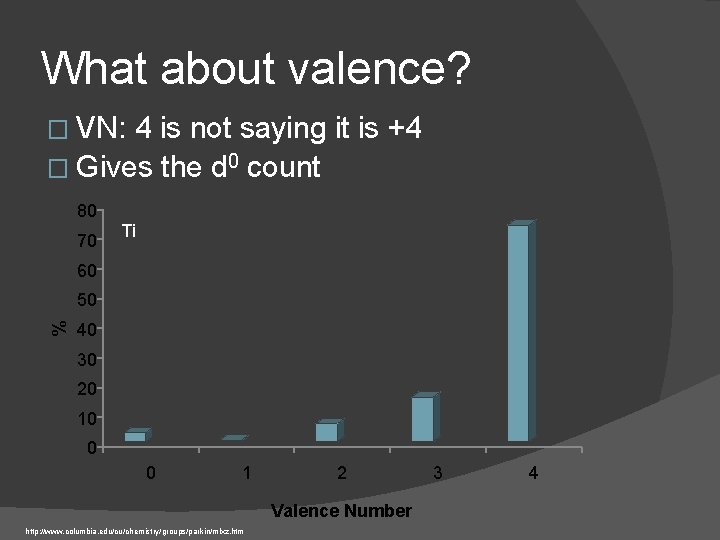
What about valence? � VN: 4 is not saying it is +4 � Gives the d 0 count 80 70 Ti 60 % 50 40 30 20 10 0 0 1 2 Valence Number http: //www. columbia. edu/cu/chemistry/groups/parkin/mlxz. htm 3 4
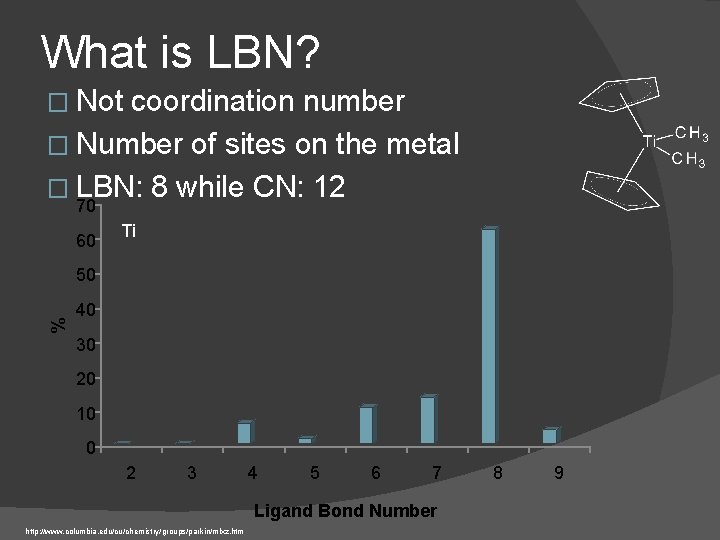
What is LBN? � Not coordination number � Number of sites on the metal � LBN: 8 while CN: 12 70 60 Ti % 50 40 30 20 10 0 2 3 4 5 6 7 Ligand Bond Number http: //www. columbia. edu/cu/chemistry/groups/parkin/mlxz. htm 8 9

Further readings � M. L. H. Green J. Organomet. Chem. 1995, 500, 127 -148. � G. Parkin, in Comprehensive Organometallic Chemistry III, Volume 1, Chapter 1; R. H. Crabtree and D. M. P. Mingos (Eds), Elsevier, Oxford, 2006. � http: //www. columbia. edu/cu/chemistry/grou ps/parkin/cbc. htm
- Slides: 13