Five As AssessClinical Problem Ask Question Acquire Best


Five A’s • Assess:了解臨床的需求(Clinical Problem) • Ask :發現問題的所在(Question) • Acquire :找尋最好的資料(Best Evidence) • Appraise :分析資料(Validity, Importance) • Apply :應用在病人身上(Patient) 2

Clinical Situation • Mr. Cheng is a 60 year old male patient. He was diagnosed as hepatitis B and C virus infection related hepatocellular carcinoma (HCC) on 2003. • He has received radiotherapy during 2003/1/24~3/6, transarterial chemoembolization (TACE) during 2006/3/4~2007/12/4, computer tomography-guided radiofrequency ablation (RFA) on 2008/8/5. • In 2009 April, abdominal sonography showed that HCC progress to retroperitoneal lymph nodes, which is suspected extrahepatic seeding tumor between abdominal wall and right lobe liver. At that time, the lab data showed elevated alpha-fetoprotein (AFP) 93. 46 ng/m. L. • At that time, the doctor considered sorafenib 400 mg PO BID for this patient. Is sorafenib effective for this patient? 3

Five A’s • Assess:了解臨床的需求(Clinical Problem) • Ask :發現問題的所在(Question) • Acquire :找尋最好的資料(Best Evidence) • Appraise :分析資料(Validity, Importance) • Apply :應用在病人身上(Patient) 4



Risk Factors • • 7 Liver cirrhosis Hepatitis B Virus Hepatitis C Virus Alcohol Cigarette smoking Betel nut Afaltoxin • Obesity • Fatty liver • Non-alcohol steatohepatitis (NASH) • Diabetes • Hereditary tyrosinemia • Hepatic porphyria • Genetic hemochromatosis • α 1 antitrypsin deficiency 林志陵, 高嘉宏. 肝癌的流行病學. 中華癌醫會誌. 2008; 24(5): 277 -281

Clinical Features • Common Symptoms • Common Physical Sign – – – – 8 Abdominal pain Weight loss Weakness Fullness and anorexia Abdominal Swelling Jaundice Vomiting – – – – Hepatomegaly Hepatic bruit Ascites Splenomegaly Jaundice Wasting Fever De. Vita VT, et al. Devita, Hellman & Rosenberg’s Cancer: Principles & Practice of Oncology, 8 th Edition

Diagnosis • Radiology – – X-ray Sonography Computed tomography (CT) Magnetic resonance imaging (MRI) • Biopsy • Alpha-fetoprotein (AFP) Serology 9 De. Vita VT, et al. Devita, Hellman & Rosenberg’s Cancer: Principles & Practice of Oncology, 8 th Edition

Treatment Option • Surgery – Partial hepatectomy – Liver transplantation • Local Ablative Therapy – – – 10 Cryosurgery Microwave ablation Ethanol injection Acetic acid injection Radiofrequecy ablation (RFA) • Regional Therapy (hepatic artery transcatheter treatments) – Transarterial chemotherapy – Transarterial embolization (TAE) – Transarterial chemoembolization (TACE) – Transarterial radiotherapy – 90 Y microspheres – 131 I lipiodol • Conformal external-beam radiation therapy • Systemic Therapy De. Vita VT, et al. Devita, Hellman & Rosenberg’s Cancer: Principles & Practice of Oncology, 8 th Edition


Staging System • Prognosis – – TNM Okuda CLIP (Cancer of the Liver Italian Program) JIS (Japanese Integrated Score) • Treatment – BCLC (Barcelona Clinic Liver Cancer) 12
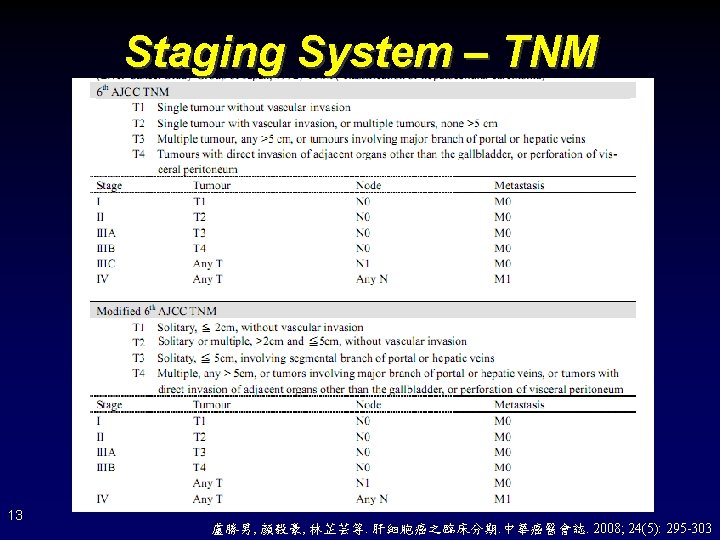
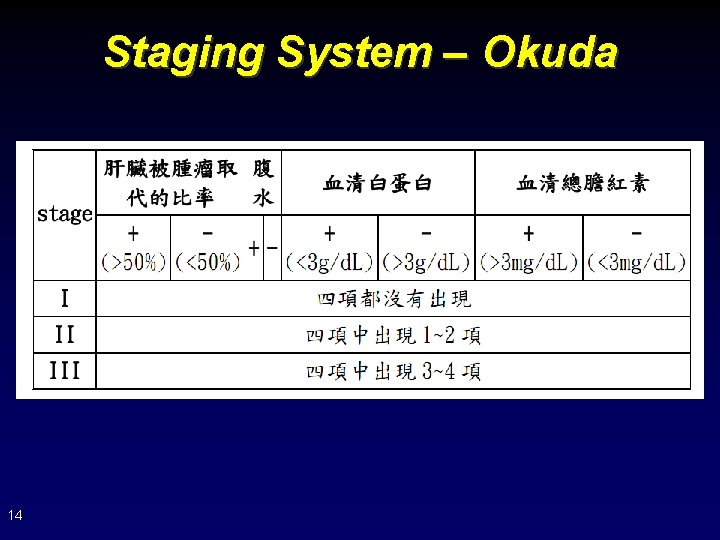
Staging System – Okuda 14
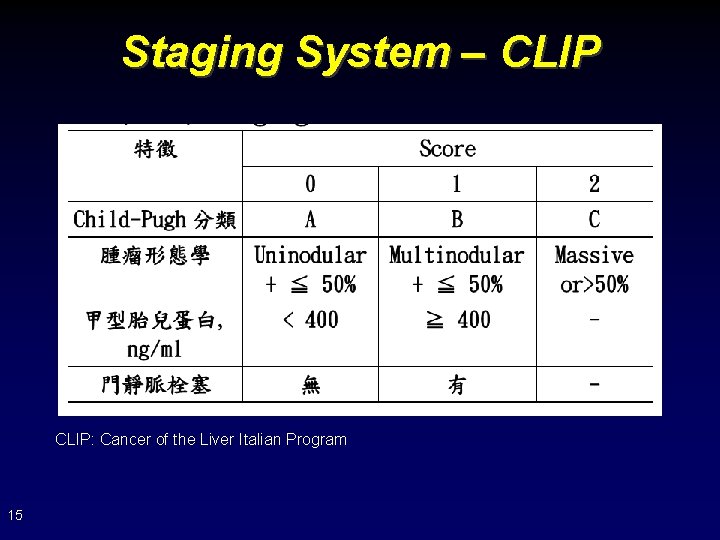
Staging System – CLIP: Cancer of the Liver Italian Program 15

Staging System – JIS 16 JIS: Japanese Integrated Score
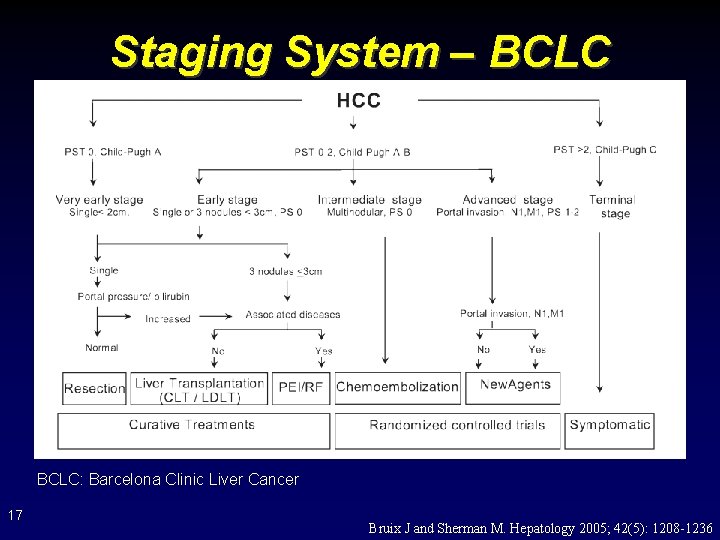
Staging System – BCLC: Barcelona Clinic Liver Cancer 17 Bruix J and Sherman M. Hepatology 2005; 42(5): 1208 -1236
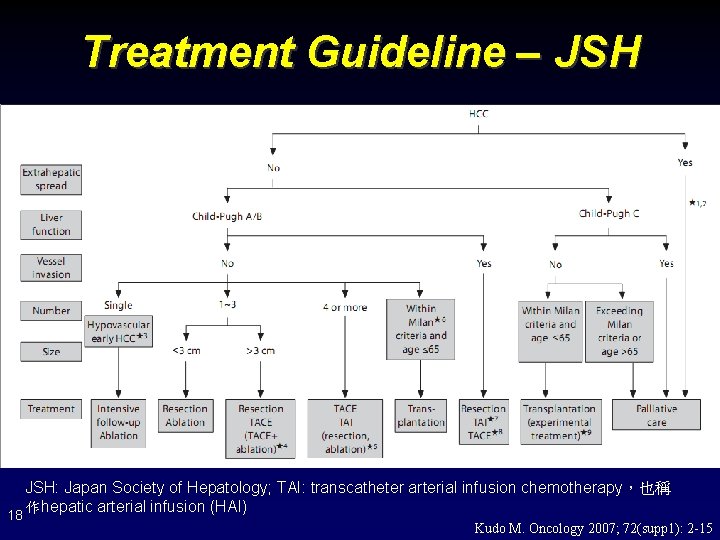
Treatment Guideline – JSH: Japan Society of Hepatology; TAI: transcatheter arterial infusion chemotherapy,也稱 作hepatic arterial infusion (HAI) 18 Kudo M. Oncology 2007; 72(supp 1): 2 -15


P. I. C. O. One-sentenced Question(請用一句話表達你的問題) As to advanced HCC patient, can sorafenib improve survival ? Patient or Problem (描述病患、疾病或病徵的型態) Hepatocellular carcinoma Intervention or Investigation (包括治療、檢驗、或預後因子) Sorafenib Comparison Placebo (通常用於比較目前的治療或診斷) Outcome (對病患有意義的臨床結果) Survival Type of Question ■ Therapy □ Diagnosis □ Prognosis □ Harm □ Others______ Type of Study Design ■ Meta-analysis ■ RCT □ Cohort Studies □ Case Control Studies □ Case Series □ Case Reports 20

Five A’s • Assess:了解臨床的需求(Clinical Problem) • Ask :發現問題的所在(Question) • Acquire :找尋最好的資料(Best Evidence) • Appraise :分析資料(Validity, Importance) • Apply :應用在病人身上(Patient) 21
22
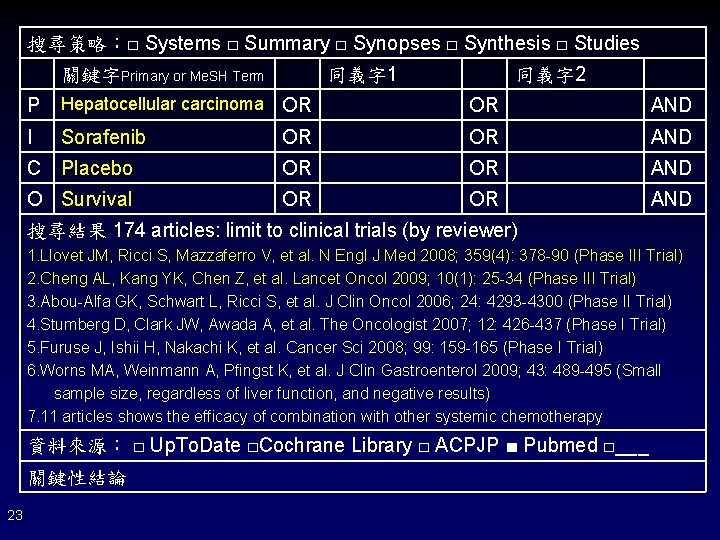
搜尋策略:□ Systems □ Summary □ Synopses □ Synthesis □ Studies 關鍵字Primary or Me. SH Term 同義字 1 同義字 2 P Hepatocellular carcinoma OR OR AND I Sorafenib OR OR AND C Placebo OR OR AND O Survival OR OR AND 搜尋結果 174 articles: limit to clinical trials (by reviewer) 1. Llovet JM, Ricci S, Mazzaferro V, et al. N Engl J Med 2008; 359(4): 378 -90 (Phase III Trial) 2. Cheng AL, Kang YK, Chen Z, et al. Lancet Oncol 2009; 10(1): 25 -34 (Phase III Trial) 3. Abou-Alfa GK, Schwart L, Ricci S, et al. J Clin Oncol 2006; 24: 4293 -4300 (Phase II Trial) 4. Stumberg D, Clark JW, Awada A, et al. The Oncologist 2007; 12: 426 -437 (Phase I Trial) 5. Furuse J, Ishii H, Nakachi K, et al. Cancer Sci 2008; 99: 159 -165 (Phase I Trial) 6. Worns MA, Weinmann A, Pfingst K, et al. J Clin Gastroenterol 2009; 43: 489 -495 (Small sample size, regardless of liver function, and negative results) 7. 11 articles shows the efficacy of combination with other systemic chemotherapy 資料來源: □ Up. To. Date □Cochrane Library □ ACPJP ■ Pubmed □___ 關鍵性結論 23

Five A’s • Assess:了解臨床的需求(Clinical Problem) • Ask :發現問題的所在(Question) • Acquire :找尋最好的資料(Best Evidence) • Appraise :分析資料(Validity, Importance) • Apply :應用在病人身上(Patient) 24

Sorafenib in Advanced Hepatocellular Carcinoma N Engl J Med 2008; 359: 378 -90 Llovet JM, Ricci S, Mazzaferro V, Hilgard P, Gane E, et al. 25

Study Design • Study Design: Randomized, double-blinded, placebo-controlled trial • Study Place: 121 centers in 21 countries in Europe, North America, South America, and Australia • Intervention: sorafenib 400 mg BID or placebo • Inclusion Criteria: 1. Advanced HCC, confirmed by pathological analysis 2. ECOG performance status of 2 or less 3. Child-Pugh liver function class A 4. Adequate hematological (PLT, Hb), hepatic (Alb, Bil-T, GOT, GPT), and renal function (s. Cr) • Exclusion Criteria: 1. Previously received molecularly targeted therapy or systemic treatment 26

Outcome Assessment • The Primary Outcome – Overall Survival – Time to Symptomatic Progression: FHSI 8↓ 4, ECOG progress to 4, or death • The Secondary Outcome – Time to Radiological Progression: by RECIST – The Disease-control Rate: CR+PR+SD – Safety 27 FHSI 8 Questionnaire:用以評估HCC病人症狀的嚴重程度 CR: complete response; PR: partial response; SD: stable disease
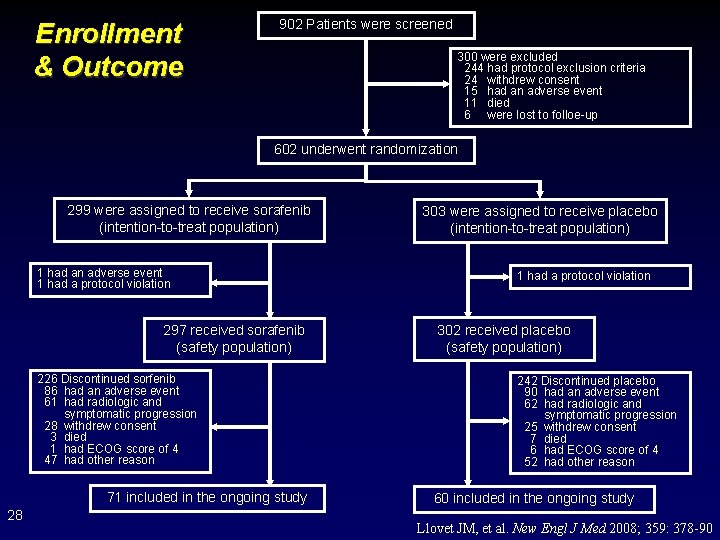
Enrollment & Outcome 902 Patients were screened 300 were excluded 244 had protocol exclusion criteria 24 withdrew consent 15 had an adverse event 11 died 6 were lost to folloe-up 602 underwent randomization 299 were assigned to receive sorafenib (intention-to-treat population) 1 had an adverse event 1 had a protocol violation 297 received sorafenib (safety population) 226 Discontinued sorfenib 86 had an adverse event 61 had radiologic and symptomatic progression 28 withdrew consent 3 died 1 had ECOG score of 4 47 had other reason 71 included in the ongoing study 28 303 were assigned to receive placebo (intention-to-treat population) 1 had a protocol violation 302 received placebo (safety population) 242 Discontinued placebo 90 had an adverse event 62 had radiologic and symptomatic progression 25 withdrew consent 7 died 6 had ECOG score of 4 52 had other reason 60 included in the ongoing study Llovet JM, et al. New Engl J Med 2008; 359: 378 -90
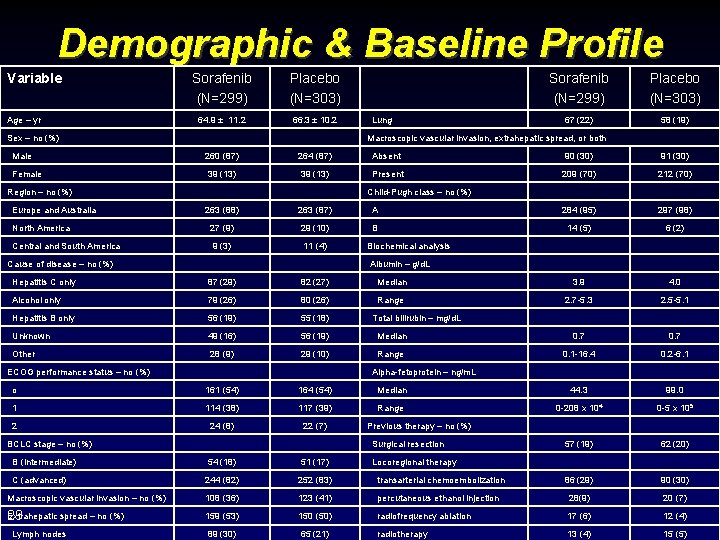
Demographic & Baseline Profile Variable Age – yr Sorafenib (N=299) Placebo (N=303) 64. 9 ± 11. 2 66. 3 ± 10. 2 Sex – no (%) Lung Sorafenib (N=299) Placebo (N=303) 67 (22) 58 (19) Macroscopic vascular invasion, extrahepatic spread, or both Male 260 (87) 264 (87) Absent 90 (30) 91 (30) Female 39 (13) Present 209 (70) 212 (70) Region – no (%) Europe and Australia Child-Pugh class – no (%) 263 (88) 263 (87) A 284 (95) 297 (98) North America 27 (9) 29 (10) B 14 (5) 6 (2) Central and South America 9 (3) 11 (4) Cause of disease – no (%) Biochemical analysis Albumin – g/d. L Hepatitis C only 87 (29) 82 (27) Median 3. 9 4. 0 Alcohol only 79 (26) 80 (26) Range 2. 7 -5. 3 2. 5 -5. 1 Hepatitis B only 56 (19) 55 (18) Unknown 49 (16) 56 (19) Median 0. 7 Other 28 (9) 29 (10) Range 0. 1 -16. 4 0. 2 -6. 1 44. 3 99. 0 ECOG performance status – no (%) o Total bilirubin – mg/d. L Alpha-fetoprotein – ng/m. L 161 (54) 164 (54) 1 114 (38) 117 (39) 2 24 (8) 22 (7) BCLC stage – no (%) Median Range 0 -208 x 104 0 -5 x 105 Previous therapy – no (%) Surgical resection 57 (19) 62 (20) 86 (29) 90 (30) B (intermediate) 54 (18) 51 (17) C (advanced) 244 (82) 252 (83) transarterial chemoembolization Macroscopic vascular invasion – no (%) 108 (36) 123 (41) percutaneous ethanol injection 28(9) 20 (7) Extrahepatic spread – no (%) 29 159 (53) 150 (50) radiofrequency ablation 17 (6) 12 (4) 89 (30) 65 (21) radiotherapy 13 (4) 15 (5) Lymph nodes Locoregional therapy

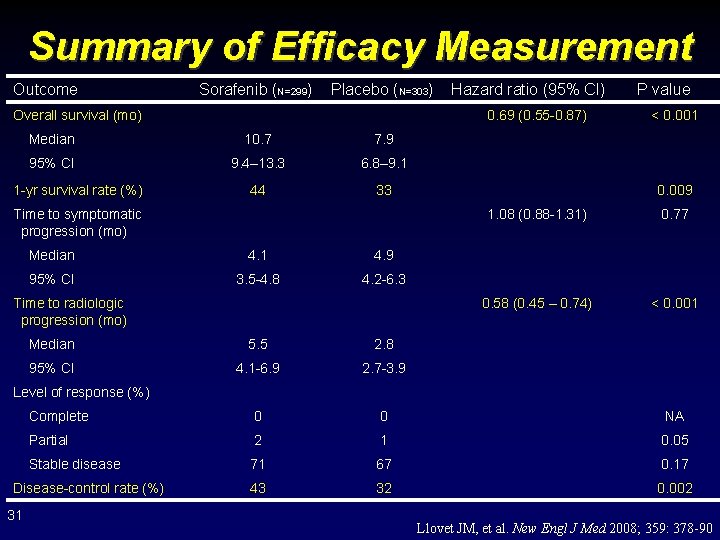
Summary of Efficacy Measurement Outcome Sorafenib (N=299) Placebo (N=303) Overall survival (mo) 0. 69 (0. 55 -0. 87) Median 10. 7 7. 9 95% CI 9. 4– 13. 3 6. 8– 9. 1 44 33 1 -yr survival rate (%) Hazard ratio (95% CI) Time to symptomatic progression (mo) Median 4. 1 4. 9 95% CI 3. 5 -4. 8 4. 2 -6. 3 Time to radiologic progression (mo) P value < 0. 001 0. 009 1. 08 (0. 88 -1. 31) 0. 77 0. 58 (0. 45 – 0. 74) < 0. 001 Median 5. 5 2. 8 95% CI 4. 1 -6. 9 2. 7 -3. 9 Complete 0 0 NA Partial 2 1 0. 05 Stable disease 71 67 0. 17 43 32 0. 002 Level of response (%) Disease-control rate (%) 31 Llovet JM, et al. New Engl J Med 2008; 359: 378 -90
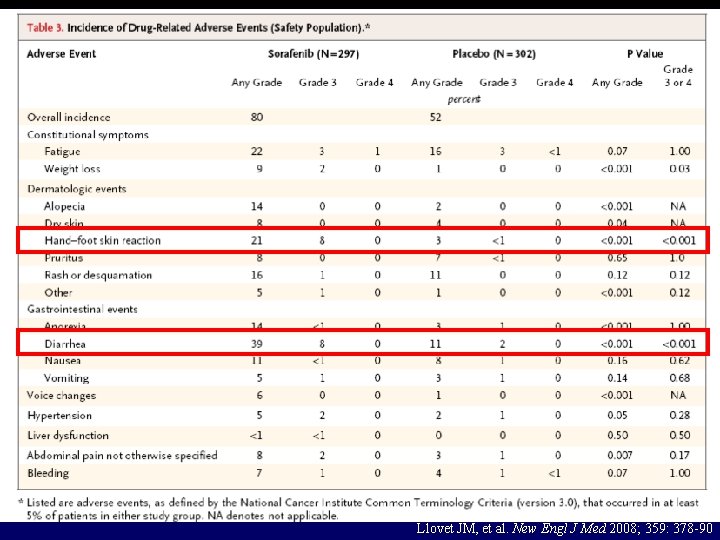
32 Llovet JM, et al. New Engl J Med 2008; 359: 378 -90
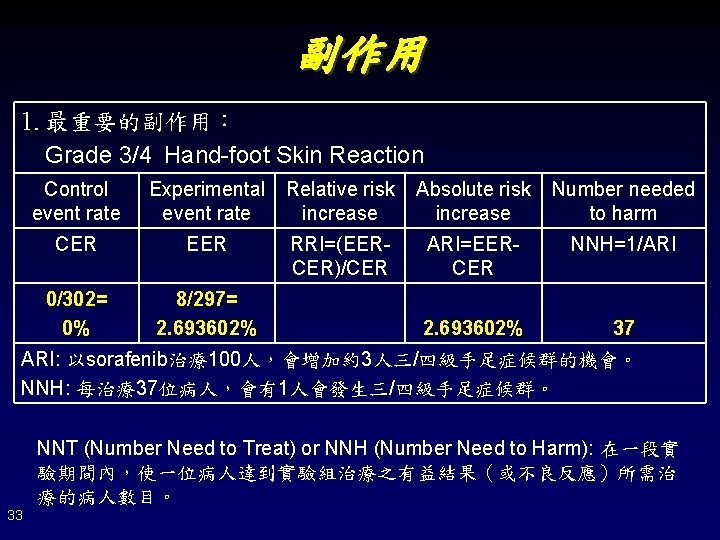
副作用 1. 最重要的副作用: Grade 3/4 Hand-foot Skin Reaction Control event rate Experimental event rate Relative risk increase Absolute risk increase Number needed to harm CER EER RRI=(EERCER)/CER ARI=EERCER NNH=1/ARI 0/302= 0% 8/297= 2. 693602% 37 ARI: 以sorafenib治療100人,會增加約3人三/四級手足症候群的機會。 NNH: 每治療37位病人,會有1人會發生三/四級手足症候群。 NNT (Number Need to Treat) or NNH (Number Need to Harm): 在一段實 驗期間內,使一位病人達到實驗組治療之有益結果(或不良反應)所需治 療的病人數目。 33
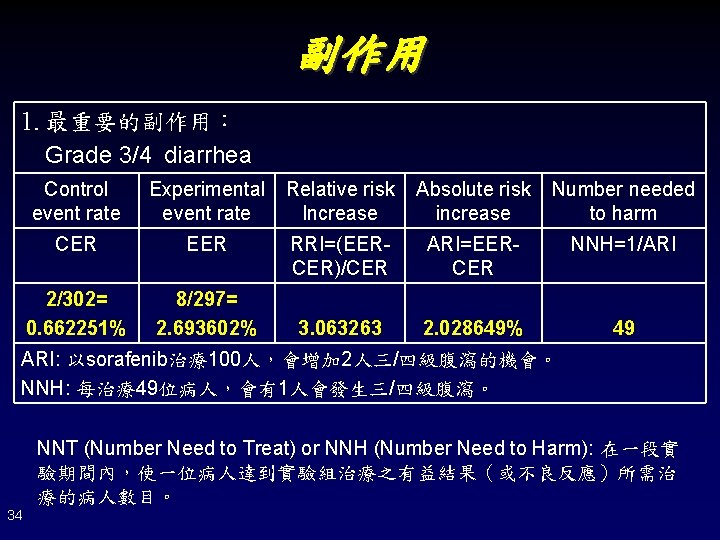
副作用 1. 最重要的副作用: Grade 3/4 diarrhea Control event rate Experimental event rate Relative risk Increase Absolute risk increase Number needed to harm CER EER RRI=(EERCER)/CER ARI=EERCER NNH=1/ARI 2/302= 0. 662251% 8/297= 2. 693602% 3. 063263 2. 028649% 49 ARI: 以sorafenib治療100人,會增加 2人三/四級腹瀉的機會。 NNH: 每治療49位病人,會有1人會發生三/四級腹瀉。 NNT (Number Need to Treat) or NNH (Number Need to Harm): 在一段實 驗期間內,使一位病人達到實驗組治療之有益結果(或不良反應)所需治 療的病人數目。 34

Efficacy and Safety of Sorafenib in Patients in The Asia-Pacific Region With Advanced Hepatocellular Carcinoma: A Phase III Randomized, Double-blind, Placebo-Controlled Trial Lancet Oncol 2009; 10: 25 -34 Cheng AL, Kang YK, Chen ZD, Tsao CJ, Qin SK, et al. 35
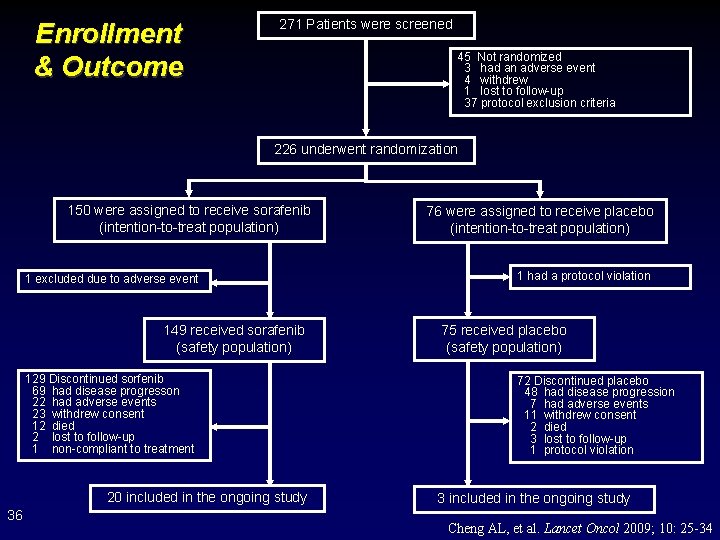
Enrollment & Outcome 271 Patients were screened 45 Not randomized 3 had an adverse event 4 withdrew 1 lost to follow-up 37 protocol exclusion criteria 226 underwent randomization 150 were assigned to receive sorafenib (intention-to-treat population) 1 excluded due to adverse event 149 received sorafenib (safety population) 129 Discontinued sorfenib 69 had disease progresson 22 had adverse events 23 withdrew consent 12 died 2 lost to follow-up 1 non-compliant to treatment 20 included in the ongoing study 36 76 were assigned to receive placebo (intention-to-treat population) 1 had a protocol violation 75 received placebo (safety population) 72 Discontinued placebo 48 had disease progression 7 had adverse events 11 withdrew consent 2 died 3 lost to follow-up 1 protocol violation 3 included in the ongoing study Cheng AL, et al. Lancet Oncol 2009; 10: 25 -34
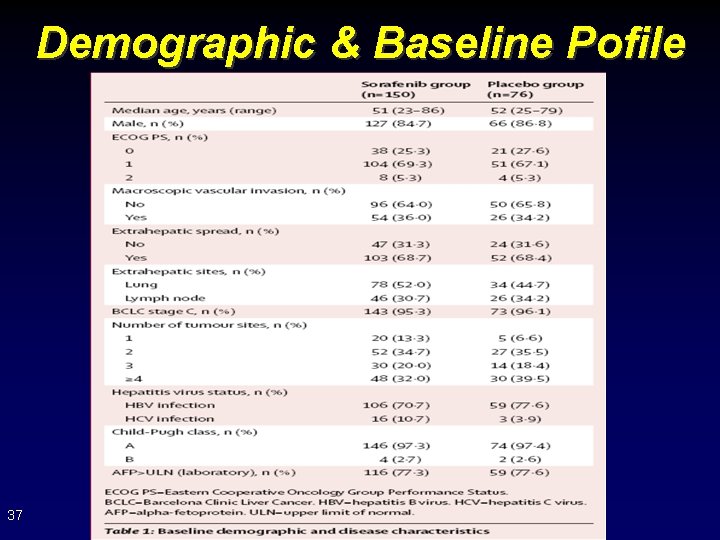
Demographic & Baseline Pofile 37

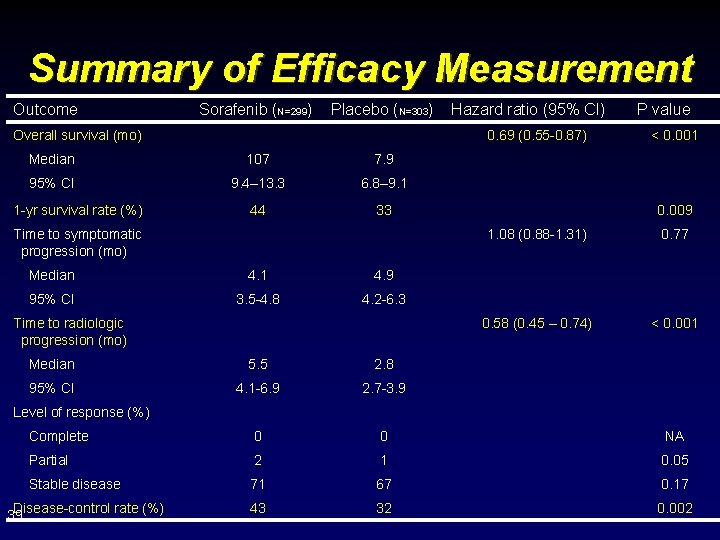
Summary of Efficacy Measurement Outcome Sorafenib (N=299) Placebo (N=303) Overall survival (mo) 0. 69 (0. 55 -0. 87) Median 107 7. 9 95% CI 9. 4– 13. 3 6. 8– 9. 1 44 33 1 -yr survival rate (%) Hazard ratio (95% CI) Time to symptomatic progression (mo) Median 4. 1 4. 9 95% CI 3. 5 -4. 8 4. 2 -6. 3 Time to radiologic progression (mo) P value < 0. 001 0. 009 1. 08 (0. 88 -1. 31) 0. 77 0. 58 (0. 45 – 0. 74) < 0. 001 Median 5. 5 2. 8 95% CI 4. 1 -6. 9 2. 7 -3. 9 Complete 0 0 NA Partial 2 1 0. 05 Stable disease 71 67 0. 17 43 32 0. 002 Level of response (%) Disease-control rate (%) 39
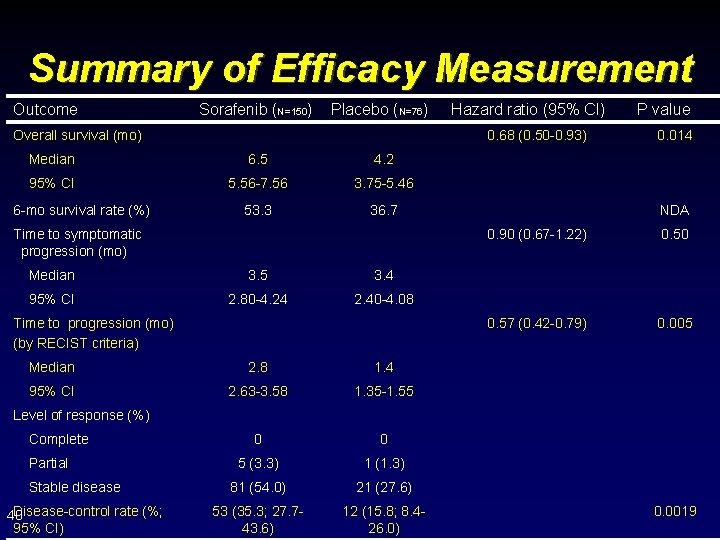
Summary of Efficacy Measurement Outcome Sorafenib (N=150) Placebo (N=76) Overall survival (mo) 0. 68 (0. 50 -0. 93) Median 6. 5 4. 2 95% CI 5. 56 -7. 56 3. 75 -5. 46 53. 3 36. 7 6 -mo survival rate (%) Hazard ratio (95% CI) Time to symptomatic progression (mo) Median 3. 5 3. 4 95% CI 2. 80 -4. 24 2. 40 -4. 08 Time to progression (mo) (by RECIST criteria) Median 2. 8 1. 4 95% CI 2. 63 -3. 58 1. 35 -1. 55 0 0 5 (3. 3) 1 (1. 3) 81 (54. 0) 21 (27. 6) 53 (35. 3; 27. 743. 6) 12 (15. 8; 8. 426. 0) P value 0. 014 NDA 0. 90 (0. 67 -1. 22) 0. 50 0. 57 (0. 42 -0. 79) 0. 005 Level of response (%) Complete Partial Stable disease Disease-control rate (%; 40 95% CI) 0. 0019
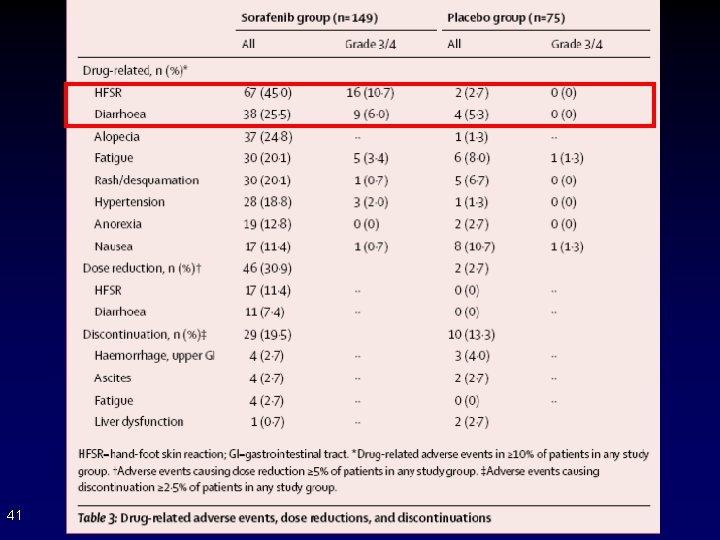
41
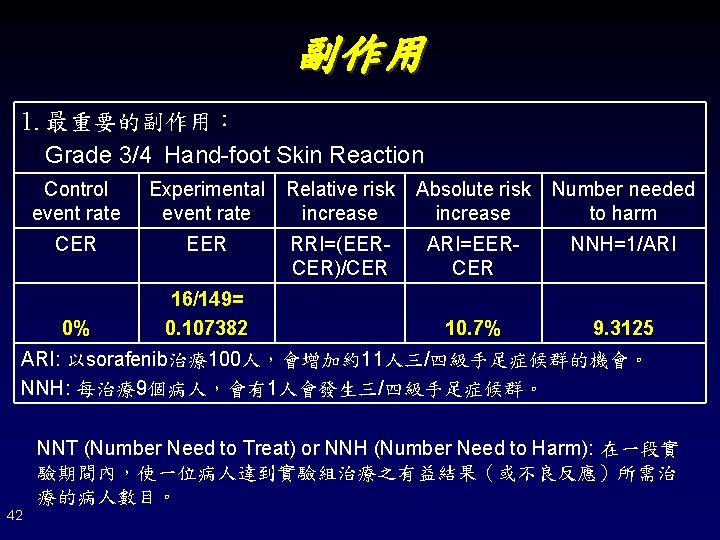
副作用 1. 最重要的副作用: Grade 3/4 Hand-foot Skin Reaction Control event rate Experimental event rate Relative risk increase Absolute risk increase Number needed to harm CER EER RRI=(EERCER)/CER ARI=EERCER NNH=1/ARI 0% 16/149= 0. 107382 10. 7% 9. 3125 ARI: 以sorafenib治療100人,會增加約11人三/四級手足症候群的機會。 NNH: 每治療9個病人,會有1人會發生三/四級手足症候群。 NNT (Number Need to Treat) or NNH (Number Need to Harm): 在一段實 驗期間內,使一位病人達到實驗組治療之有益結果(或不良反應)所需治 療的病人數目。 42
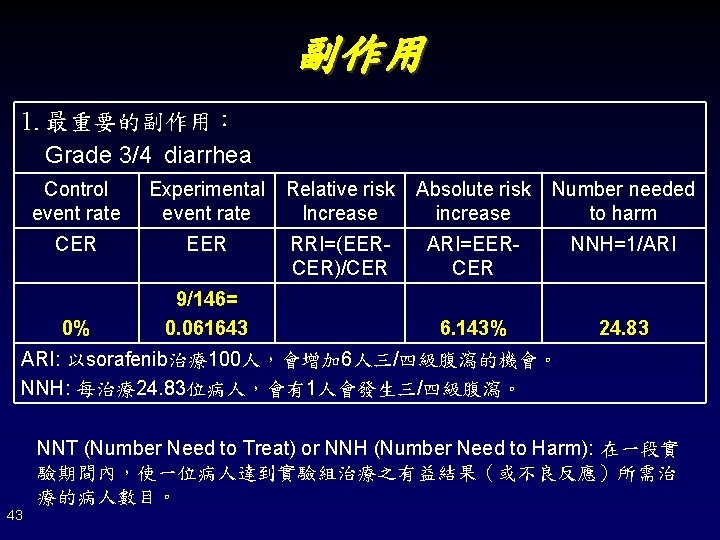
副作用 1. 最重要的副作用: Grade 3/4 diarrhea Control event rate Experimental event rate Relative risk Increase Absolute risk increase Number needed to harm CER EER RRI=(EERCER)/CER ARI=EERCER NNH=1/ARI 0% 9/146= 0. 061643 6. 143% 24. 83 ARI: 以sorafenib治療100人,會增加 6人三/四級腹瀉的機會。 NNH: 每治療24. 83位病人,會有1人會發生三/四級腹瀉。 NNT (Number Need to Treat) or NNH (Number Need to Harm): 在一段實 驗期間內,使一位病人達到實驗組治療之有益結果(或不良反應)所需治 療的病人數目。 43
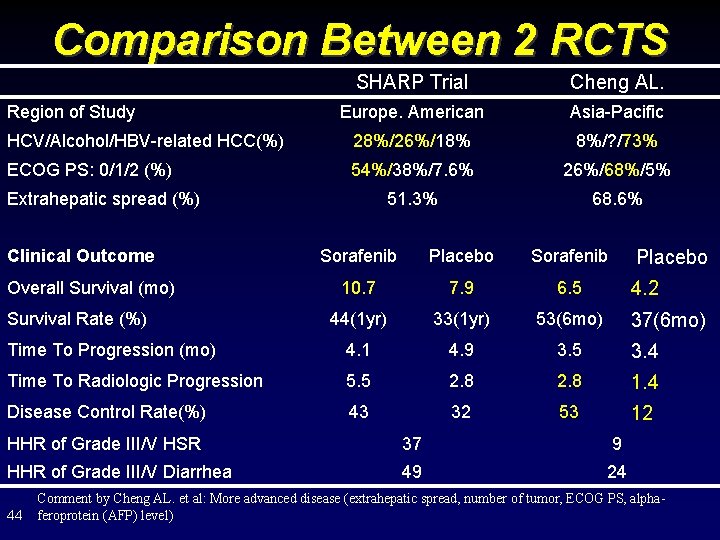
Comparison Between 2 RCTS SHARP Trial Cheng AL. Europe. American Asia-Pacific HCV/Alcohol/HBV-related HCC(%) 28%/26%/18% 8%/? /73% ECOG PS: 0/1/2 (%) 54%/38%/7. 6% 26%/68%/5% 51. 3% 68. 6% Region of Study Extrahepatic spread (%) Clinical Outcome Sorafenib Placebo Sorafenib 10. 7 7. 9 6. 5 44(1 yr) 33(1 yr) 53(6 mo) Time To Progression (mo) 4. 1 4. 9 3. 5 3. 4 Time To Radiologic Progression 5. 5 2. 8 1. 4 Disease Control Rate(%) 43 32 53 12 Overall Survival (mo) Survival Rate (%) Placebo 4. 2 37(6 mo) HHR of Grade III/V HSR 37 9 HHR of Grade III/V Diarrhea 49 24 44 Comment by Cheng AL. et al: More advanced disease (extrahepatic spread, number of tumor, ECOG PS, alphaferoprotein (AFP) level)
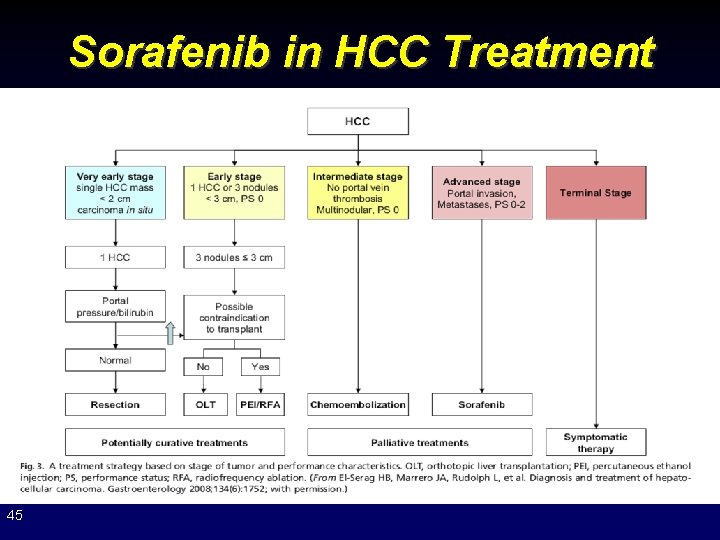
Sorafenib in HCC Treatment 45

Five A’s • Assess:了解臨床的需求(Clinical Problem) • Ask :發現問題的所在(Question) • Acquire :找尋最好的資料(Best Evidence) • Appraise :分析資料(Validity, Importance) • Apply :應用在病人身上(Patient) 46

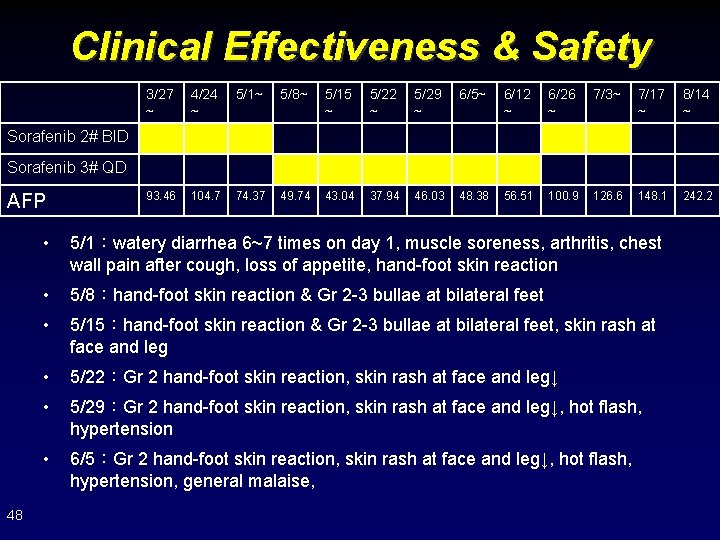
Clinical Effectiveness & Safety 3/27 ~ 4/24 ~ 5/1~ 5/8~ 5/15 ~ 5/22 ~ 5/29 ~ 6/5~ 6/12 ~ 6/26 ~ 7/3~ 7/17 ~ 8/14 ~ 93. 46 104. 7 74. 37 49. 74 43. 04 37. 94 46. 03 48. 38 56. 51 100. 9 126. 6 148. 1 242. 2 Sorafenib 2# BID Sorafenib 3# QD AFP 48 • 5/1:watery diarrhea 6~7 times on day 1, muscle soreness, arthritis, chest wall pain after cough, loss of appetite, hand-foot skin reaction • 5/8:hand-foot skin reaction & Gr 2 -3 bullae at bilateral feet • 5/15:hand-foot skin reaction & Gr 2 -3 bullae at bilateral feet, skin rash at face and leg • 5/22:Gr 2 hand-foot skin reaction, skin rash at face and leg↓ • 5/29:Gr 2 hand-foot skin reaction, skin rash at face and leg↓, hot flash, hypertension • 6/5:Gr 2 hand-foot skin reaction, skin rash at face and leg↓, hot flash, hypertension, general malaise,

Thank You For Your Listening 49

Reference • • • 50 教科書 De. Vita VT, et al. Devita, Hellman & Rosenberg's Cancer: Principles & Practice of Oncology, 8 th Edition 臨床準則 AASLD Practice Guideline: Bruix J and Sherman M. Management of hepatocellular carcinoma. Hepatology 2005; 42(5): 1208 -1236 JSH Guideline: Kudo M, Okunoue T, and Clinical Practice Manual of HCC Expert Panel. Management of hepatocellular carcinoma in Japan: consensus-based clinical practice manual proposed by the Japan Society of Hepatology. Oncology 2007; 72(supp 1): 2 -15 英文期刊 Llovet JM, Ricci S, Mazzaferro V, Hilgard P, Gane E, et al. Sorafenib in Advanced Hepatocellular Carcinoma. N Engl J Med 2008; 359: 378 -90 Cheng AL, Kang YK, Chen ZD, Tsao CJ, Qin SK, et al. Efficacy and Safety of Sorafenib in Patients in The Asia-Pacific Region With Advanced Hepatocellular Carcinoma: A Phase III Randomized, Double-blind, Placebo-Controlled Trial. Lancet Oncol 2009; 10: 25 -34 中文期刊 林志陵, 高嘉宏. 肝癌的流行病學. 中華癌醫會誌. 2008; 24(5): 277 -281 盧勝男, 顏毅豪, 林芷芸等. 肝細胞癌之臨床分期. 中華癌醫會誌. 2008; 24(5): 295 -303

Sorafenib in Advanced Hepatocellular Carcinoma N Engl J Med 2008; 359: 378 -90 Llovet JM, Ricci S, Mazzaferro V, Hilgard P, Gane E, et al. 51

Efficacy and Safety of Sorafenib in Patients in The Asia-Pacific Region With Advanced Hepatocellular Carcinoma: A Phase III Randomized, Double-blind, Placebo-Controlled Trial Lancet Oncol 2009; 10: 25 -34 Cheng AL, Kang YK, Chen ZD, Tsao CJ, Qin SK, et al. 52

53

54

55

56

57

Outline • 5 Steps in EBM (5 As) – Assess – Ask • Background or foreground question, PICO – Acquire • Level of evidence, Searching skills – Appraise • Validity, importance, and practicability – Apply 58

What is Evidence-based Medicine ? • Evidence-based medicine is the integration of best research evidence with clinical expertise and patient values • ~ Dr. Sackett, et al 2001 Patient Concerns EBM Best research Clinical Expertise evidence 59


Ask Questions • Background Question – How does HBV infection progress to HCC? – How could we diagnose HCC? – How could we determine the stage of HCC? – When should we consider liver transplantation? • Foreground Question – Is ultrasound more accurate than computed tomography? – Is sorafenib more effective than doxorubicin-based chemotherapy? 61



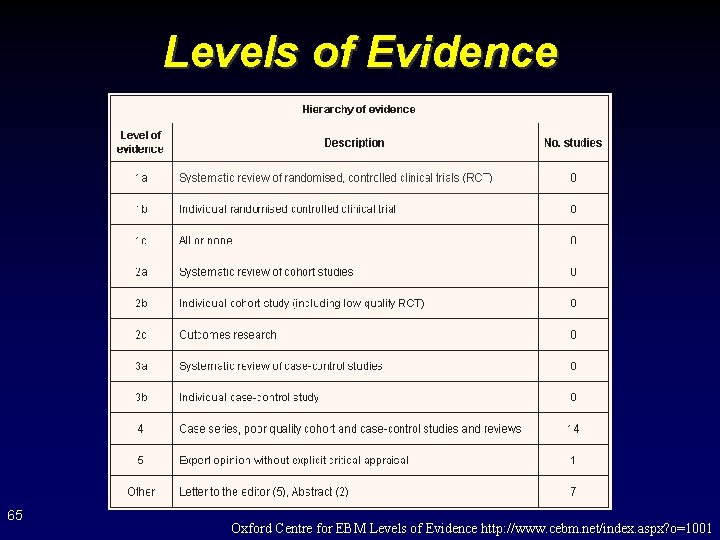
Levels of Evidence 65 Oxford Centre for EBM Levels of Evidence http: //www. cebm. net/index. aspx? o=1001

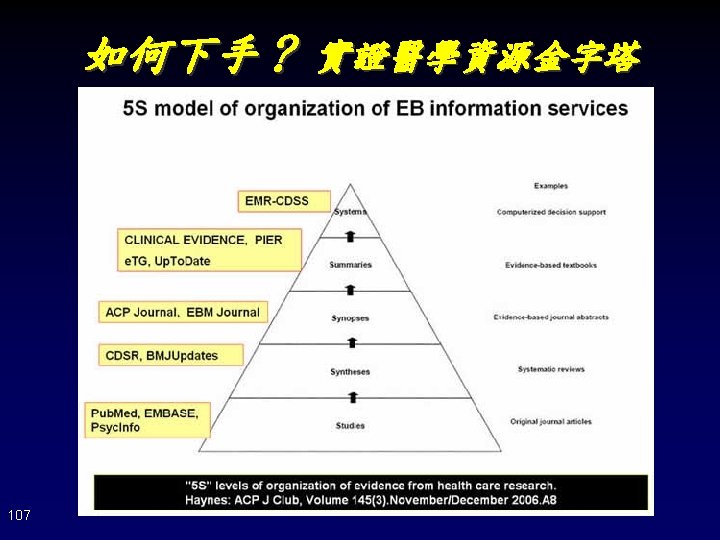
如何下手? 實證醫學資源金字塔 Systems 連結個別病歷的臨床知識與支援決策系統 Summaries 整合証據提供特定臨床問題之概述與建議 Up. To. Date Dyna. Med BMJ Clinical Evidence First. Consult ACP PIER Synopses 對單篇研究或回顧性文獻作摘要與評述 ACP Journal Club, Evidece-Based Medicine (Pub. Med, Ovid Medline) Syntheses 特定臨床問題的系統性評論文獻 Cochrane Database of Systematic Reviews Database of Abstracts of Reviews of Effects (Pub. Med, Ovid Medline): Systematic Reviews Studies 原始文獻 (Pub. Med, Ovid Medline, CINAHL, EMBASE Cochrane CENTRAL, Google scholar CEPS中文電子期刊, 中文期刊篇目索引) 66 越上層 • 資訊精粹 • 簡單的關鍵字 • 省時搜尋與評讀 • 由下層累積 越下層 • 文獻雜多 • 完整的關鍵字 • 費時搜尋與評讀 • 注重檢索技巧 • 資訊新穎

資訊檢索 - 關鍵字 • 選用Me. SH term (並避免雙引號、切截字元) – COX-2 selective inhibitor >> COX-2 inhibitor • 列舉同義字 – Conventional >> conventional OR traditional • 避免使用縮寫 – CVD >> cardiovascular diseases • 使用明確詞彙 – Cardiovascular risk >> cardiovascular diseases AND risk • 調整廣狹義詞 – Cardiovascular diseases <<>> myocardial infarction 68
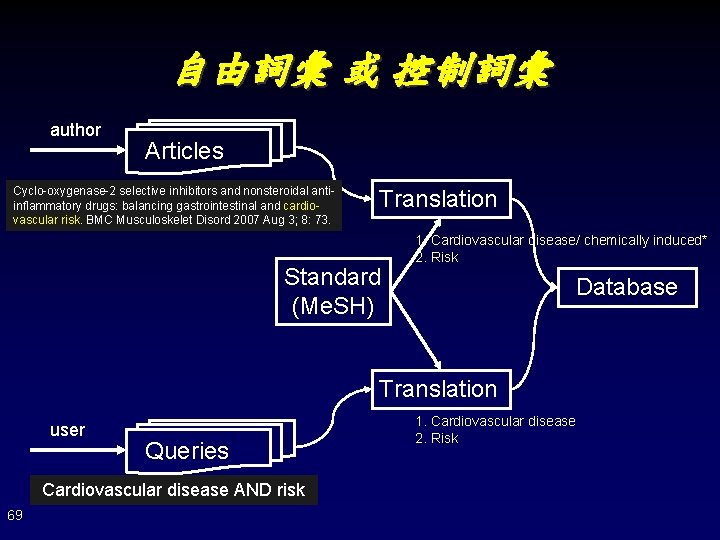
自由詞彙 或 控制詞彙 author Articles Cyclo-oxygenase-2 selective inhibitors and nonsteroidal antiinflammatory drugs: balancing gastrointestinal and cardiovascular risk. BMC Musculoskelet Disord 2007 Aug 3; 8: 73. Translation Standard (Me. SH) 1. Cardiovascular disease/ chemically induced* 2. Risk Database Translation user Queries Cardiovascular disease AND risk 69 1. Cardiovascular disease 2. Risk

資訊檢索概念 • 單字 或 片語 – Cardiovascular diseases OR "cardiovascular diseases" • 切截字 – Therap*: therapy, therapeutics • 同義字(用Me. SH可避免大部分的困擾) – 單複數:inhibitor/inhibitors;英美拼法:edema/oedema; 全名縮寫:cardiovascular disease/CVD;同義異形字: carotid ultrasonography/carotid ultrasound/carotid Doppler • 廣狹義詞 – Cardiovascular disease > myocardial infarction 70

72
Llovet JM, et al. Sorafenib in Advanced Hepatocellular Carcinoma. New Engl J Med 2008; 359: 378 -90 研究設計 人/動物、年齡、性別 73

資訊檢索 - 關鍵字 • 選用Me. SH term (並避免雙引號、切截字元) – COX-2 selective inhibitor >> COX-2 inhibitor • 列舉同義字 – Conventional >> conventional OR traditional • 避免使用縮寫 – CVD >> cardiovascular diseases • 使用明確詞彙 – Cardiovascular risk >> cardiovascular diseases AND risk • 調整廣狹義詞 – Cardiovascular diseases <<>> myocardial infarction 74

資訊檢索概念 • 單字 或 片語 – Cardiovascular diseases OR "cardiovascular diseases" • 切截字 – Therap*: therapy, therapeutics • 同義字(用Me. SH可避免大部分的困擾) – 單複數:inhibitor/inhibitors;英美拼法:edema/oedema; 全名縮寫:cardiovascular disease/CVD;同義異形字: carotid ultrasonography/carotid ultrasound/carotid Doppler • 廣狹義詞 – Cardiovascular disease > myocardial infarction 75

資訊檢索 - 關鍵字 • 選用Me. SH term (並避免雙引號、切截字元) – COX-2 selective inhibitor >> COX-2 inhibitor • 列舉同義字 – Conventional >> conventional OR traditional • 避免使用縮寫 – CVD >> cardiovascular diseases • 使用明確詞彙 – Cardiovascular risk >> cardiovascular diseases AND risk • 調整廣狹義詞 – Cardiovascular diseases <<>> myocardial infarction 76

80
Llovet JM, et al. Sorafenib in Advanced Hepatocellular Carcinoma. New Engl J Med 2008; 359: 378 -90 研究設計 人/動物、年齡、性別 81

Appraise The Evidence • Validity 可信度 • Importance 重要性 • Practicability 應用性 82

Validity • • Internal Validity: Is there a causal relationship between two variables? Criteria to establishing a cause-effect relationship: 1. Temporal Precedence: the cause must happen before the effect. 2. Co-variation: If X then Y, and if not X then not Y. Or a dose -response relationship: if more of the X then more of Y, and if less of X then less of Y. 3. Without other Plausible Explanations: the problem with the “third variable” or “missing variable”. Other variable(s) causes the outcome. It is important but not always possible to “rule out” plausible explanations. 83

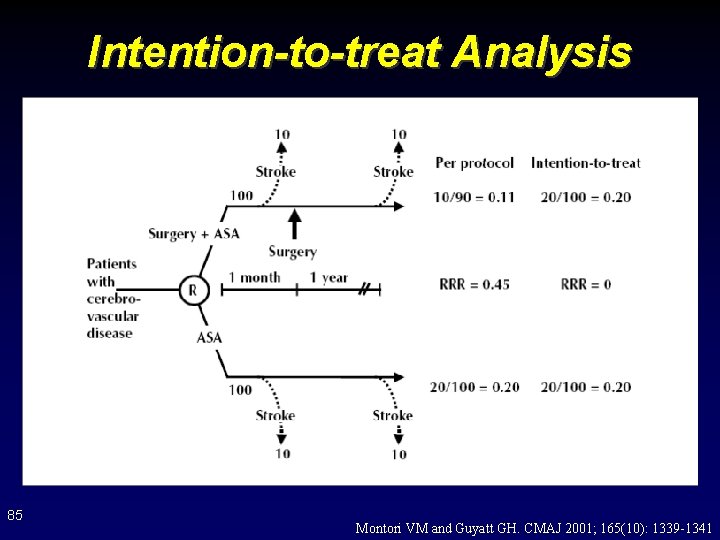
Intention-to-treat Analysis 85 Montori VM and Guyatt GH. CMAJ 2001; 165(10): 1339 -1341


Appraise The Evidence • Validity 可信度 • Importance 重要性 • Practicability 應用性 87

Critically Appraisal • 本篇文獻研究的結果在臨床上重要嗎? • 強度:本實驗是否有效或有害 – 有效: • RRR: relative risk reduction • ARR: absolute risk reduction • NNT: number need to treat – 有害: • RR: relative risk • OR: odd ratio • 效度:本實驗是否準確? – 95% confidence interval (95%CI) 88

Appraise The Evidence • Validity 可信度 • Importance 重要性 • Practicability 應用性 89



92

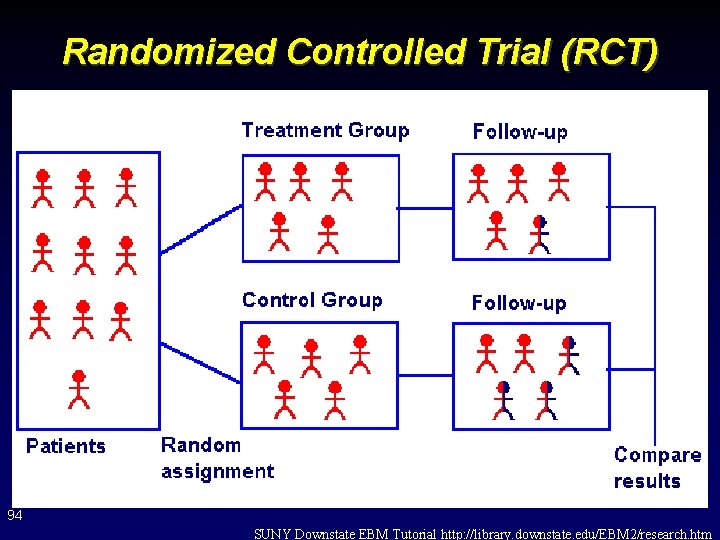
Randomized Controlled Trial (RCT) 94 SUNY Downstate EBM Tutorial http: //library. downstate. edu/EBM 2/research. htm
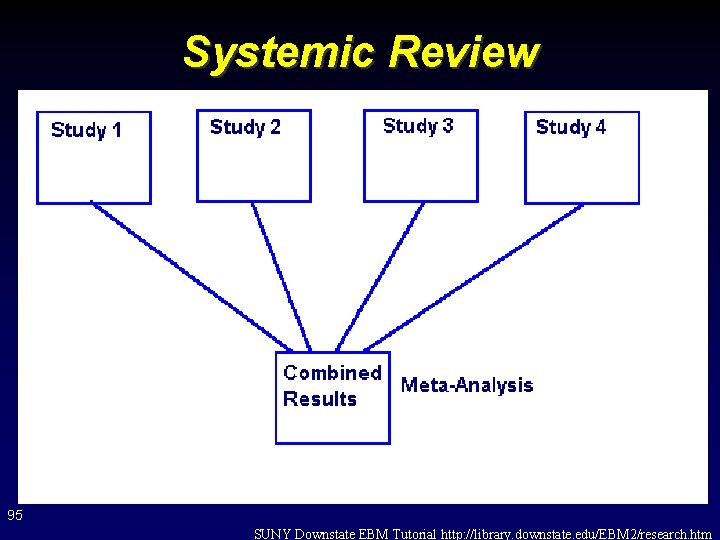
Systemic Review 95 SUNY Downstate EBM Tutorial http: //library. downstate. edu/EBM 2/research. htm
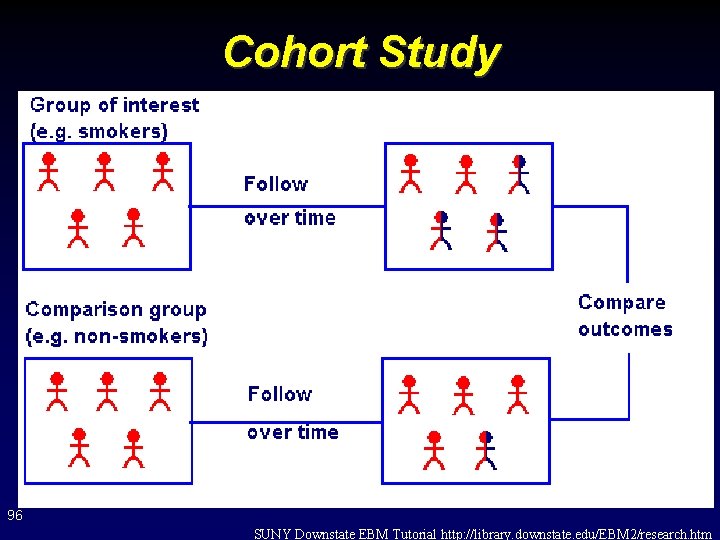
Cohort Study 96 SUNY Downstate EBM Tutorial http: //library. downstate. edu/EBM 2/research. htm
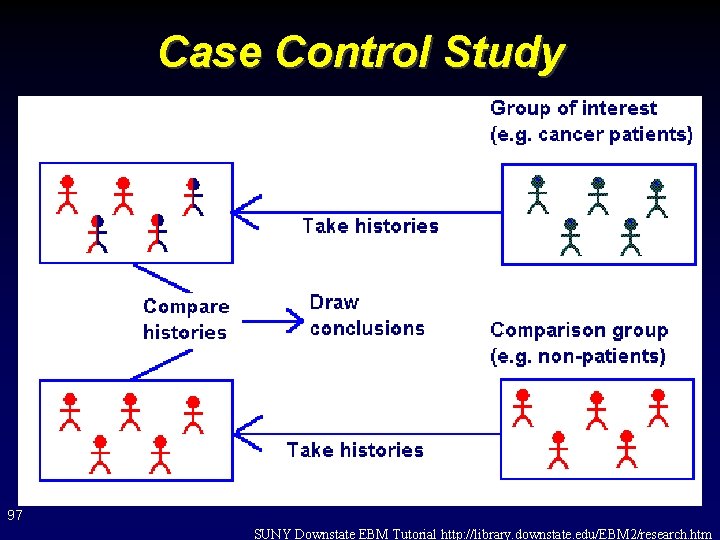
Case Control Study 97 SUNY Downstate EBM Tutorial http: //library. downstate. edu/EBM 2/research. htm
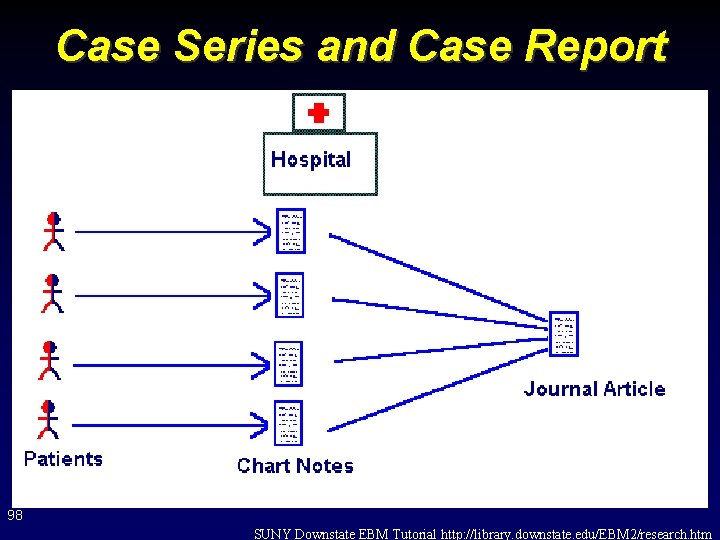
Case Series and Case Report 98 SUNY Downstate EBM Tutorial http: //library. downstate. edu/EBM 2/research. htm

99


Practice 實用性 (2) 2) 根據疾病的發生率(PEER),計算本治療在國內的 NNT、NNH。 • PEER method,假設RRR,RRI不受地區影響改變。 • NNT = 1/(PEER x RRR) = 1/(0. 162 x 0. 13) = 47. 48 • NNH = 1/(PEER x RRI) = 1/(0. 07 x 0. 142) = 100. 60 3) 根據個人經驗粗估本疾病治療好處及副作用的危險性是 論文中的 f 倍 101 • ft = 1 NNTpatient = NNTreported/ft = 47. 48 • fh =1 NNHpateint = NNHreported/fh = 100. 60

Practice 實用性 (3) 4) 根據病人的期望,比較患者接受此治療的好處與 壞處。 • • • Sdisease(得到疾病的嚴重度)= 0. 1 Sharm(進行治療的副作用)= 0. 9 S(severity factor) = Sharm/Sdisease = 9 5) 患者接受此治療得到的利害比(LHH, likelihood of being helped or harmed) • • 102 原始LHH = ARR: ARI = (1/NNT) : (1/NNH) = 2. 12 Adjusted LHH = [(1/NNT) x ft x S] : [(1/NNH) x fh] = 19. 08

Validity (1) • Definition: the best available approximation to the truth or falsity of a given proposition, inference, or conclusion. • Typology of validity – Conclusion Validity (statistical conclusion validity): Is there a relationship between the two variables? Ø The confidence we have on the conclusions we reach about relationships in our data (i. e. there is a significant correlation between two variables. ) It concerns only whether such a relationship exists, but not whether such a relationship is a causal one. – Internal Validity: Assuming that there is a relationship in this study, is the relationship a causal one? – External Validity: Assuming that there is a causal relationship between the constructs of the cause and the effect, can it be generalized to other persons, places or times. 103

Validity (2) • Typology of Validity (~ continued) – Content Validity is evaluated by showing how well the content of the test samples the class of situations or subject matter about which conclusions are to be drawn. – Criterion-related Validity is evaluated by comparing the test scores with one or more external variables (called criteria) considered to provide a direct measure of the characteristic or behavior in question. Ø Predictive validity indicates the extent to which an individual’s future level on the criterion is predicated from prior test performance. Ø Concurrent Validity indicates the extent to which the test scores estimate an individual’s present standing on the criterion. – Construct Validity is evaluated by investigating what qualities a test measures, that is, by determining the degree to which certain explanatory concepts or constructs account for performance on the test. 104
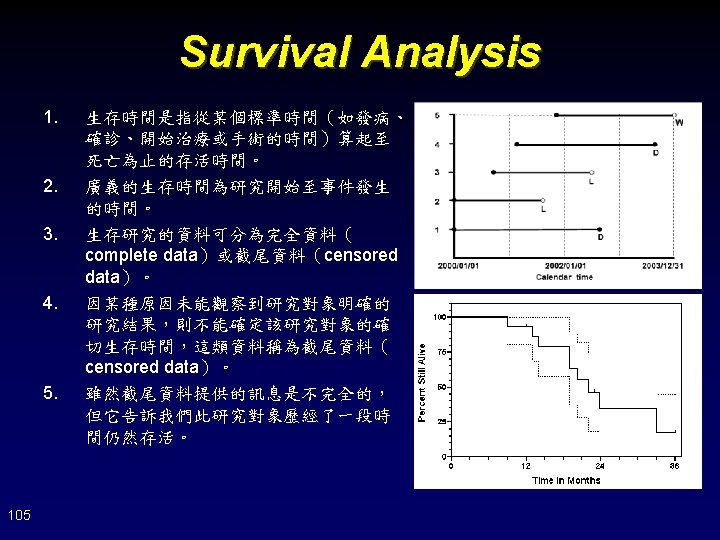
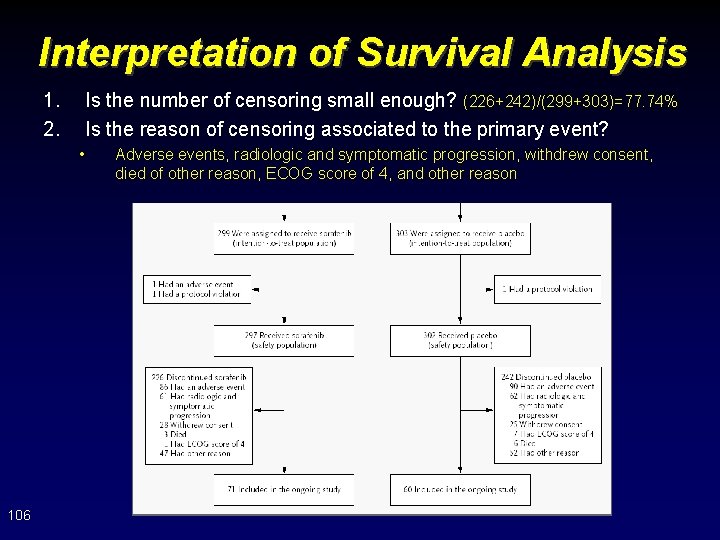
Interpretation of Survival Analysis 1. 2. Is the number of censoring small enough? (226+242)/(299+303)=77. 74% Is the reason of censoring associated to the primary event? • 106 Adverse events, radiologic and symptomatic progression, withdrew consent, died of other reason, ECOG score of 4, and other reason
前景問題 Foreground Questions 背景問題 Background Questions 經驗的累積 108
- Slides: 108