First Pharmacogenomic Analysis Using Whole Exome Sequencing to










- Slides: 20

First Pharmacogenomic Analysis Using Whole Exome Sequencing to Identify Novel Genetic Determinants of Clopidogrel Response Variability GIFT-EXOME ACC/i 2 2012

Exome Disclosures Consulting honoraria: Bristol-Myers Squibb/sanofi-aventis, Accumetrics, DSI/Lilly & Co. , Merck, Janssen, Astra. Zeneca, The Medicines Company, Medicure Equity Interest: Iverson Genetics Speaker Honoraria: DSI/Lilly, Astra. Zeneca Research Support: Bristol Meyers Squibb/sanofi-aventis, Quest Diagnostics, Accumetrics, Molecular Response GIFT was supported through an grant from BMS/sanofi aventis, and exome analysis was made possible by a grant from Molecular Response and in-kind support from Agilent technologies GRAVITAS was sponsored by Accumetrics

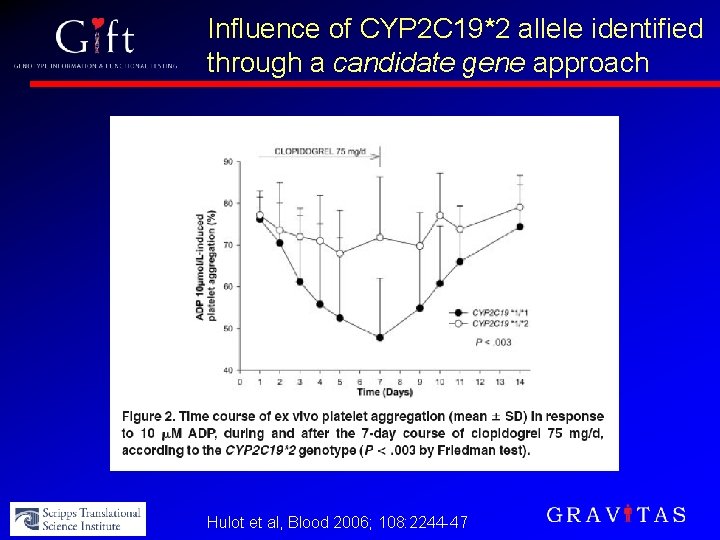
Influence of CYP 2 C 19*2 allele identified through a candidate gene approach Hulot et al, Blood 2006; 108: 2244 -47

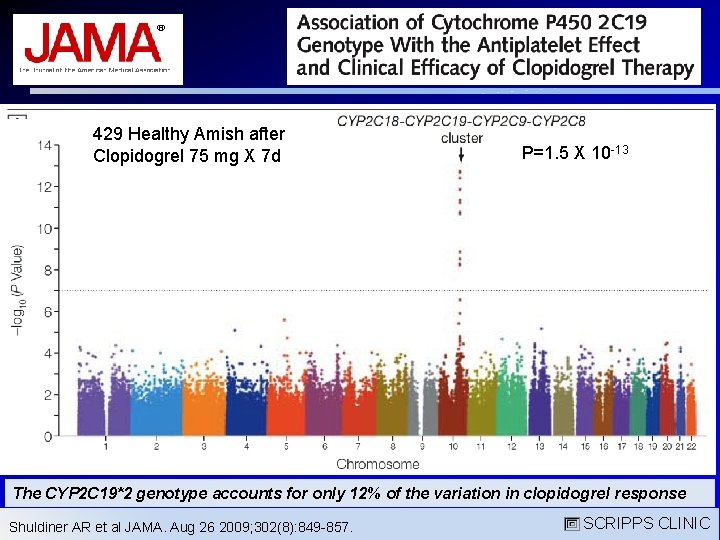
429 Healthy Amish after Clopidogrel 75 mg X 7 d P=1. 5 X 10 -13 The CYP 2 C 19*2 genotype accounts for only 12% of the variation in clopidogrel response Shuldiner AR et al JAMA. Aug 26 2009; 302(8): 849 -857. SCRIPPS CLINIC

What Is Whole Exome Analysis, and Why Do it? • Sequencing of the entire protein coding regions of the human genome • Exploratory approach, non hypothesis-driven (don’t need to know the mechanism of effect). • Identifies both SNPs and insertion/deletions (indels) • Unlike GWAS, can identify actual, causative variant associated with disease, rather than a SNP in linkage dysequilibrium (i. e. , in keeping with bad company) • More likely to detect mutations with a greater impact on disease • Enriched portion of the genome that can be used to search for variants with large effect sizes

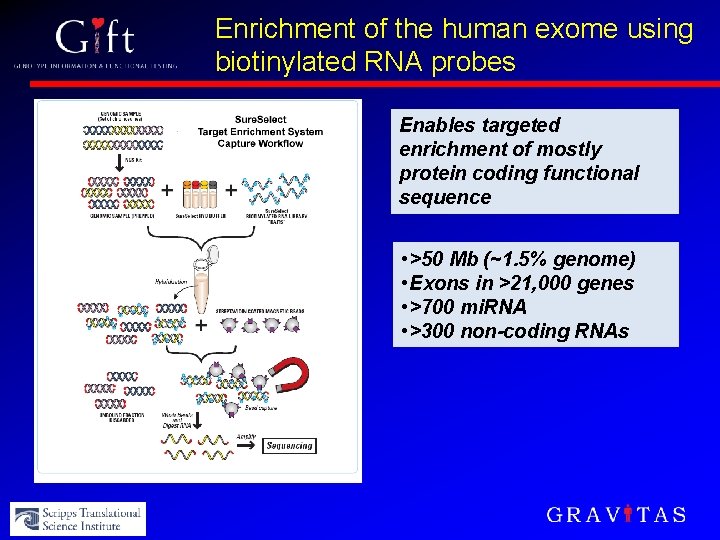
Enrichment of the human exome using biotinylated RNA probes Enables targeted enrichment of mostly protein coding functional sequence • >50 Mb (~1. 5% genome) • Exons in >21, 000 genes • >700 mi. RNA • >300 non-coding RNAs

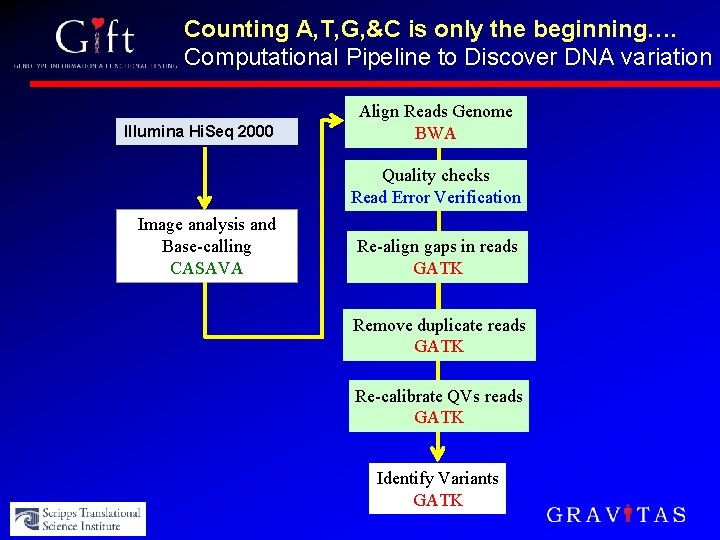
Counting A, T, G, &C is only the beginning…. Computational Pipeline to Discover DNA variation Illumina Hi. Seq 2000 Align Reads Genome BWA Quality checks Read Error Verification Image analysis and Base-calling CASAVA Re-align gaps in reads GATK Remove duplicate reads GATK Re-calibrate QVs reads GATK Identify Variants GATK

Counting A, T, G, &C is only the beginning…. Computational Pipeline to Discover DNA variation 32 -600 processor units employed for 7 -10 days at a time to compute DNA variants on 192 exome samples. Total serial compute time: >400 days

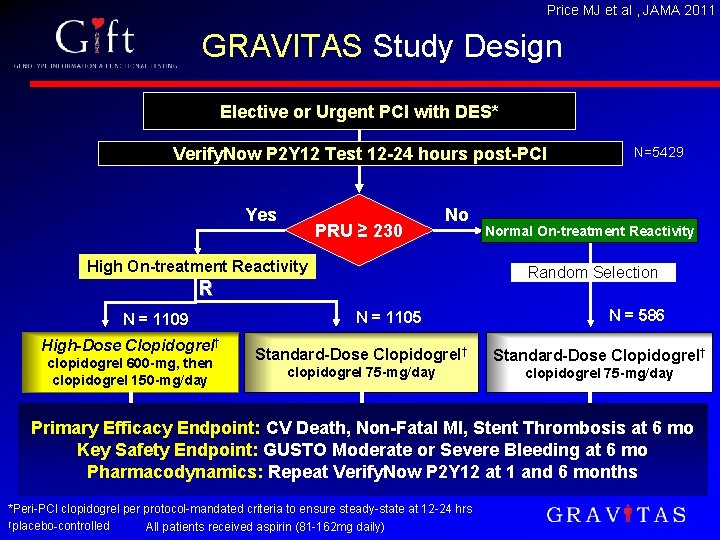
Price MJ et al , JAMA 2011 GRAVITAS Study Design Elective or Urgent PCI with DES* Verify. Now P 2 Y 12 Test 12 -24 hours post-PCI Yes PRU ≥ 230 No High On-treatment Reactivity High-Dose Clopidogrel† clopidogrel 600 -mg, then clopidogrel 150 -mg/day Normal On-treatment Reactivity Random Selection R N = 1109 N=5429 N = 1105 N = 586 Standard-Dose Clopidogrel† clopidogrel 75 -mg/day Primary Efficacy Endpoint: CV Death, Non-Fatal MI, Stent Thrombosis at 6 mo Key Safety Endpoint: GUSTO Moderate or Severe Bleeding at 6 mo Pharmacodynamics: Repeat Verify. Now P 2 Y 12 at 1 and 6 months *Peri-PCI clopidogrel per protocol-mandated criteria to ensure steady-state at 12 -24 hrs †placebo-controlled All patients received aspirin (81 -162 mg daily)

Sample Acquisition • Samples (N=1, 152) obtained at platelet function screening or during follow-up from patients participating in GRAVITAS at 42 participating sites • OTR assessed using Verify. Now P 2 Y 12 test per GRAVITAS protocol • Baseline: 12 -24 hours post-PCI, after standard periprocedural clopidogrel regimen • 30± 7 days • N=192 self-identified Caucasians selected for exome analysis

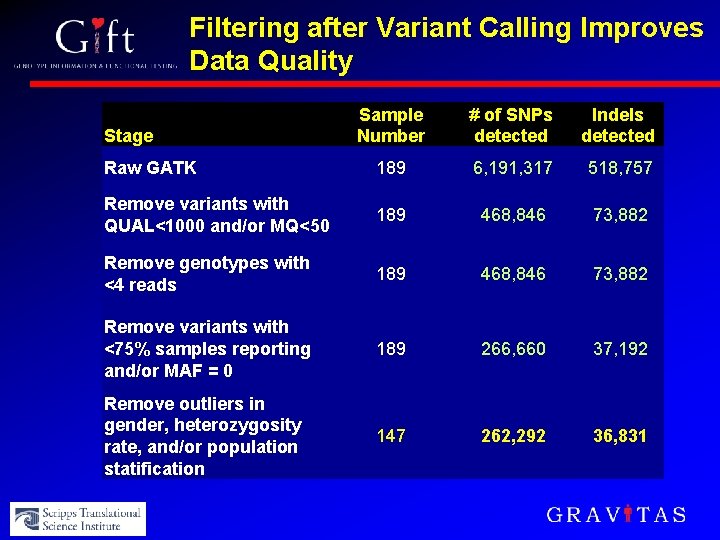
Filtering after Variant Calling Improves Data Quality Sample Number # of SNPs detected Indels detected Raw GATK 189 6, 191, 317 518, 757 Remove variants with QUAL<1000 and/or MQ<50 189 468, 846 73, 882 Remove genotypes with <4 reads 189 468, 846 73, 882 Remove variants with <75% samples reporting and/or MAF = 0 189 266, 660 37, 192 Remove outliers in gender, heterozygosity rate, and/or population statification 147 262, 292 36, 831 Stage

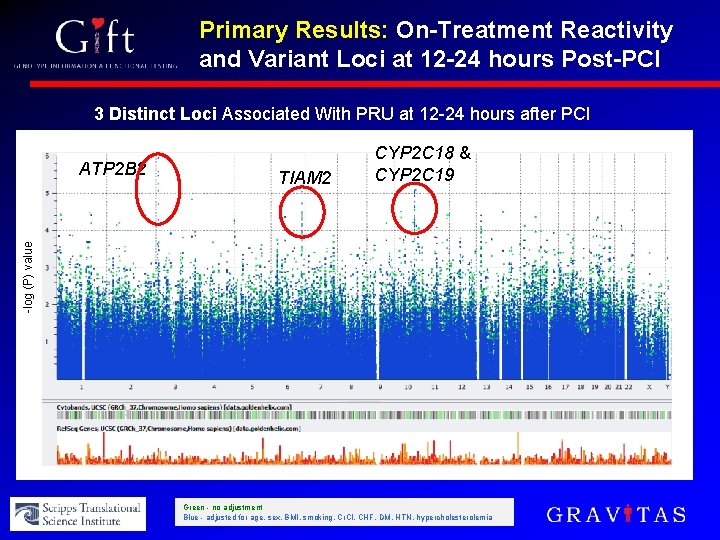
Primary Results: On-Treatment Reactivity and Variant Loci at 12 -24 hours Post-PCI 3 Distinct Loci Associated With PRU at 12 -24 hours after PCI TIAM 2 -log (P) value ATP 2 B 2 CYP 2 C 18 & CYP 2 C 19 Green - no adjustment Blue - adjusted for age, sex, BMI, smoking, Cr. Cl, CHF, DM, HTN, hypercholesterolemia

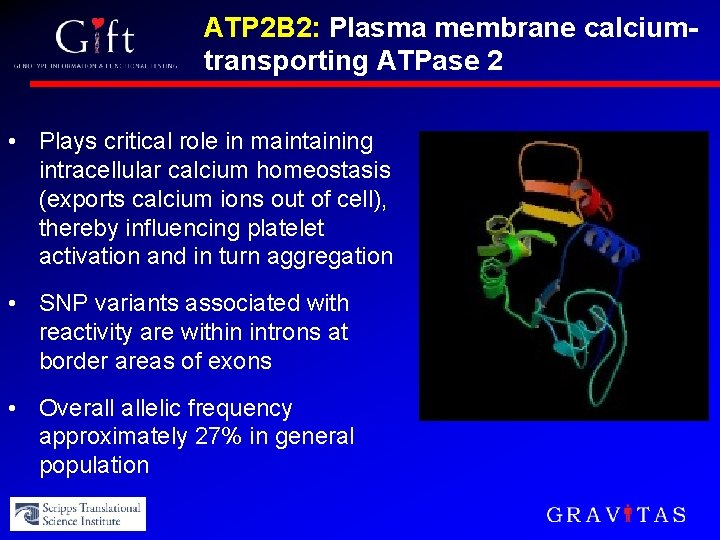
ATP 2 B 2: Plasma membrane calciumtransporting ATPase 2 • Plays critical role in maintaining intracellular calcium homeostasis (exports calcium ions out of cell), thereby influencing platelet activation and in turn aggregation • SNP variants associated with reactivity are within introns at border areas of exons • Overall allelic frequency approximately 27% in general population

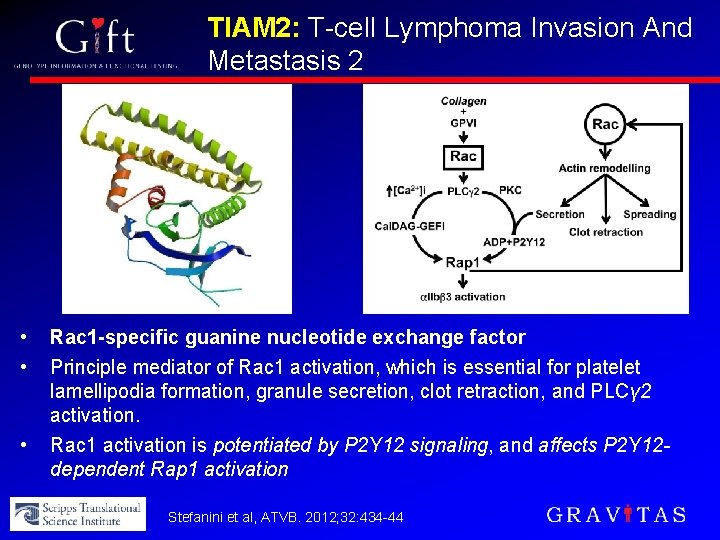
TIAM 2: T-cell Lymphoma Invasion And Metastasis 2 • • • Rac 1 -specific guanine nucleotide exchange factor Principle mediator of Rac 1 activation, which is essential for platelet lamellipodia formation, granule secretion, clot retraction, and PLCγ 2 activation. Rac 1 activation is potentiated by P 2 Y 12 signaling, and affects P 2 Y 12 dependent Rap 1 activation Stefanini et al, ATVB. 2012; 32: 434 -44

TIAM 2 Variants: Clinical Effects • >10 SNPs in TIAM 2 weakly associated with PRU • Most significant SNP: • Arg to Cys (CGC to TGC): non-synonymous, “damaging” substitution according to computational analysis (SIFT) • Associated with lower levels of on-treatment reactivity • Phenotype is consistent with predicted TIAM 2 loss-of-function variant (i. e. , decreased Rac 1 activation) • Allelic frequency approx 13% in general pop’n

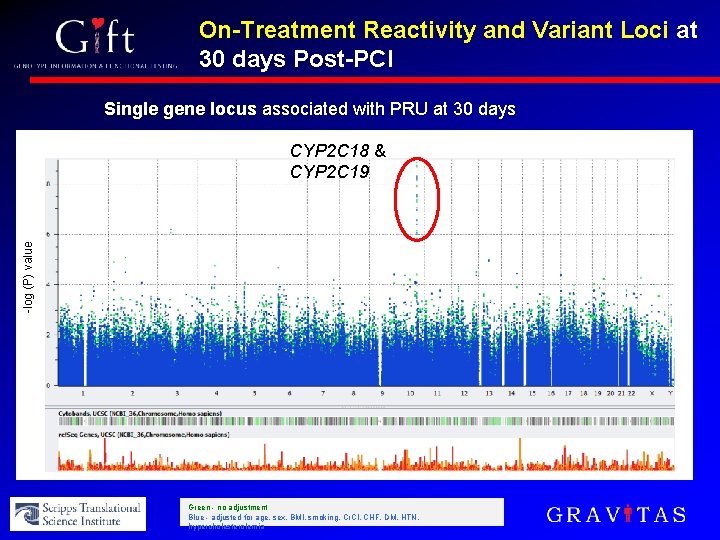
On-Treatment Reactivity and Variant Loci at 30 days Post-PCI Single gene locus associated with PRU at 30 days -log (P) value CYP 2 C 18 & CYP 2 C 19 Green - no adjustment Blue - adjusted for age, sex, BMI, smoking, Cr. Cl, CHF, DM, HTN, hypercholesterolemia

Summary • Exome analysis identified 2 novel loci that appear to be associated with early on-treatment reactivity. • Findings preliminary, but identification of 2 genes critical to platelet function among the 21, 000 sequenced genes lends credibility to the validity of the result • Singular influence of CYP 2 C 18/9 locus at 30 days of maintenance clopidogrel after PCI • Unlikely that other protein-coding variant has large effect on response variability at this timepoint

Next Steps • Validating variants in >1, 000 subjects via genotyping • Increase exome sequencing sample size • Functional modeling

Conclusion • Novel variants in genes downstream of clopidogrel metabolism appear to influence early on-treatment reactivity • Over longer-term follow-up, CYP 2 C 18/19 locus is the primary protein-coding determinant of clopidogrel response variability • Our findings demonstrate the feasibility and potential of exploratory pharmacogenomics using exome sequencing to identify unanticipated mechanisms of drug response

The STSI Team Andrew Carson Ph. D: Computational biology Samuel Levy Ph. D, Director, Genomic Sciences, STSI Guangfa Zhang – Image analysis and base calling Janel Lee, Tiereny Phillips – Exome enrichment Erin Lee Sarah Shaw Murray Rebecca Tisch Eric Topol