Finalizing the Libby Action Plan Research Program Maureen

Finalizing the Libby Action Plan Research Program Maureen R. Gwinn 1, David Berry 2, Andrea Kirk 3, Thomas Bateson 1, Bob Benson 2, Stephen Gavett 1, Annie Jarabek 1, Leonid Kopylev 1, David Bussard 1, Ronald Hines 1, Deborah Mc. Kean 2, Michael Scozzafava 3 1 Office of Research and Development, U. S. Environmental Protection Agency 2 Region 8, U. S. Environmental Protection Agency 3 Office of Land Emergency Management, U. S. Environmental Protection Agency The views expressed in this presentation are that of the presenters and do not necessarily represent the views and/or policies of the U. S. Environmental Protection Agency.

Outline • History of Libby Action Plan (LAP) • Goal and Results of LAP studies – Human health assessments – Analytical methods studies – Laboratory in vitro studies – Laboratory in vivo (animal) toxicity studies – Dosimetry model development – Human epidemiology studies 2

History of Libby Action Plan • Jan 2007 – Cross Agency Meeting to Initiate Development of Libby Action Plan (LAP) • March-June 2007 – Development of work plans with external Peer Review • • July 2007 – Funding Approval for LAP July 2007 – December 2016 – Organize cross-Agency LAP team (OLEM, ORD, Region 8) – Design, perform and complete LAP studies 3
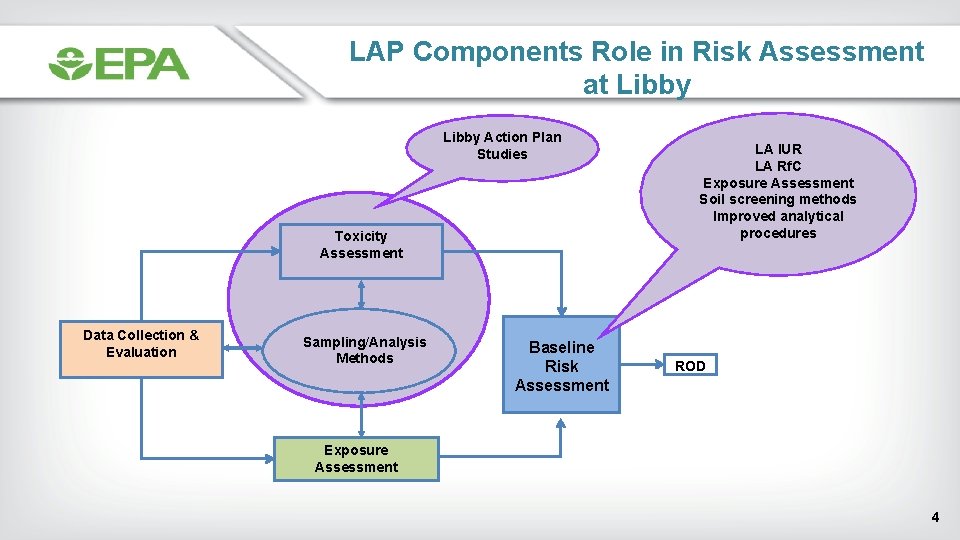
LAP Components Role in Risk Assessment at Libby Action Plan Studies Toxicity Assessment Data Collection & Evaluation Sampling/Analysis Methods Baseline Risk Assessment LA IUR LA Rf. C Exposure Assessment Soil screening methods Improved analytical procedures ROD Exposure Assessment 4
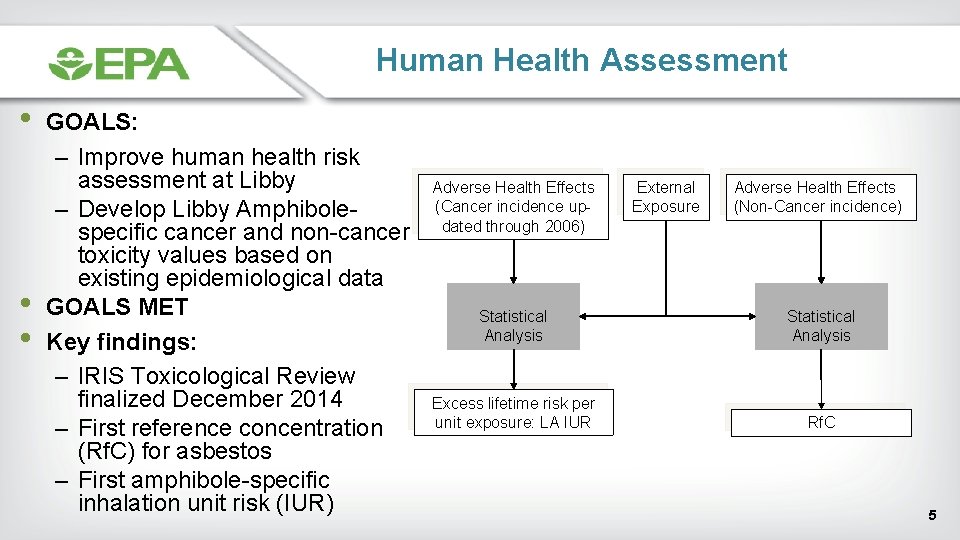
Human Health Assessment • • • GOALS: – Improve human health risk assessment at Libby – Develop Libby Amphibolespecific cancer and non-cancer toxicity values based on existing epidemiological data GOALS MET Key findings: – IRIS Toxicological Review finalized December 2014 – First reference concentration (Rf. C) for asbestos – First amphibole-specific inhalation unit risk (IUR) Adverse Health Effects (Cancer incidence updated through 2006) External Exposure Adverse Health Effects (Non-Cancer incidence) Statistical Analysis Excess lifetime risk per unit exposure: LA IUR Rf. C 5

Analytical Methods • GOALS: – – Support exposure assessment for risk characterization Develop better soil analysis methods for screening Develop better analytical procedures for fiber identification and quantification Provide high quality exposure data for the laboratory toxicity studies • GOALS MET • Key Findings: – Fluidized bed asbestos segregator (FBAS) preparation method demonstrated that low-level LA soil concentrations in the range of 0. 005% can be measured – Preparation of large quantities of test material [LAA 2007] and fiber size characterization – Improved Activity-based sampling (ABS) exposure assessment 6

Laboratory In Vitro Studies • • • GOALS: – Compare biological effects of LA to other types of asbestos; relative potency – Compare dissolution characteristics of LA to other types of asbestos – Needed for dosimetry model development GOALS MET Key Findings: – Demonstrated that particle length and surface area contribute significantly to asbestos toxicity in airway epithelial cells – Showed that LA may be as potent as purified amosite samples in inducing inflammatory responses in vitro and cleavage fragments elicited a strong inflammatory response – Indicated greater potential for biopersistence of LA and maintenance of retained dose and size dimensions in vivo 7

Laboratory In Vivo Studies • GOALS: – Compare biological effects of LA to other types of asbestos – Evaluate non-cancer health effects in rats (including autoimmune and cardiovascular effects) – Inform uncertainty analysis of non-cancer LA risk assessment • GOALS MET • Key Findings: – Multiple exposures are likely more dangerous than single exposures – Physical properties of asbestos samples such as length, aspect ratio, surface area, density, and fiber number help explain why some forms of asbestos are more toxic than others – Inhalation of LA produced persistent inflammatory and fibrotic responses in the lung, and a positive dose trend for incidence of bronchiolar/alveolar adenoma or carcinoma – Inflammatory response leading to pulmonary injury from LA may increase risk of cardiovascular disease 8
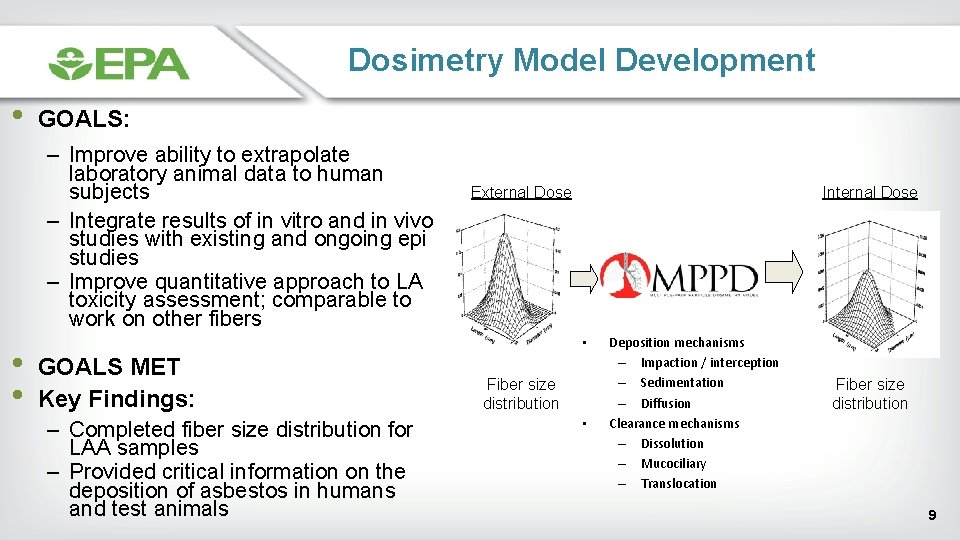
Dosimetry Model Development • GOALS: – Improve ability to extrapolate laboratory animal data to human subjects – Integrate results of in vitro and in vivo studies with existing and ongoing epi studies – Improve quantitative approach to LA toxicity assessment; comparable to work on other fibers • • External Dose Internal Dose • GOALS MET Key Findings: – Completed fiber size distribution for LAA samples – Provided critical information on the deposition of asbestos in humans and test animals Fiber size distribution • Deposition mechanisms – Impaction / interception – Sedimentation – Diffusion Clearance mechanisms – Dissolution – Mucociliary – Translocation Fiber size distribution 9

Human Epidemiology Studies • • • GOALS – explore the health effects in individuals exposed to LA, specifically: • health effects from childhood exposure • health effects from environmental/community exposures • non-pulmonary health effects • susceptible populations GOALS MET Key Findings: – Provided first evidence of impact of exposure to LA to measures of respiratory function – Showed that even relatively low cumulative lifetime exposures can increase risk of pleural diseases (LPT, SIO and DPT) – Illustrated a broad spectrum of ARD diseases heretofore not ascribed to asbestos exposure(s) – Supported use of digital radiographs as replacements for film – Helped clarify risks to children incurred during normal childhood activities 10

Ongoing Studies • The Libby Action Plan is complete, but publications continue! – Mode of Action • Finalize publication – Dosimetry model • Complete analysis and publish results – Epidemiology Studies • Complete collection, analysis, and publication of data 11

Thank you for your attention. Publication list available upon request. 12

Supplemental Slides 13

Studies in the LAP Project Objective Completion Date Preparation of Libby Testing Material To provide consistent samples of LAA for LAP laboratory studies. May 2009 Characterization of Fiber Size Distribution in Libby Vermiculite Analyze asbestos fibers in vermiculite ore samples representative of material used in the O. M. Scott Company plant to support development of the LAA Reference Concentration (Rf. C). August 2008 Dosimetry Model Development and Simulation Studies To develop a model of deposition and clearance of inhaled LAA in rats March 2015 and humans to relate external dose to internal dose in the respiratory tract. In Vitro Dissolution Assays To determine dissolution rates for LAA fibers as an indication of durability relative to other types of mineral fibers. March 2015 In Vitro Toxicity Endpoints To screen the biologic activity of asbestos samples and investigate mechanisms by which fibers may damage lung cells. September 2015 Comparative Toxicology in Rats To compare acute and chronic toxic effects of LAA with those of other naturally occurring asbestos fibers using intratracheal exposures. September 2015 Inhalation Toxicology in Rats To determine the inhalation toxicity of LAA and provide data for the dosimetry modeling project. To evaluate the potential for LAA to exacerbate the development and progression of rheumatoid arthritis. January 2016 To evaluate the potential for LAA to induce cardiovascular effects. September 2015 Assessment of Autoimmune Disease in Rodents Exposed to LAA Assessment of Cardiovascular Effects in Rodents Exposed to LAA December 2012 14

Studies in the LAP Region 8: Analytical Methods Studies for Environmental Monitoring Project Filter Pore Size Comparison Studies Low-Level Soil Method Development Objective Completion Date To determine whethere is a difference in collection September 2011 efficiency between 0. 8 -µm (pore size) filters and 0. 45µm filters. To develop and validate a reliable method for analysis September 2015 of low levels of LAA in soil. and ongoing Comparison of Direct & Indirect Preparations To determine whether the indirect preparation method March 2014 [ISO 13794] provides equivalent accuracy and sensitivity to the direct preparation method [ISO 10312] for quantifying Libby Amphibole fibers in air samples. Ambient Air Collection Method Verification Verify method in support of continued monitoring and documentation of LAA fiber concentrations in ambient air in Libby, MT. February 2009 15

Studies in the LAP ATSDR: Epidemiologic Research in Support of the Toxicity Assessment Completion Date Project Objective New Epidemiologic Information from Libby MT Cohorts Intramural research: Further study of health effects seen in the Libby Miner cohort and the ATSDR community screening January 2012 Libby Epidemiology Research Program Further explore the health effects in individuals exposed to Libby amphibole asbestos specifically: health effects from childhood exposure, health effects from environmental/community exposures, non-pulmonary health effects and susceptible populations. Many studies complete. Monitoring of exposed individuals continues and papers will continue to be published. New Epidemiologic Information from Other Cohorts To determine whether low-level exposure to Libby Amphibole January 2015 and is associated with lung abnormalities, loss of lung function, ongoing and increased morbidity/mortality due to pulmonary disease. 16
- Slides: 16