Final Quantum Light Model Lab Electron Potpourri Config
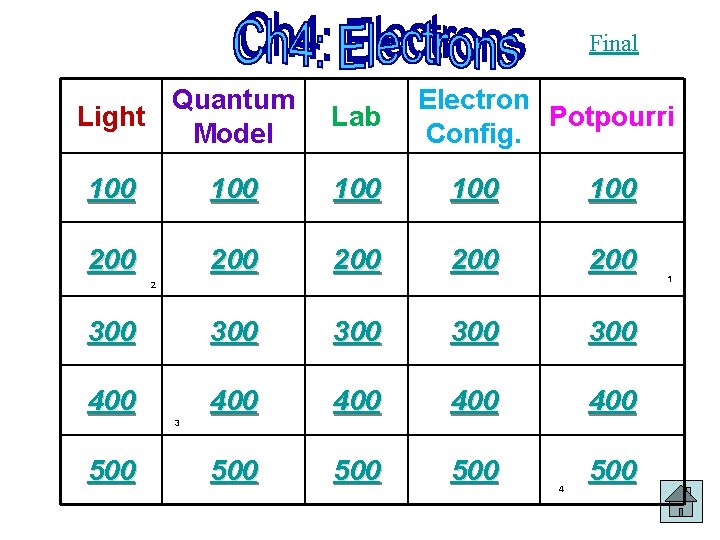
Final Quantum Light Model Lab Electron Potpourri Config. 100 100 100 200 200 200 300 300 300 400 400 400 500 500 2 3 500 4 1

100 Light Name three wave properties of light. Light behaves as a wave when it _______. Answer

Light 100 Refraction, diffraction, interference, reflection Light behaves as a wave when it propagates (travels).

200 Light When an electron in an atom falls from the 5 th to the 2 nd energy level, 4. 58 x 10 19 J of energy are released. What is the frequency of this energy? Answer All Play!

Light 200 E = h 4. 58 x 10 19 J = 6. 626 x 10 34 Jsec X Answer = 6. 91 x 1014 Hz

300 Light A bright line in the spectrum of sodium has a wavelength of 5. 90 x 102 nm. What is the frequency? Answer All Play!!

Light 300 ? m = 5. 90 x 102 nm x 1 m_ = 5. 90 x 10 -7 m 1 x 109 nm Use c = 3. 00 x 108 m/sec = 5. 90 x 10 -7 m X Answer X= 5. 08 x 1014 Hz

400 Light Explain the photoelectric effect. Is this a wave or particle property? Give an example of how this is useful. Answer

Light 400 Light of certain frequencies striking a metal causes the metal to release electrons. (electric current is created) This is a particle property. Useful: solar panels, garage door opener safety

500 Light A device emits light with frequency of 9. 37 x 107 Hz. Find the wavelength. Is the device a tanning bed, red laser or a radio tower? All Play! Answer

Light 500 Use c = 3. 00 x 108 m/s = 9. 37 x 107 Hz l = 3. 20 m ? nm = 3. 20 m X 1 x 109 nm = 3. 20 x 109 nm 1 meter 3. 20 x 109 nm is longer wavelength than visible light, thus it is Radio

100 Quantum Model Name the scientist who stated that electrons occupy orbitals of equal energy singly, with parallel spins, before doubling up. Answer All Play

Quantum Model 100 Hund

200 Quantum Model A 3 -dimensional region about the nucleus where an electron may be found is a(n) All Play!!!! Answer

Quantum Model 200 orbital

300 Quantum Model Name the 1 st quantum number, describe what it indicates and what its possible values are. Answer

Final Jeopardy A photon of yellow light has 3. 58 x 10 -19 Joules of energy. What is the wavelength of this light in nanometers? Answer

Final Jeopardy Answer E = hc/ = hc = 6. 626 x 10 -34 Jsec x 3. 00 x 108 m/s E 3. 58 x 10 -19 Joules = 5. 55 x 10 -7 m = 555 nm

Quantum Model 300 Principle Quantum Number Indicates energy level of electron – distance from nucleus Values are positive integers 1, 2, 3, etc.
![400 Quantum Model The noble gas configuration of an element is: [Kr]5 s 24 400 Quantum Model The noble gas configuration of an element is: [Kr]5 s 24](http://slidetodoc.com/presentation_image_h/11b81bb55eb75fc27cdbc54537bf91e1/image-20.jpg)
400 Quantum Model The noble gas configuration of an element is: [Kr]5 s 24 d 105 p 3. Identify the element and state how many unpaired electrons it has. All Play!!
![Quantum Model 400 1. [Kr]5 s 24 d 105 p 3 -- this element Quantum Model 400 1. [Kr]5 s 24 d 105 p 3 -- this element](http://slidetodoc.com/presentation_image_h/11b81bb55eb75fc27cdbc54537bf91e1/image-21.jpg)
Quantum Model 400 1. [Kr]5 s 24 d 105 p 3 -- this element is Sb. 2. It has 3 unpaired electrons.

500 Quantum Model Give the letters naming the 4 types of sublevels (shapes) and indicate the maximum # of electrons each can hold. Answer

Quantum Model 500 s= 2 p= 6 d = 10 f = 14

100 Lab An illuminated tungsten light bulb, a hot toaster coil, and molten lava are all examples of _____. The spectrum of these light sources would be _____. Answer All Play!!

Lab 100 Incandescence continuous spectrum

200 Lab Explain the difference between phosphorescence and fluorescence. Give one example of each. Answer

Lab 200 Both are the absorption of energy(usually UV) with the emission of visible light. In fluorescence the object stops glowing as soon as the energy source is removed. In phosphorescence, the glowing persists for a time after the energy source is removed. An example of phosphorescence is: a glow-in-the -dark toothbrush. An example of fluorescence is: the bright white glow of dirty shoelaces under a UV light.

300 Lab A flame test is performed on a metal compound. The flame is a hot pink/magenta color. What metal must be present? (Give name and symbol) swer

Lab 300 Li Lithium

400 Lab What piece of lab equipment is needed to view a spectrum? What in this room emits a bright line spectrum? Answer

Lab 400 A spectroscope The fluorescent lights or a flame test.

500 Lab What is the main difference between luminescence and incandescence? Give 2 examples each of incandescence and chemiluminescence. Answer

Lab 500 Luminescence is light produced without heat. Incandescence is light from a material heated to glowing. Incandescence – a W filament light bulb, a metal wire held in a flame until it glows red. Chemiluminescence- glowsticks, fireflies, moon jellyfish, the plants on Pandora

100 Electron Configuration Identify each of the following atoms based on their electron configurations. A. [Ne]3 s 2 B. [Ar]3 d 64 s 2 C. [Xe]6 s 24 f 145 d 7 All Play!! Answer

Electron Configuration 100 A. Mg – magnesium B. Fe – iron C. Ir -- iridium

200 Electron Configuration Write the orbital diagram for phosphorus. Answer

Electron Config 200 Answer 1 s 2 s 2 px py pz 3 s 3 px py pz

300 Electron Configuration Write the noble gas configuration for Re, Rhenium Answer
![Electron Configuration 300 Re [Xe]6 s 24 f 145 d 5 Electron Configuration 300 Re [Xe]6 s 24 f 145 d 5](http://slidetodoc.com/presentation_image_h/11b81bb55eb75fc27cdbc54537bf91e1/image-39.jpg)
Electron Configuration 300 Re [Xe]6 s 24 f 145 d 5

400 Electron Configuration Give the electron configuratio and noble gas configuration for strontium. Answer

Electron Configuration 400 2 2 6 2 10 6 2 1 s 2 s 2 p 3 s 3 p 4 s 3 d 4 p 5 s [Kr]5 s 2 Sr:

500 Electron Configuration Write an electron configuration for an excited carbon atom. Answer All Play!!

Electron Configuration 500 Ground state: 1 s 22 p 2 Excited configuration: 1 s 22 p 13 s 1 one of the 2 p electrons is in a higher energy orbital (can be any higher orbital)

100 Potpourri Which of the following configurations is impossible according to the Quantum Model? A. B. C. D. 1 s 22 s 23 p 1 1 s 22 p 62 d 1 [Rn]7 s 26 d 1 [Xe]6 s 25 d 1 All Play! Answer

Potpourri 100 B. Is impossible because there is no d sublevel allowed in the 2 nd energy level.

200 Potpourri Determine the % by mass of nitrogen in TNT. The formula of TNT is C 7 H 5 N 3 O 6 Answer All Play!!
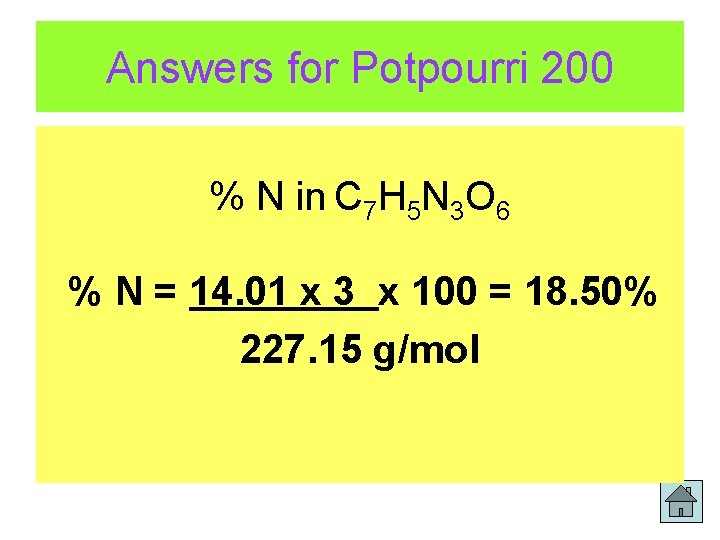
Answers for Potpourri 200 % N in C 7 H 5 N 3 O 6 % N = 14. 01 x 3 x 100 = 18. 50% 227. 15 g/mol

300 Potpourri A line in the spectrum of an element has a wavelength of 7. 5 x 10 -7 m. 1. Can you see this light? 2. What is the energy of the light? Answer
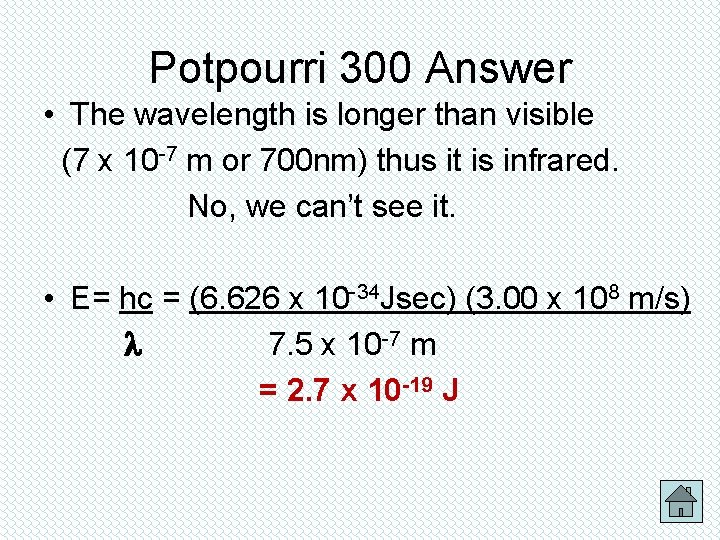
Potpourri 300 Answer • The wavelength is longer than visible (7 x 10 -7 m or 700 nm) thus it is infrared. No, we can’t see it. • E= hc = (6. 626 x 10 -34 Jsec) (3. 00 x 108 m/s) 7. 5 x 10 -7 m = 2. 7 x 10 -19 J

400 Potpourri State and explain the Pauli Exclusion Principle. Answer

Potpourri 400 PEP: No two electrons in the same atom have the same 4 quantum numbers. If two electrons have the same first 3 quantum numbers, they are in the same orbital and thus must have different spins.

500 Potpourri A sample has the composition of 85. 5% C and 14. 5% H. It has a molar mass of 98. What is the molecular formula of this compound? (hint: find empirical formula 1 st!) All Play!! Answer

Potpourri 500 mol C = 85. 5 g x 1 mol = 7. 12 mol C/7. 12 = 1 12. 01 g mol H = 14. 5 g x 1 mol = 14. 4 mol H/7. 12 = 2. 02 1. 01 g Empirical = C 1 H 2 molar mass of empirical = 14. 03 98/14. 03 = 7 Molecular formula = C 7 H 14
- Slides: 53