Final Exam Instructions 1 You must bring a

Final Exam Instructions: 1. You must bring a #2 pencil. YOU MAY NOT USE A CALCULATOR. You MAY NOT use the formula sheet. 2. Cell phones are not allowed. If you are observed with a cell phone in use or in sight, you will receive a grade of ZERO on the final exam. 3. You may not leave the testing room for any reason. If you must leave the room during the exam, you must turn in your exam first, and you will NOT be allowed to continue after it is turned in. 4. You will have exactly 120 minutes to complete 70 multiple choice questions. If you are late, you will have less than 120 minutes to complete the exam. 5. Your grade will be determined as a percentile of the national average or as a raw percentage, which ever score is higher. 6. You will be provided with a test booklet which has abbreviations, a section of the periodic table, and some molecular structures, answer sheet, and scratch paper.
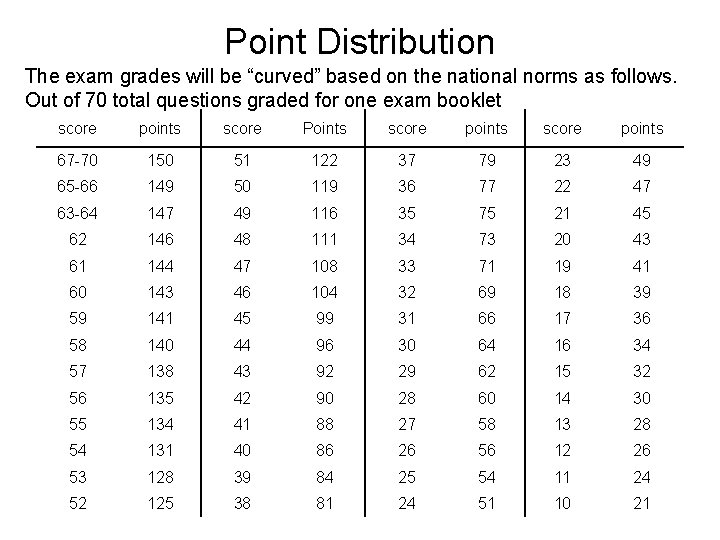
Point Distribution The exam grades will be “curved” based on the national norms as follows. Out of 70 total questions graded for one exam booklet score points score Points score points 67 -70 150 51 122 37 79 23 49 65 -66 149 50 119 36 77 22 47 63 -64 147 49 116 35 75 21 45 62 146 48 111 34 73 20 43 61 144 47 108 33 71 19 41 60 143 46 104 32 69 18 39 59 141 45 99 31 66 17 36 58 140 44 96 30 64 16 34 57 138 43 92 29 62 15 32 56 135 42 90 28 60 14 30 55 134 41 88 27 58 13 28 54 131 40 86 26 56 12 26 53 128 39 84 25 54 11 24 52 125 38 81 24 51 10 21
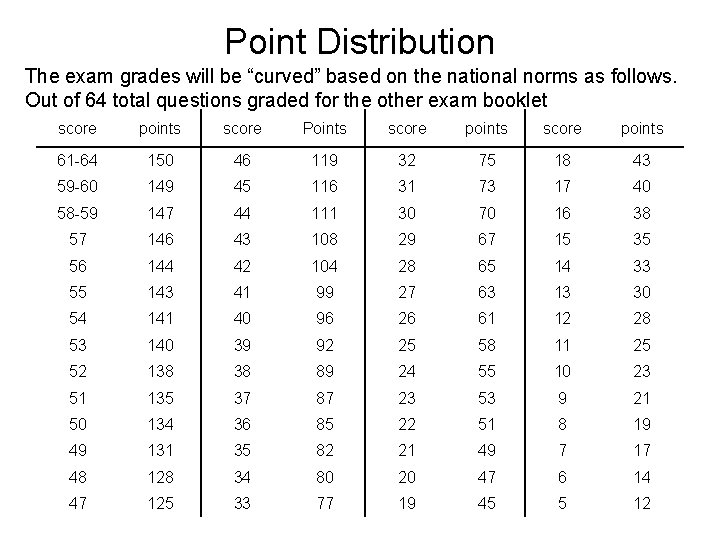
Point Distribution The exam grades will be “curved” based on the national norms as follows. Out of 64 total questions graded for the other exam booklet score points score Points score points 61 -64 150 46 119 32 75 18 43 59 -60 149 45 116 31 73 17 40 58 -59 147 44 111 30 70 16 38 57 146 43 108 29 67 15 35 56 144 42 104 28 65 14 33 55 143 41 99 27 63 13 30 54 141 40 96 26 61 12 28 53 140 39 92 25 58 11 25 52 138 38 89 24 55 10 23 51 135 37 87 23 53 9 21 50 134 36 85 22 51 8 19 49 131 35 82 21 49 7 17 48 128 34 80 20 47 6 14 47 125 33 77 19 45 5 12

Final Exam Topics: Naming: IUPAC and common names we covered, E/Z notation Reactivity: all reaction conditions we discussed are fair game, strengths of organic acids and bases (based on stability of their conjugates), strengths of nulcleophiles and electrophiles, electron withdrawing or donating groups and their effects on the ring, drawing transition states, reaction coordinate diagrams for reaction mechanisms, polymerizations, kinetics and rate law, thermodynamics Drawing molecules, formal charge, resonance, recognizing aromaticity, sp 2 sp 3 hybridization and molecular geometry, Stability: Newman projections, gauche interactions, cyclohexanes (axial and equitorial position), 3˚, 2˚, 1˚, and resonance stabilized carbocations, HOMO and LUMO, degree of substitution on a C=C, Isomers: constitutional, chirality, stereo (diastereomers, enantiomers), stereocenters, R and S configuration, meso compounds, racemic mixtures Spectroscopy: IR, MS, NMR (proton and carbon), optical activity, UV

Organic Chemistry Olympiad 2010 Rules • The class will be split into 2 teams. • Each team will send a team member to the front of the room to compete heads up (no team member may participate in heads up twice until all team members have participated at least once). No coaching will be allowed during heads-up competition. • Heads-up competition will result in one of the following outcomes. 1. Both competitors choose not to answer the question • In this event, the competitors will consult with their team. During team consultation, a free-for-all will ensue, and any team member may submit an answer on the board. The first team to officially submit a correct answer will receive 1 point. 2. Both competitors answer the question correctly • In this event, the competitor who submitted their answer first will receive 2 points for their team, and the competitor will get one point for their team. 3. One competitor answers correctly and the other does not • The competitor who answers correctly will earn 2 points for their team. 4. Both competitors answer the question incorrectly • No points will be awarded. • Competitors may NOT look at each others work until they have both officially submitted their answers.
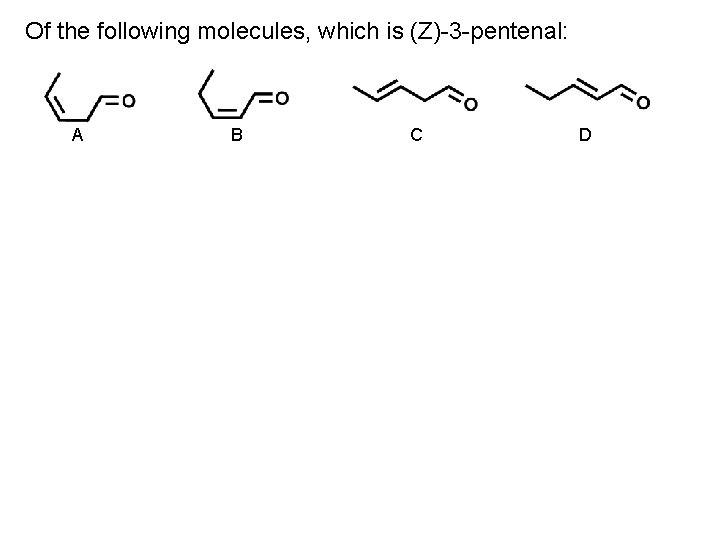
Of the following molecules, which is (Z)-3 -pentenal: A B C D
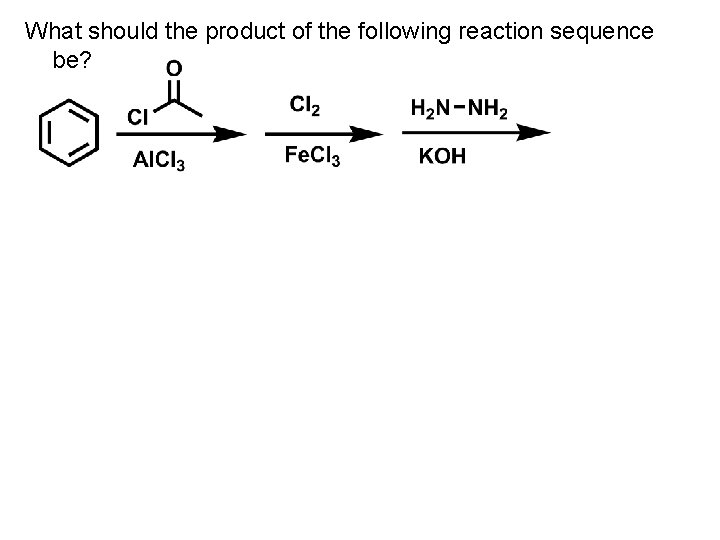
What should the product of the following reaction sequence be?
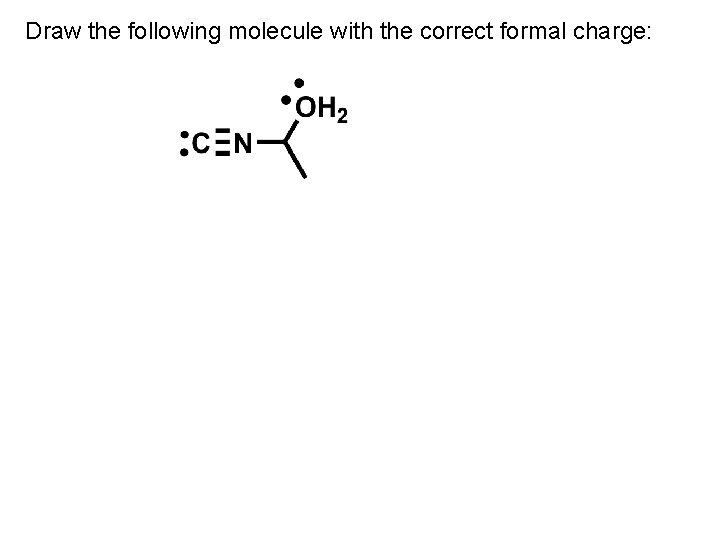
Draw the following molecule with the correct formal charge:
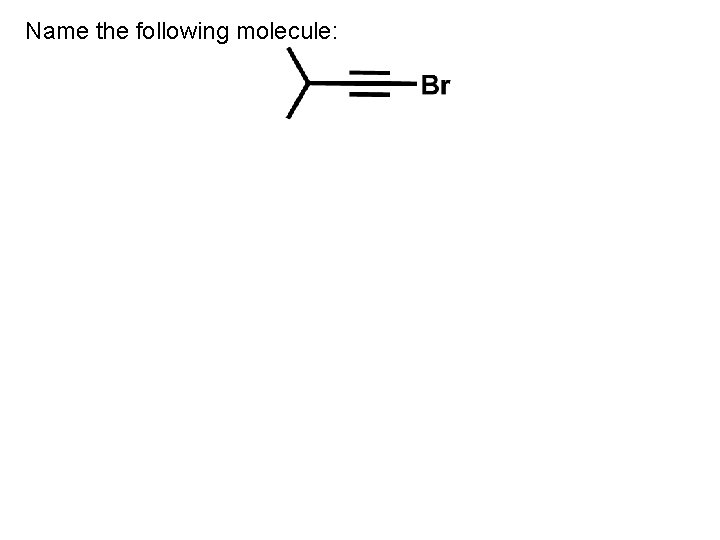
Name the following molecule:
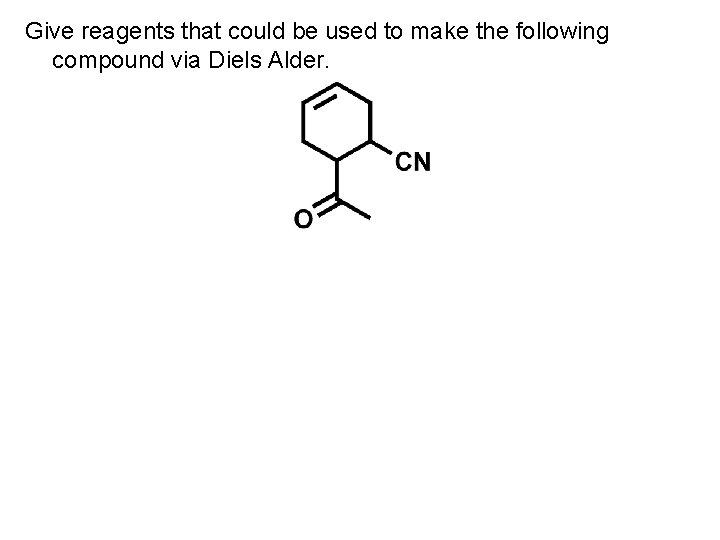
Give reagents that could be used to make the following compound via Diels Alder.
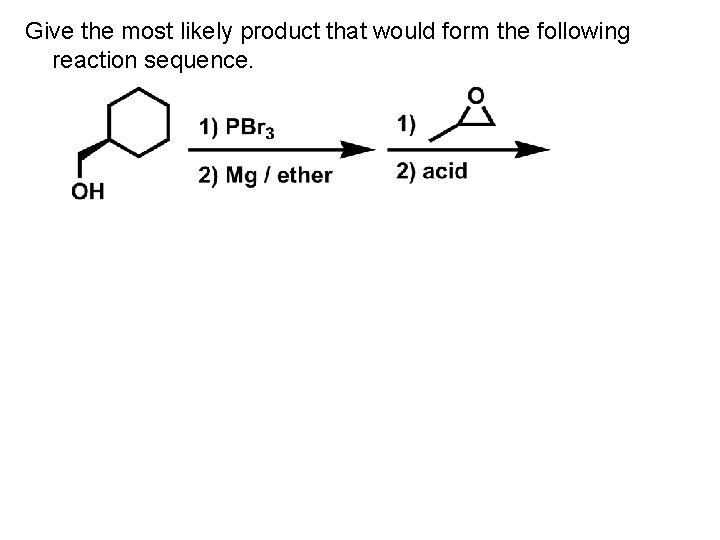
Give the most likely product that would form the following reaction sequence.
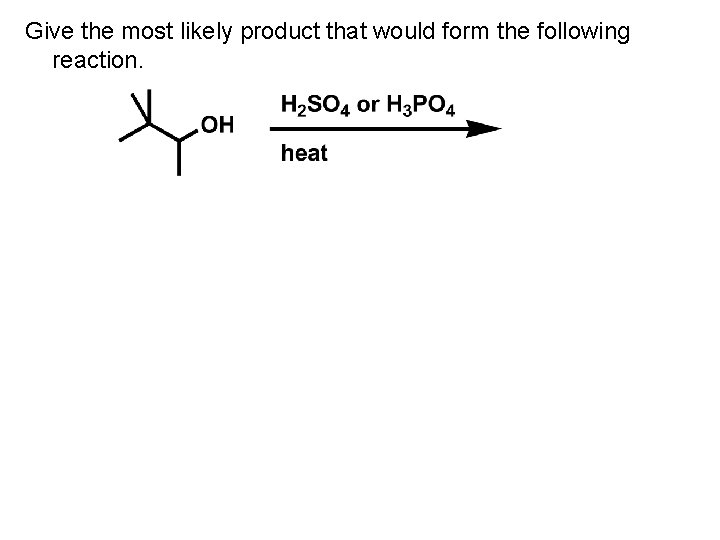
Give the most likely product that would form the following reaction.
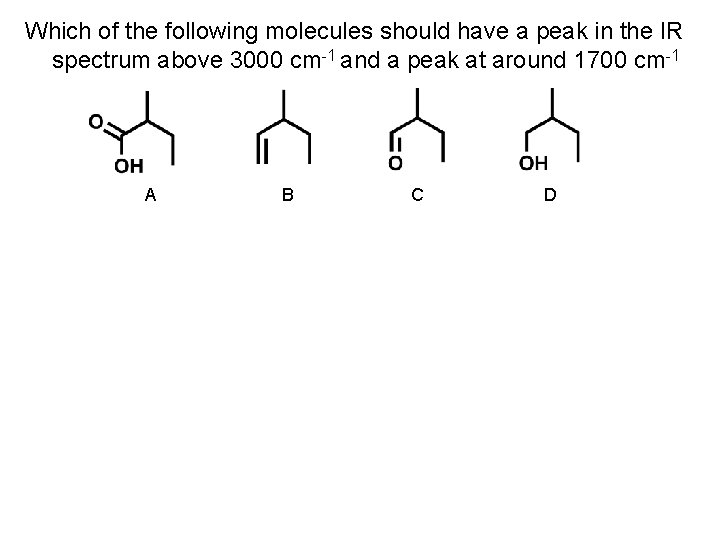
Which of the following molecules should have a peak in the IR spectrum above 3000 cm-1 and a peak at around 1700 cm-1 A B C D

True or False: If benzoic acid were substituted with an electron withdrawing group in the para position, the resulting compound will have a lower p. Ka than benzoic acid.

Give the structure of a ketone that would give 2 13 C NMR peaks but has 3 total carbons.
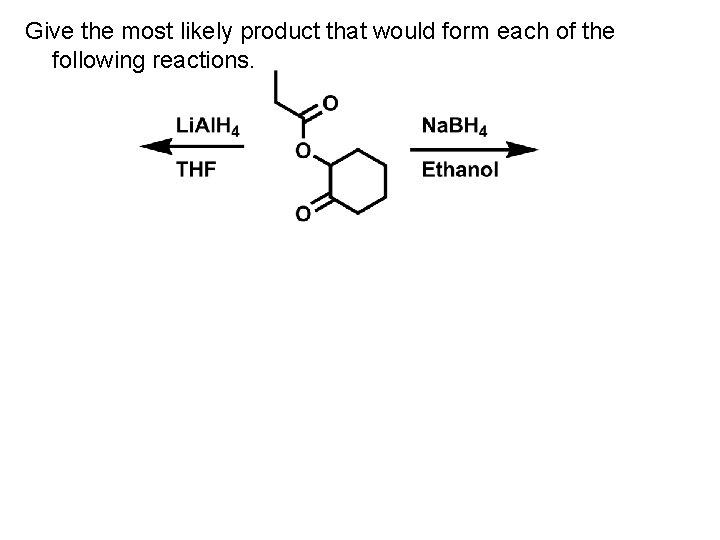
Give the most likely product that would form each of the following reactions.
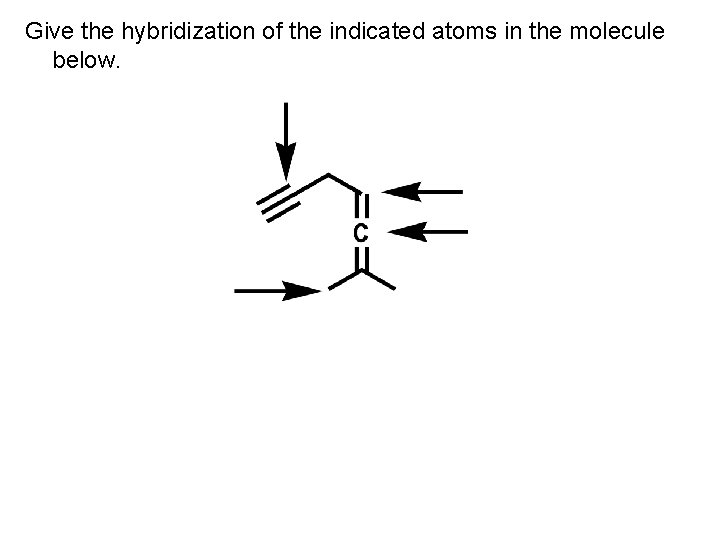
Give the hybridization of the indicated atoms in the molecule below.
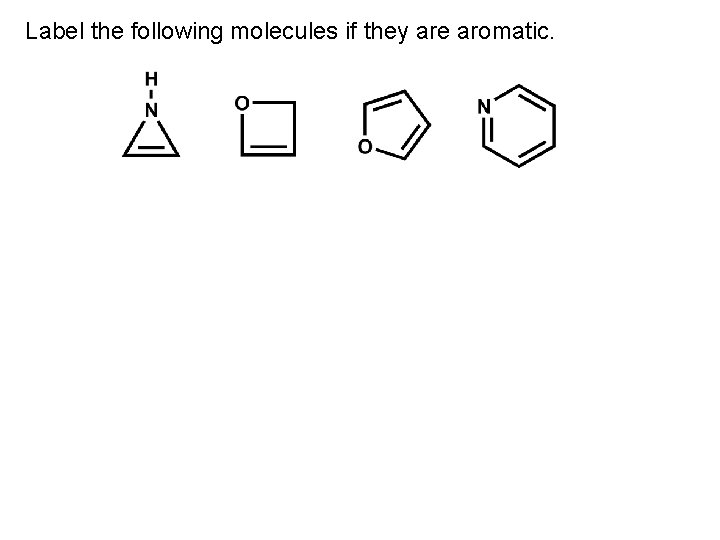
Label the following molecules if they are aromatic.
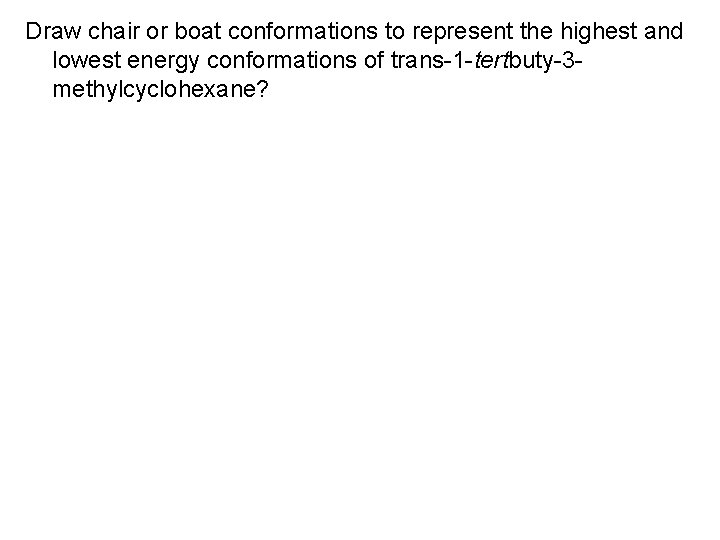
Draw chair or boat conformations to represent the highest and lowest energy conformations of trans-1 -tertbuty-3 methylcyclohexane?
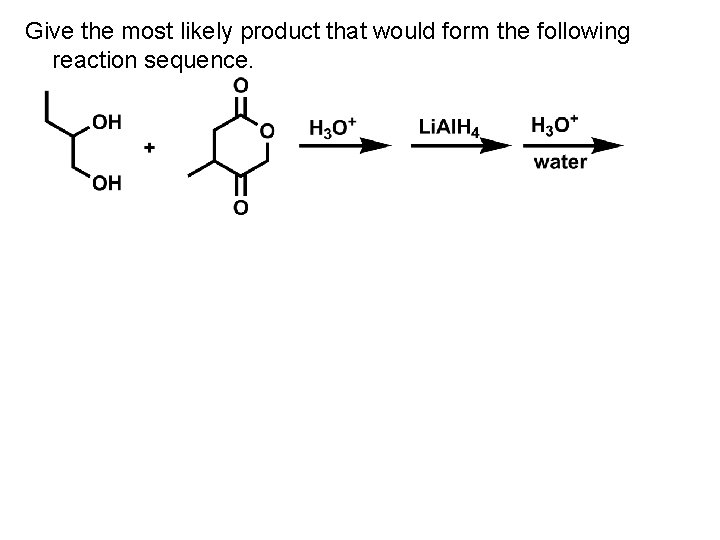
Give the most likely product that would form the following reaction sequence.
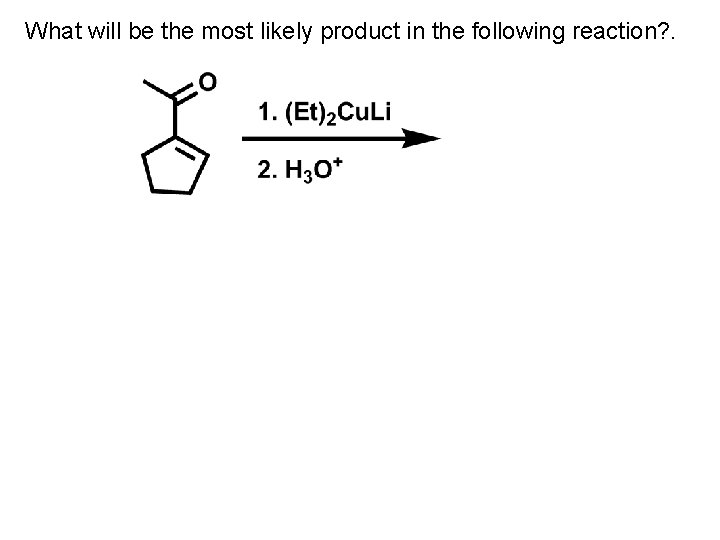
What will be the most likely product in the following reaction? .
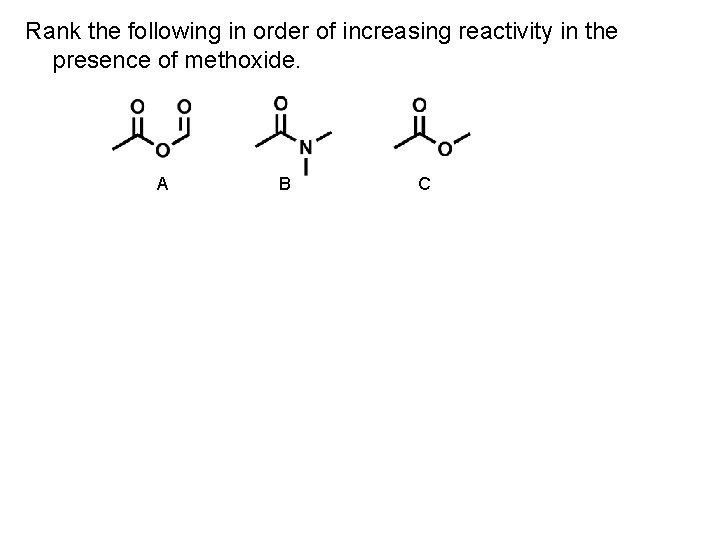
Rank the following in order of increasing reactivity in the presence of methoxide. A B C

Draw Newman projections to represent the highest and lowest energy conformations of pentane.

Give the function group generally created with a primary alcohol is treated with Ts. Cl/pyr followed by Na. OEt.

Which have the smallest HOMO-LUMO gap? Which will absorb the shortest wavelenth UV light? Alkene, diene, conjugated triene, alkane

Rank the following in order of increasing p. Ka Acetylene, methyl amine, methanol

Rank the following in order of increasing basicity Aniline, p-methoxyaniline, p-nitroaniline, m-chloroaniline
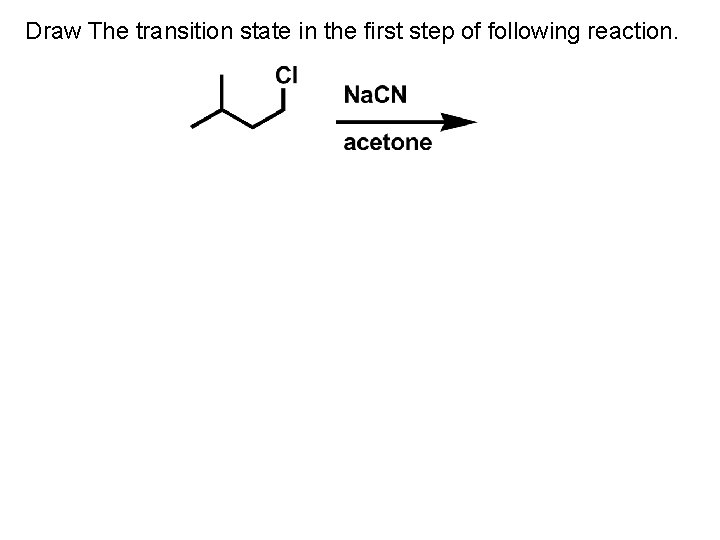
Draw The transition state in the first step of following reaction.
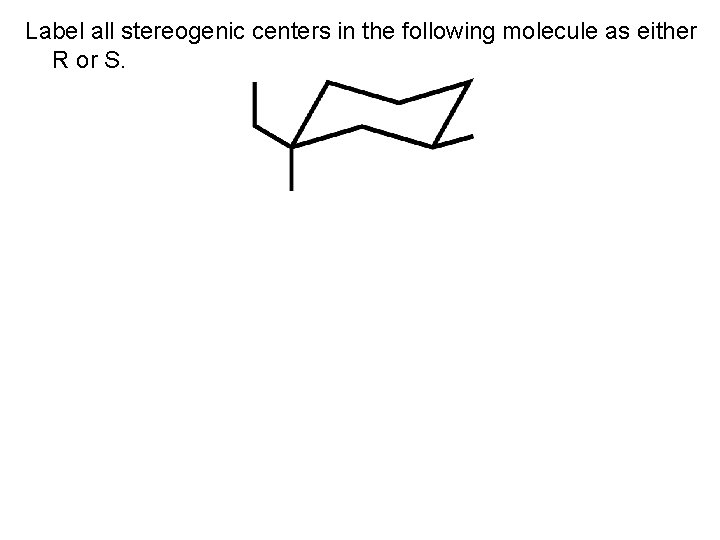
Label all stereogenic centers in the following molecule as either R or S.

Identify the key difference in the IR of aldehydes, ketones, carboxylic acids, alcohols, and amines.
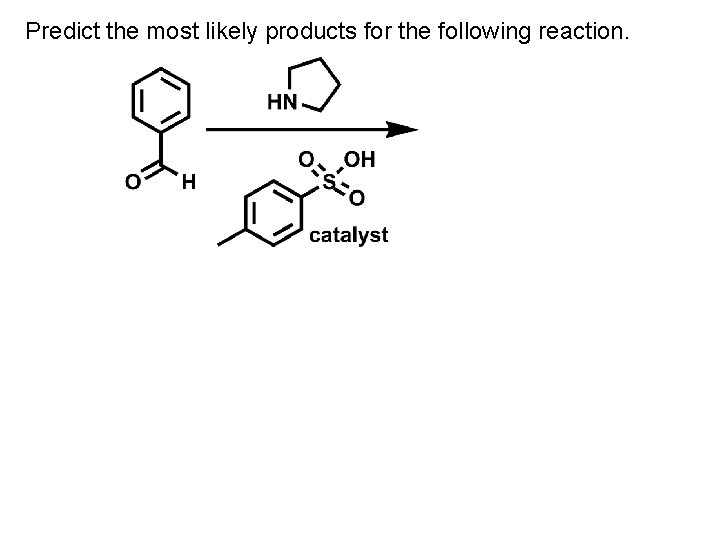
Predict the most likely products for the following reaction.
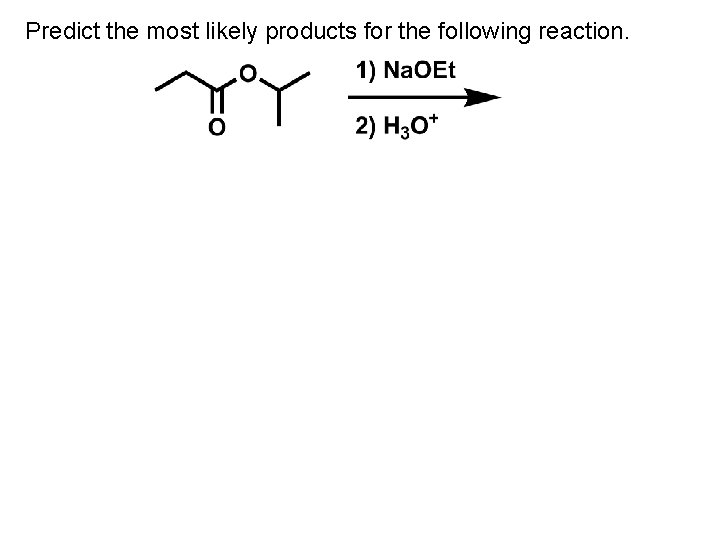
Predict the most likely products for the following reaction.

Draw a molecule that could give the following 13 C decoupled NMR data? Data: Peak 1 2 3 4– 7 shift (ppm) 20 27 30 100 -150
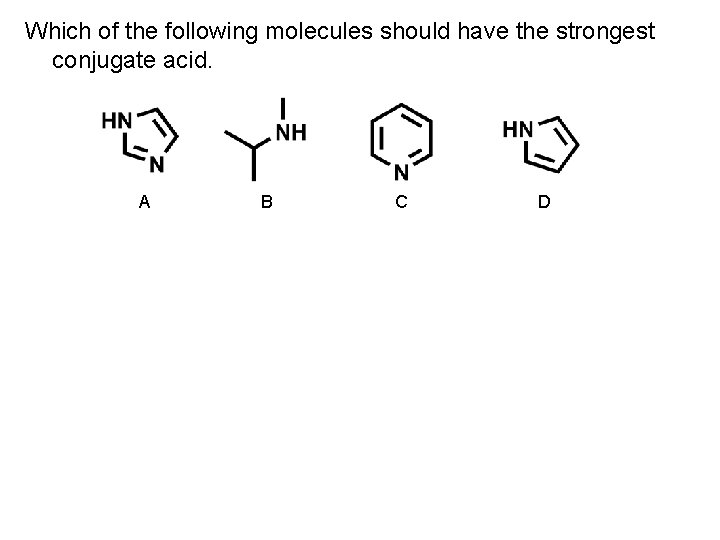
Which of the following molecules should have the strongest conjugate acid. A B C D
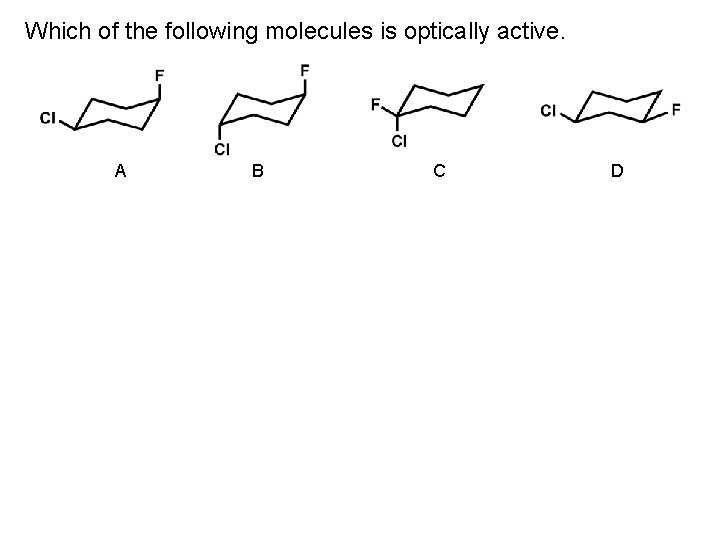
Which of the following molecules is optically active. A B C D

Draw a molecule that could be synthesized starting from an ester and two moles of a grignard reagent.

Draw the first step in the mechanism by which an ester undergoes soponification (hydrolysis) in the presence of Na. OH.
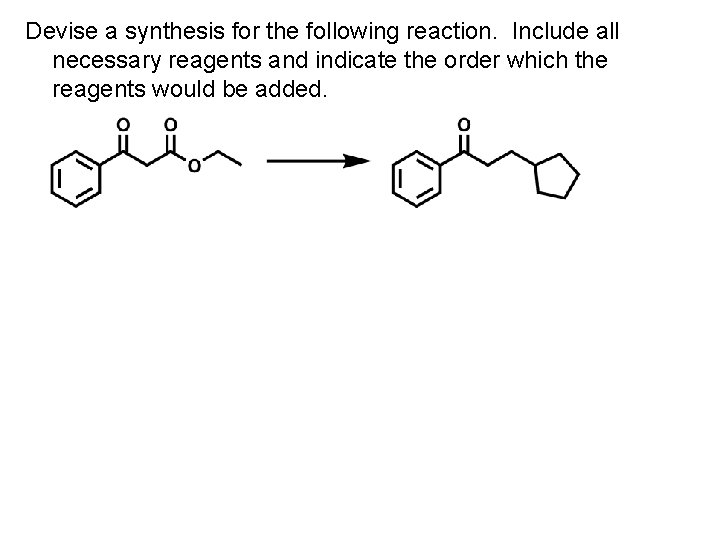
Devise a synthesis for the following reaction. Include all necessary reagents and indicate the order which the reagents would be added.
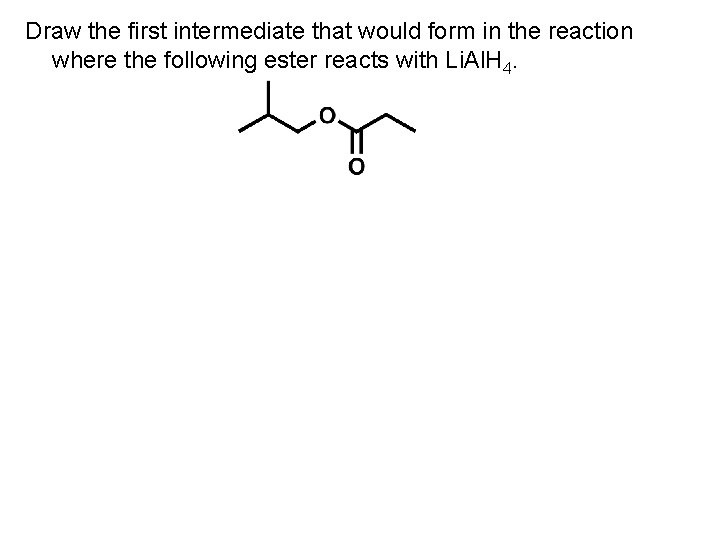
Draw the first intermediate that would form in the reaction where the following ester reacts with Li. Al. H 4.
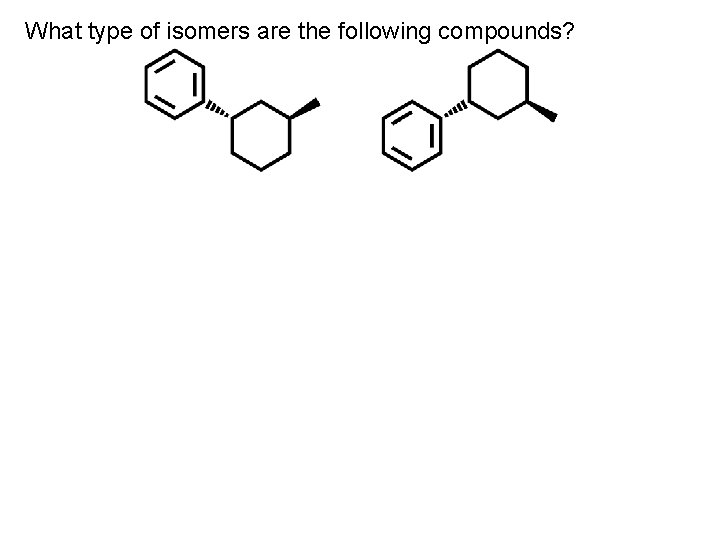
What type of isomers are the following compounds?

What could be true about a molecule with the following formula: C 10 H 16? (A) It may contain two triple bonds (B) It may contain no unsaturation (C) It may contain a ring and two double bonds (D) It may contain two rings and a triple bond

Explain why vinyl chloride will undergo free radical polymerization more readily than ethene.
Draw any molecule that would likely give a peak in the 1 H NMR with a quartet around 4. 0 ppm.

Draw a reasonable energy diagram (reaction coordinate diagram) for the substitution reaction between 2 -methyl-2 bromopentane and Na. OH.
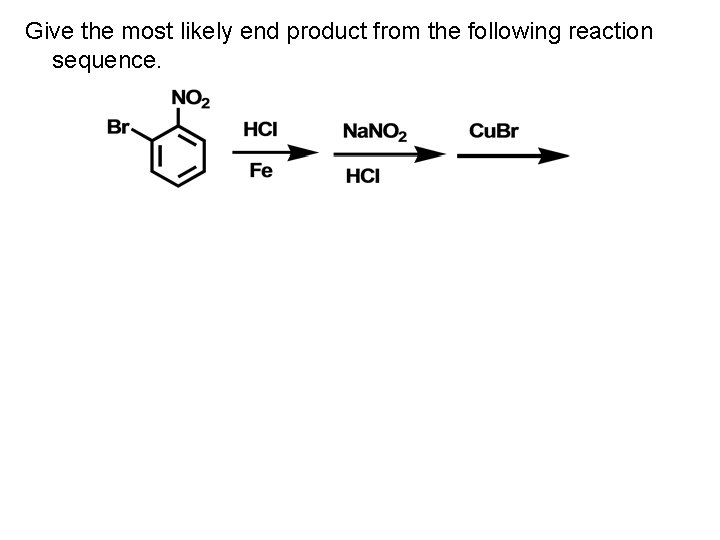
Give the most likely end product from the following reaction sequence.

Draw an alkyl halide that could readily undergo substitution by either SN 1 or SN 2.

Which should undergo SN 1 most readily? Benzyl bromide, 4 -methylbenzyl bromide, 4 -nitrobenzyl bromide, 4 -methoxybenzyl bromide

Starting from 2 -cyclopentenone, what reagents would be necessary to make 2, 3 -dipropylcyclopentanone?

If ethyl acetate were treated with ethoxide followed by dilute acid/H 2 O, draw the product that would most likely result.

Give reagents necessary to make N-methyl-1 -phenylethanamine in one step from acetophenone.

Show the antiperiplanar transition state for the elimination of bromocyclohexane.
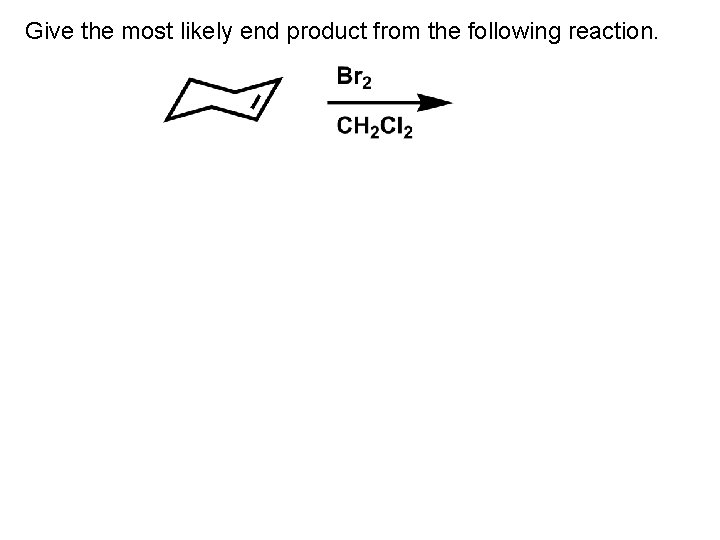
Give the most likely end product from the following reaction.

Draw 2 functional groups that can withdraw electrons through both induction and resonance.
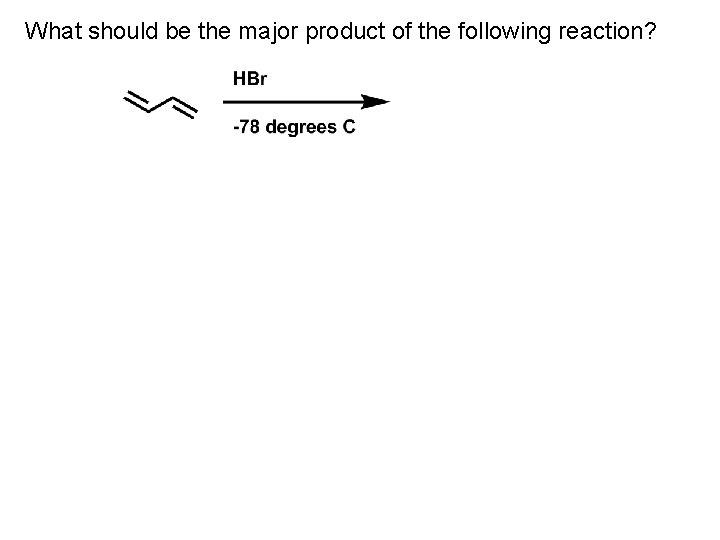
What should be the major product of the following reaction?
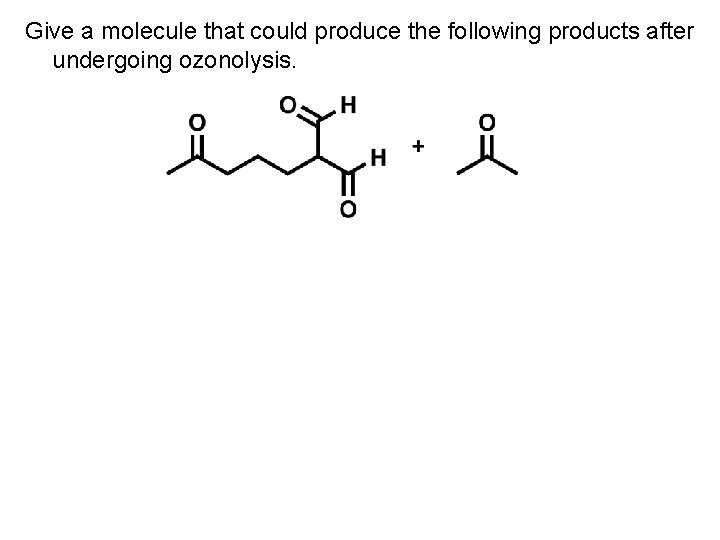
Give a molecule that could produce the following products after undergoing ozonolysis.
- Slides: 55