Figures from the book Impatient Revolution a guide

Figures from the book: #Impatient. Revolution a guide for impatient organizations Published by Dracaena Report dracaena-report@draccon. com Use and distribute freely with attribution. For noncommercial purposes only. Dracaena Report | #Impatient. Revolution
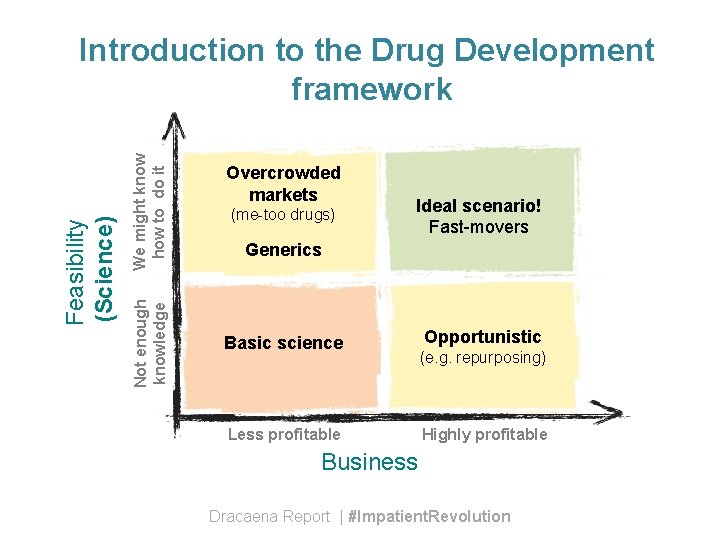
We might know how to do it Not enough knowledge Feasibility (Science) Introduction to the Drug Development framework Overcrowded markets (me-too drugs) Ideal scenario! Fast-movers Generics Basic science Less profitable Opportunistic (e. g. repurposing) Highly profitable Business Dracaena Report | #Impatient. Revolution
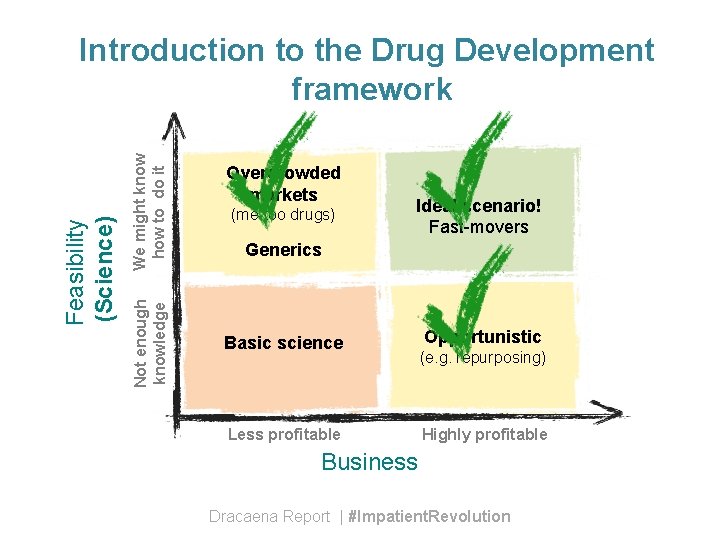
We might know how to do it Not enough knowledge Feasibility (Science) Introduction to the Drug Development framework Overcrowded markets (me-too drugs) Ideal scenario! Fast-movers Generics Basic science Less profitable Opportunistic (e. g. repurposing) Highly profitable Business Dracaena Report | #Impatient. Revolution
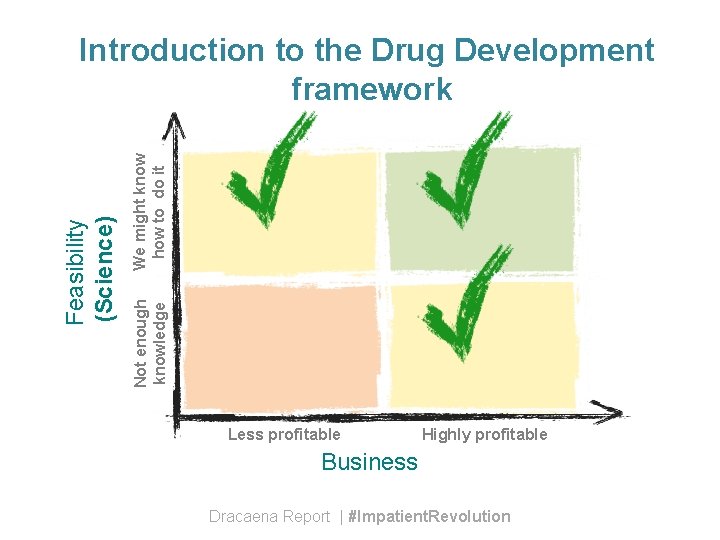
We might know how to do it Not enough knowledge Feasibility (Science) Introduction to the Drug Development framework Less profitable Highly profitable Business Dracaena Report | #Impatient. Revolution
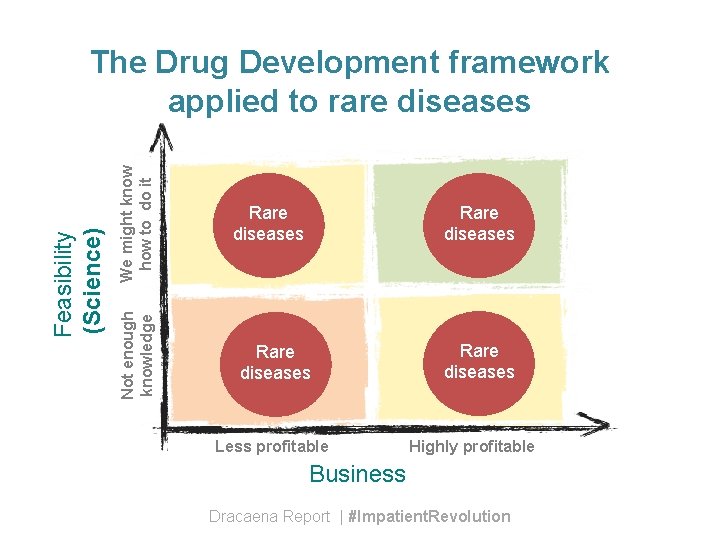
We might know how to do it Not enough knowledge Feasibility (Science) The Drug Development framework applied to rare diseases Rare diseases Less profitable Rare diseases Highly profitable Business Dracaena Report | #Impatient. Revolution
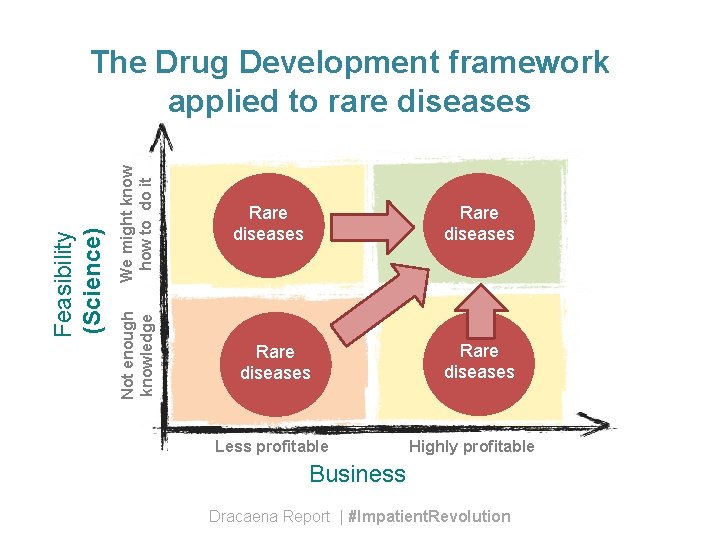
We might know how to do it Not enough knowledge Feasibility (Science) The Drug Development framework applied to rare diseases Rare diseases Less profitable Rare diseases Highly profitable Business Dracaena Report | #Impatient. Revolution
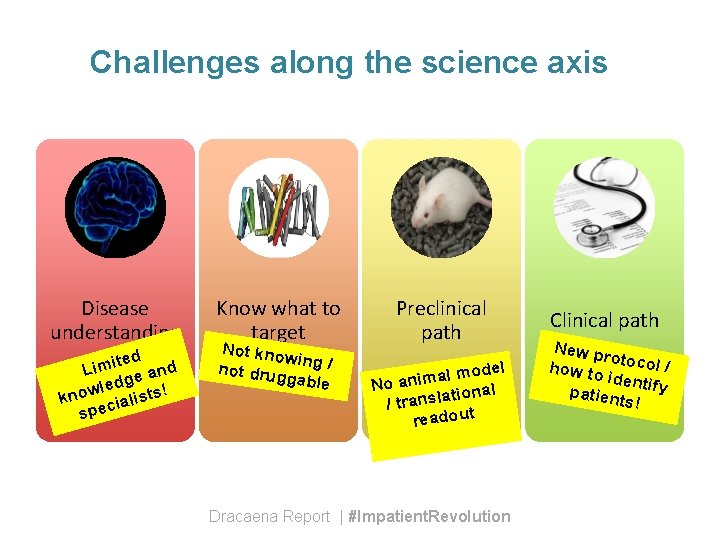
Challenges along the science axis Disease understanding ted d i m i L e an g d e l know cialists! spe Know what to target Not kno wi not drug ng / gable Preclinical path l model a m i n a No ional t a l s n a r /t readout Dracaena Report | #Impatient. Revolution Clinical path New p ro how to tocol / identif y patien ts!
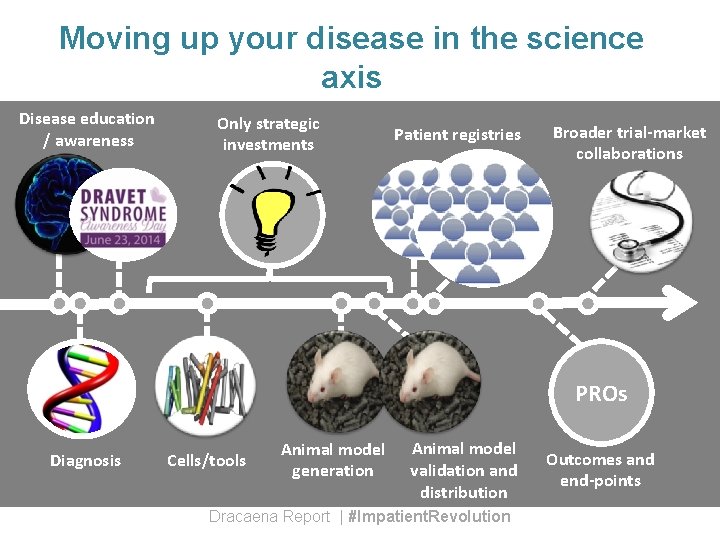
Moving up your disease in the science axis Disease education / awareness Only strategic investments Patient registries Broader trial-market collaborations PROs Diagnosis Cells/tools Animal model generation Animal model validation and distribution Dracaena Report | #Impatient. Revolution Outcomes and end-points
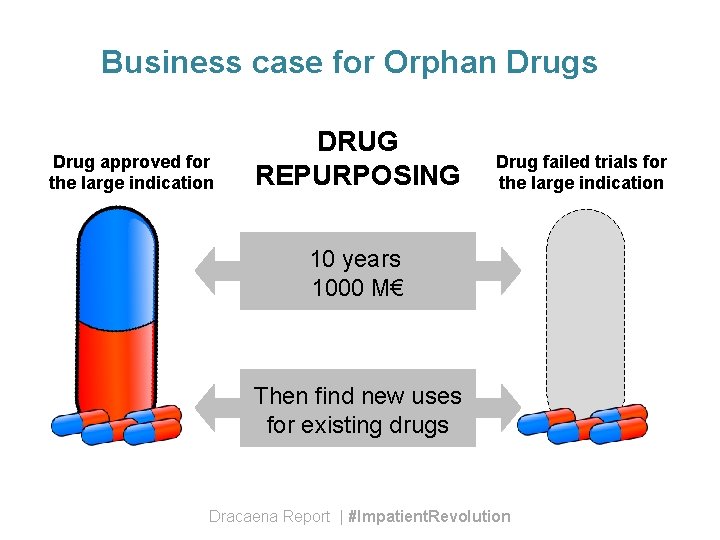
Business case for Orphan Drugs Drug approved for the large indication DRUG REPURPOSING Drug failed trials for the large indication 10 years 1000 M€ Then find new uses for existing drugs Dracaena Report | #Impatient. Revolution

Business case for Orphan Drugs Use rare diseases as a stepping stone before moving into larger (and riskier) indications • Shorter time • Less money • Test mechanism • Less risk Or the drug mechanism makes it useful for a cluster of rare diseases Dracaena Report | #Impatient. Revolution
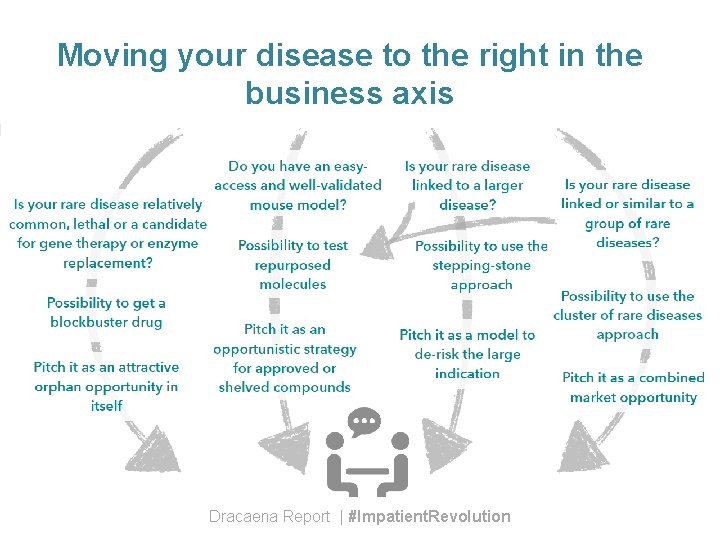
Moving your disease to the right in the business axis Dracaena Report | #Impatient. Revolution

Written by Ana Mingorance, Ph. D ana@draccon. com @CNSdrughunter Published by Dracaena Report, a division of Dracaena Consulting Calle Doctor Fleming 30, 1 izquierda 28036 Madrid, Spain www. draccon. com dracaena-report@draccon. com Copyright © 2017 Ana Mingorance
- Slides: 12