Fig 3 1 Water is the biological medium

Fig. 3 -1 Water is the biological medium on Earth All living organisms require water more than any other substance Most cells are surrounded by water, and cells themselves are about 70– 95% water The abundance of water is the main reason the Earth is habitable

Water is a ______ Molecule Diagram of a Water Molecule
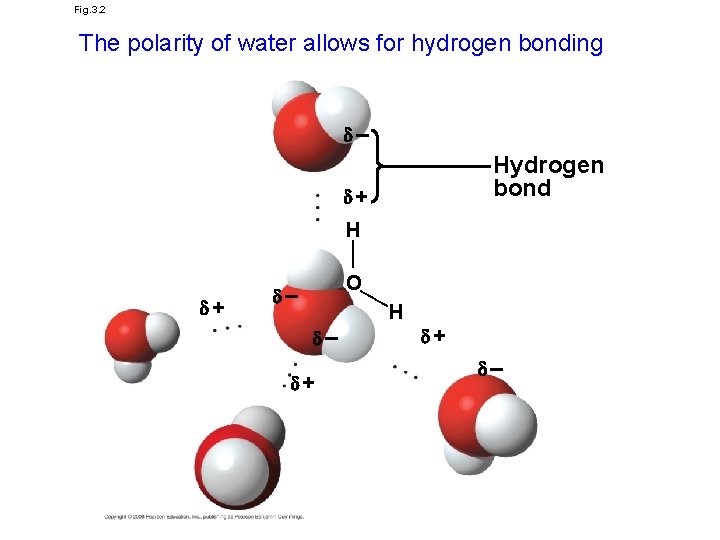
Fig. 3. 2 The polarity of water allows for hydrogen bonding – Hydrogen bond + —— H —— + O – H – + + –

Unique Properties of Water 1. 2. 3. 4. Cohesive behavior Ability to moderate temperature Expansion upon freezing Versatility as a solvent How do they facilitate an environment for life? Why is this important?

Cohesive Behavior Cohesion: attraction of like molecules ex/______ Adhesion: attraction of unlike molecules ex? _____ Why is this important?

Fig. 3 -3 Adhesion Water-conducting cells Direction of water movement Cohesion 150 µm

Fig. 3 -4 Why does water have a high surface tension (measure of how hard it is to break the surface of a liquid)?

Moderation of Temperature • Water has a high specific heat (1 cal/g/ºC) - amount of heat that must be absorbed or lost for 1 g of that substance to change its temperature by 1ºC • Water absorbs heat from warmer air and releases stored heat to cooler air (slight change in own temperature) Why is this important? How can water’s high specific heat be traced to hydrogen bonding?

• Heat of vaporization: is the heat a liquid must absorb for 1 g to be converted to gas • Evaporative Cooling: as a liquid evaporates, its remaining surface cools – helps stabilize temperatures in organisms and bodies of water

Expansion Upon Freezing Why does ice float?

Fig. 3 -6 Expansion Upon Freezing Why does ice float? Hydrogen bond Ice Hydrogen bonds are stable Less Dense: Below ____ Why is this important? Liquid water Hydrogen bonds break and re-form More Dense: Above ____

Versatility as a Solvent • Solution: is a liquid that is a homogeneous mixture of substances • Solvent: is the dissolving agent of a solution • The solute is the substance that is dissolved • An aqueous solution is one in which water is the solvent
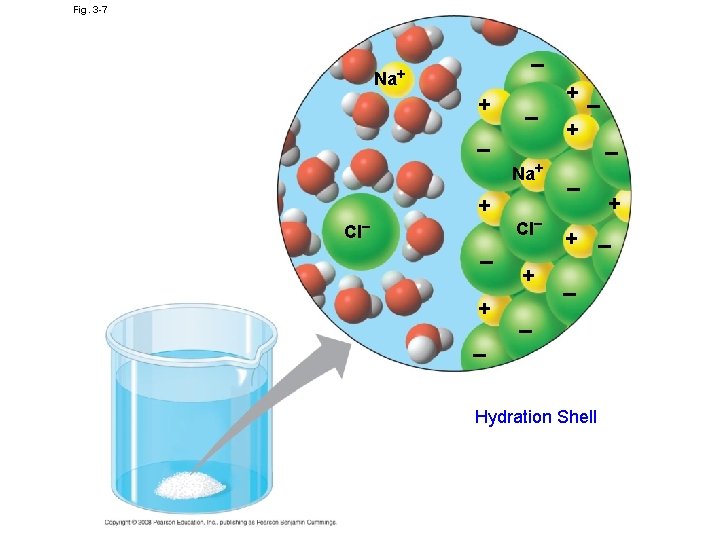
Fig. 3 -7 + – – Cl– + + + – + – – Cl– Na+ – – Na+ Hydration Shell
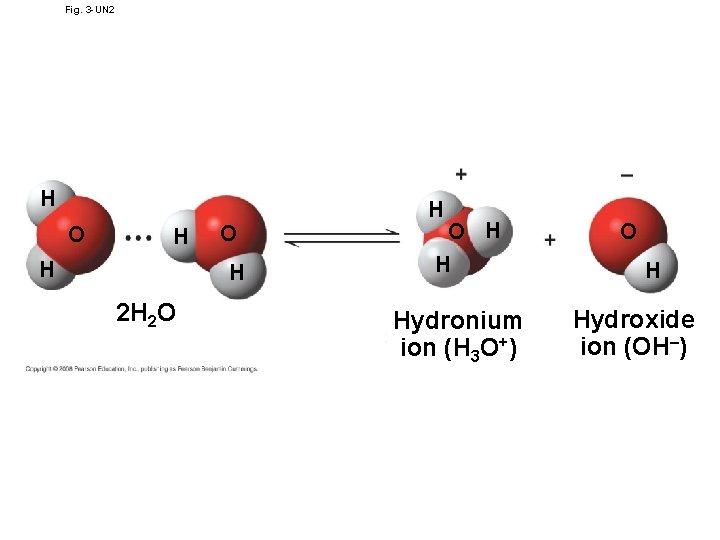
Fig. 3 -UN 2 H O H 2 H 2 O H H Hydronium ion (H 3 O+) O H Hydroxide ion (OH–)

• The dissociation of water molecules has a great effect on organisms (the chemistry of cell) • Most biological fluids have p. H values in the range of 6 to 8 • Buffers: substances that minimize changes in concentrations of H+ and OH– in a solution • Teaching the Blood Buffennnnnnnr System using a Demonstration • http: //vimeo. com/20298747
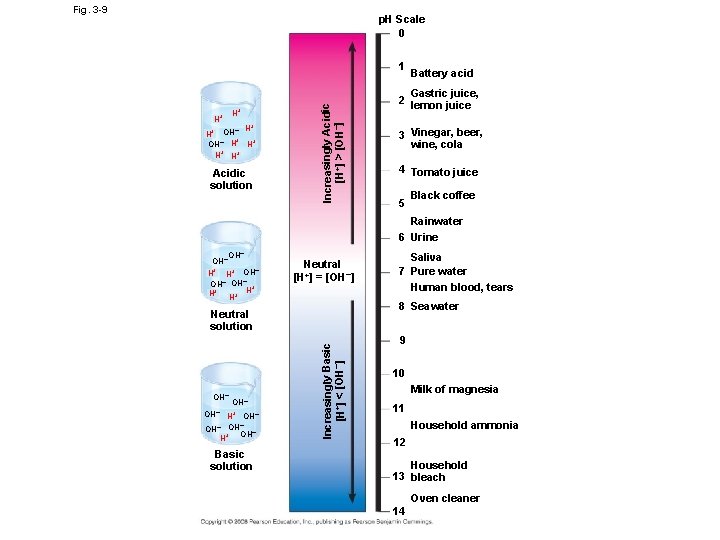
Fig. 3 -9 p. H Scale 0 H+ H+ + – H H+ OH– H H+ H+ H+ Acidic solution Increasingly Acidic [H+] > [OH–] 1 Battery acid Gastric juice, 2 lemon juice 3 Vinegar, beer, wine, cola 4 Tomato juice 5 Black coffee Rainwater 6 Urine OH– H+ OH– OH– + H+ H+ H Neutral [H+] = [OH–] 8 Seawater OH– H+ OH– – OH OH– + H Basic solution Increasingly Basic [H+] < [OH–] Neutral solution OH– Saliva 7 Pure water Human blood, tears 9 10 Milk of magnesia 11 Household ammonia 12 Household 13 bleach Oven cleaner 14

• Human activities such as burning fossil fuels threaten water quality • CO 2 is released by fossil fuel combustion and contributes to: – A warming of earth called the “greenhouse” effect – Acidification of the oceans; this leads to a decrease in the ability of corals to form calcified reefs
- Slides: 17