FemPop Stenting T N Is ZILVER PTX DES










- Slides: 20

Fem-Pop Stenting: T N Is ZILVER PTX DES PLA IM The “De Facto” Stent to Deploy? Krishna Rocha-Singh, MD Chief Scientific Officer Prairie Heart Institute Saint John’s Hospital Springfield, IL

Krishna Rocha-Singh, MD Within the past 12 months, I or my spouse/partner have had a financial interest/arrangement or affiliation with the organization(s) listed below. Affiliation/Financial Relationship • Grant/Research Support • Consulting Fees/Honoraria • • • Major Stock Shareholder/Equity Royalty Income Ownership/Founder Intellectual Property Rights Other Financial Benefit Company • None • Medtronic, Alucent, Zimmer-Bio. Met, ROX Medical, • PQ Bypass • • None Convergence Consulting, LLC Yes VIVA Board Member

Is Zilver PTX DES the De Facto IMPLANT to Deploy? Lecture Goals: • Acknowledge the importance of well-designed RCTs/adjudicated registries in specific angiographic cohorts (i. e. , long lesions, severely calcified). • Consider the importance of long-term clinical data to assess clinical utility of technologies. • Review recently released data from the REAL PTX RCT of Zilver PTX DES vs. DCB

VIVA SFA Nitinol Stent Meta-Analysis: Limitations/Caveats • Exploratory analyses of published data – full pre-specification not possible since trials well-known and published • Variation in definitions – Adds variability but may enhance generalizability of lesion length and ABI findings – However, large sample size helps to offset variability concern • Sample size constrained by available data (n=999) Rocha-Singh et al, Cath Cardiovasc Interv, In Press

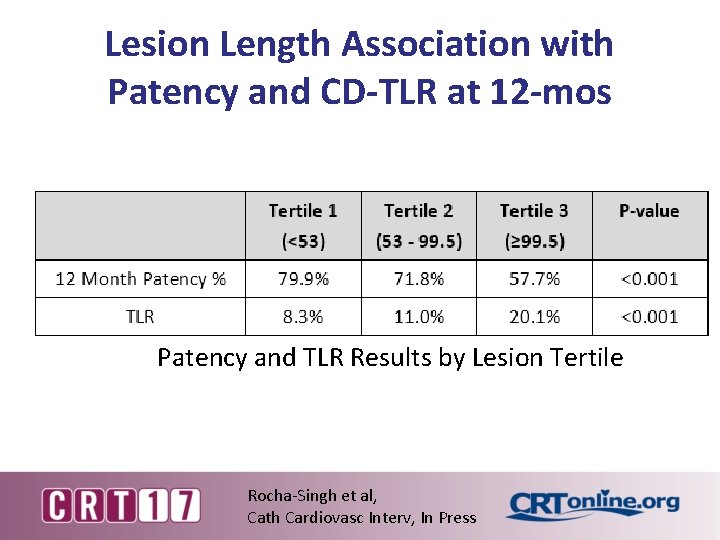
Lesion Length Association with Patency and CD-TLR at 12 -mos Patency and TLR Results by Lesion Tertile Rocha-Singh et al, Cath Cardiovasc Interv, In Press

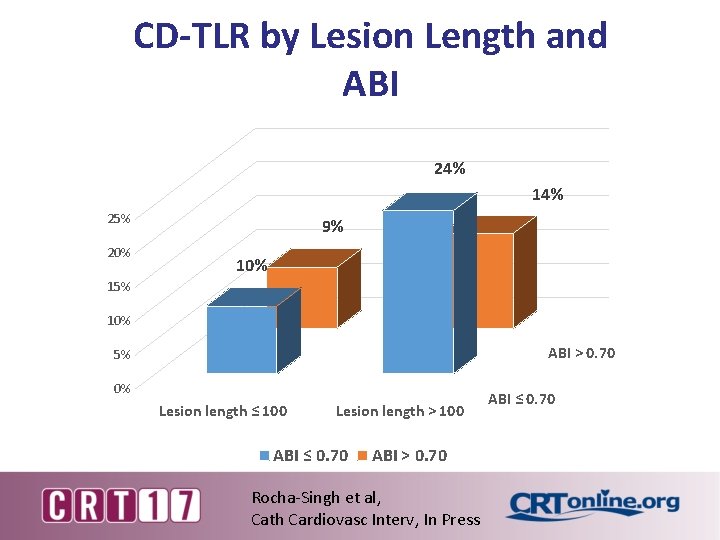
CD-TLR by Lesion Length and ABI 24% 14% 25% 20% 9% 10% 15% 10% ABI > 0. 70 5% 0% Lesion length ≤ 100 Lesion length > 100 ABI ≤ 0. 70 ABI > 0. 70 Rocha-Singh et al, Cath Cardiovasc Interv, In Press ABI ≤ 0. 70

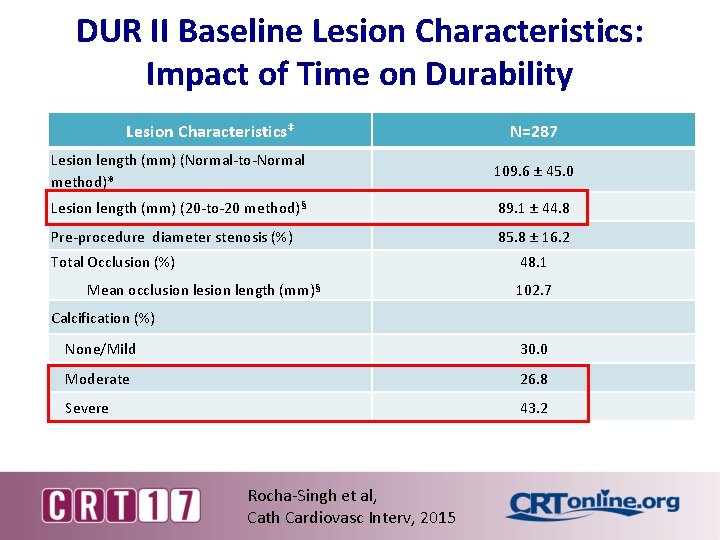
DUR II Baseline Lesion Characteristics: Impact of Time on Durability Lesion Characteristics‡ N=287 Lesion length (mm) (Normal-to-Normal method)* 109. 6 ± 45. 0 Lesion length (mm) (20 -to-20 method)§ 89. 1 ± 44. 8 Pre-procedure diameter stenosis (%) 85. 8 ± 16. 2 Total Occlusion (%) 48. 1 Mean occlusion length (mm)§ 102. 7 Calcification (%) None/Mild 30. 0 Moderate 26. 8 Severe 43. 2 Rocha-Singh et al, Cath Cardiovasc Interv, 2015

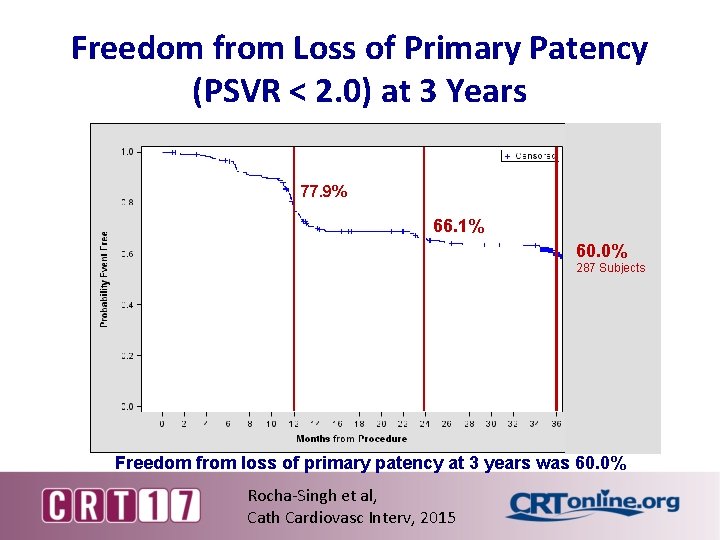
Freedom from Loss of Primary Patency (PSVR < 2. 0) at 3 Years 77. 9% 66. 1% 60. 0% 287 Subjects Freedom from loss of primary patency at 3 years was 60. 0% Rocha-Singh et al, Cath Cardiovasc Interv, 2015

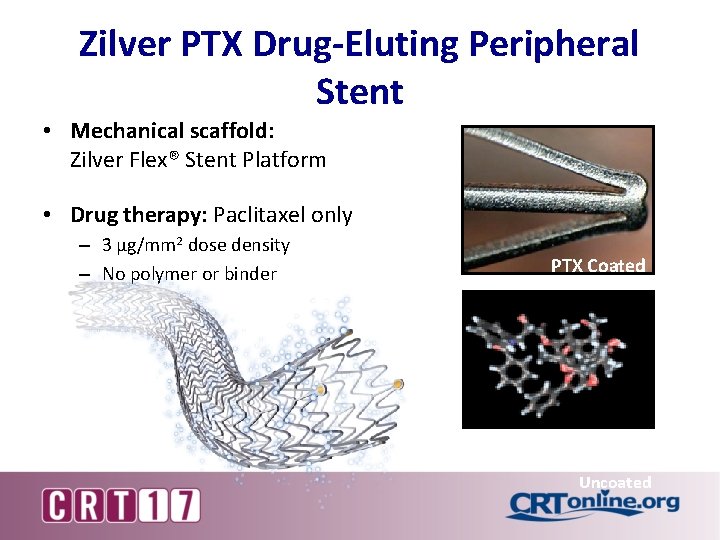
Zilver PTX Drug-Eluting Peripheral Stent • Mechanical scaffold: Zilver Flex® Stent Platform • Drug therapy: Paclitaxel only – 3 µg/mm 2 dose density – No polymer or binder PTX Coated Uncoated

Drug-Eluting Technologies: How Do We Target Their Appropriate Use? • 3 -Yr adjudicated follow-up data in TASC II A-B lesions (mean lesion length 9. 5 cm, calciumfree) note a 83. 5% freedom from CD-TLR rate • However, in longer (≥ 15 cm), calcified lesions (72%) provisional BMS use is required 40% to maintain a 1 -yr freedom from CD-TLR of 94% • Adjunct use of ‘vessel prep’ devices to reduce provisional stent rates and improve DCB results have be hypothesized.

IN. PACT Global Long Lesion Imaging Cohort: Lesion/Procedural Characteristics Lesions (N) Lesion Type: de novo restenotic (no ISR) ISR 164 Device Success [1] 99. 5% (442/444) Procedure Success [2] 99. 4% (155/156) Given the provisional BMS use in complex SFA Pre-dilatation 89. 8% (141/157) Post-dilatation 39. 1% (61/156) lesion morphologies, should primary DES Lesion Length Provisional Stent 40. 4% (63/156) - LL 15 -25 cm: 33. 3% (33/99) Total Occlusions 60. 4% (99/164) deployment be considered the primary - LL > 25 cm: 52. 6% (30/57) Calcification 71. 8% (117/163) 1. Severe 19. 6% (32/163) ‘de facto’ strategy? RVD (mm) Diameter Stenosis (pretreatment) Clinical Success [3] 83. 2% (134/161) 16. 8% (27/161) 0. 0% (0/161) 26. 40 ± 8. 61 cm 4. 594 ± 0. 819 90. 9% ± 14. 2 Dissections: 0 37. 9% (61/161) A-C 47. 2% (76/161) D-F 14. 9% (24/161) 2. 3. M. Jaff VIVA 2016 99. 4% (155/156) Device success: successful delivery, inflation, deflation and retrieval of the intact study balloon device without burst below the RBP Procedure success: residual stenosis of ≤ 50% (nonstented subjects) or ≤ 30% (stented subjects) by core lab (if core lab was not available then the site reported estimate was used) Clinical success: procedural success without procedural complications (death, major target limb amputation, thrombosis of the target lesion, or TVR) prior to discharge

REAL PTX Study Design A Pilot Study • Prospective, multicenter (5 EU sites), randomized, controlled trial • Zilver PTX DES vs DCB (~90% In. PACT Admiral) (1: 1) in native FP disease • N= 150 patients, 75 in each group • Stratification for lesion length for both groups (1: 1: 1) short: ≤ 10 cm middle: > 10 and ≤ 20 cm long: > 20 and ≤ 30 cm • Mean lesion length: 15. 3 ± 8. 8 cm • Independent angio and DUS core lab adjudication D. Scheinert, LINC 2017

Study Design Follow-up: 3 -Years Endpoints: - Primary: Primary Patency @ 12 Month (Duplex) - Secondary: Procedural sucess Major Adverse Event (Major Amputation; Death or TLR within 30 days) Primary Patency @ 24, 36 Month Clinically-driven target lesion revascularization (TLR) ABI, Improvement in Rutherford Categories, Walking capacity (WIQ) D. Scheinert, LINC 2017

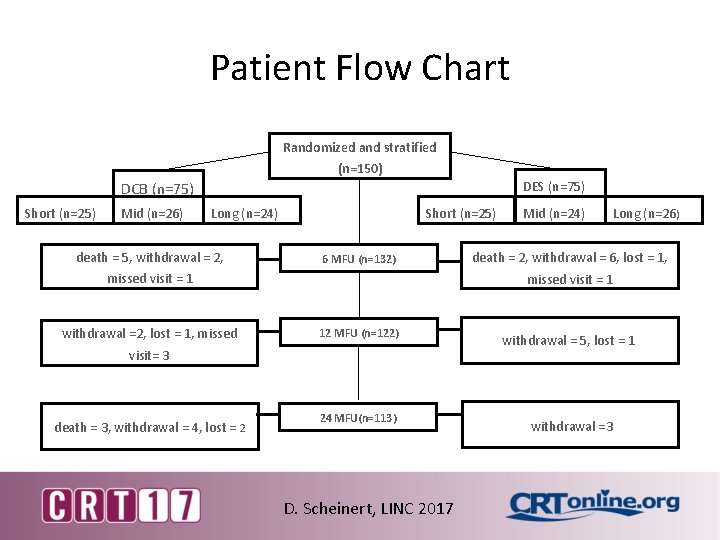
Patient Flow Chart Randomized and stratified (n=150) DES (n=75) DCB (n=75) Short (n=25) Mid (n=26) Long (n=24) Short (n=25) death = 5, withdrawal = 2, 6 MFU (n=132) missed visit = 1 Mid (n=24) Long (n=26) death = 2, withdrawal = 6, lost = 1, missed visit = 1 withdrawal =2, lost = 1, missed 12 MFU (n=122) visit= 3 withdrawal = 5, lost = 1 death = 3, withdrawal = 4, lost = 2 24 MFU(n=113) withdrawal =3 D. Scheinert, LINC 2017

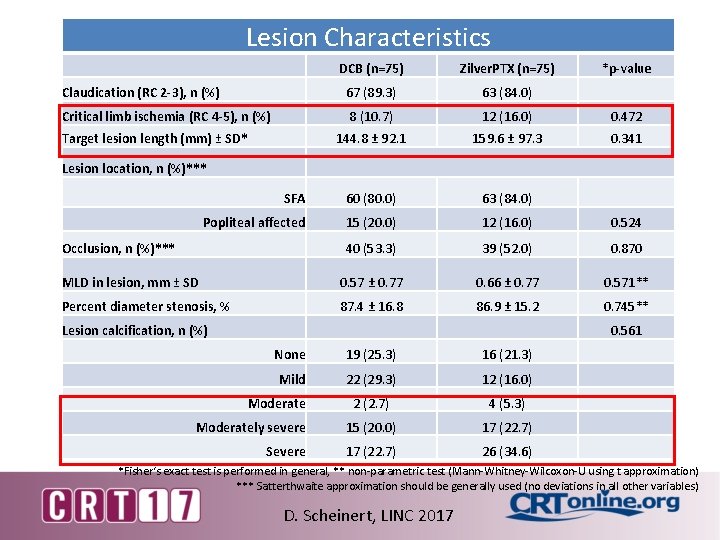
Lesion Characteristics DCB (n=75) Zilver. PTX (n=75) Claudication (RC 2 -3), n (%) 67 (89. 3) 63 (84. 0) Critical limb ischemia (RC 4 -5), n (%) 8 (10. 7) 12 (16. 0) 0. 472 144. 8 ± 92. 1 159. 6 ± 97. 3 0. 341 SFA 60 (80. 0) 63 (84. 0) Popliteal affected 15 (20. 0) 12 (16. 0) 0. 524 40 (53. 3) 39 (52. 0) 0. 870 MLD in lesion, mm ± SD 0. 57 ± 0. 77 0. 66 ± 0. 77 0. 571** Percent diameter stenosis, % 87. 4 ± 16. 8 86. 9 ± 15. 2 0. 745** Target lesion length (mm) ± SD* *p-value Lesion location, n (%)*** Occlusion, n (%)*** Lesion calcification, n (%) 0. 561 None 19 (25. 3) 16 (21. 3) Mild 22 (29. 3) 12 (16. 0) 2 (2. 7) 4 (5. 3) Moderately severe 15 (20. 0) 17 (22. 7) Severe 17 (22. 7) 26 (34. 6) Moderate *Fisher‘s exact test is performed in general, ** non-parametric test (Mann-Whitney-Wilcoxon-U using t approximation) *** Satterthwaite approximation should be generally used (no deviations in all other variables) D. Scheinert, LINC 2017

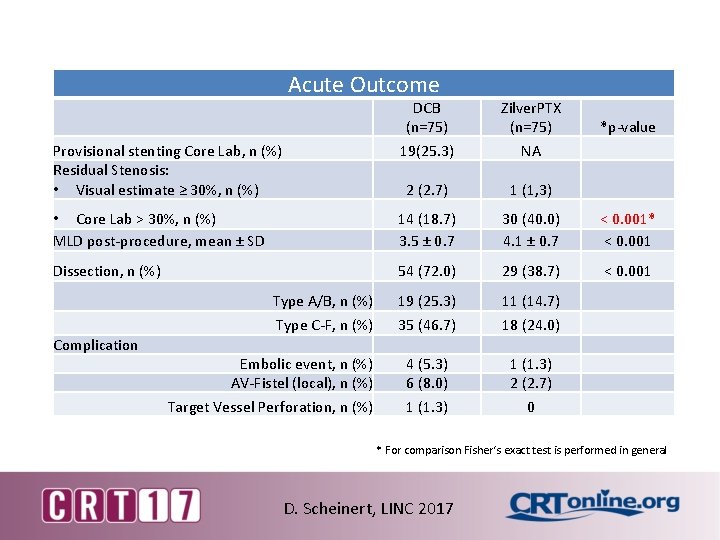
Acute Outcome DCB (n=75) Zilver. PTX (n=75) Provisional stenting Core Lab, n (%) Residual Stenosis: • Visual estimate ≥ 30%, n (%) 19(25. 3) NA 2 (2. 7) 1 (1, 3) • Core Lab > 30%, n (%) MLD post-procedure, mean ± SD 14 (18. 7) 3. 5 ± 0. 7 30 (40. 0) 4. 1 ± 0. 7 < 0. 001* < 0. 001 Dissection, n (%) 54 (72. 0) 29 (38. 7) < 0. 001 Type A/B, n (%) 19 (25. 3) 11 (14. 7) Type C-F, n (%) 35 (46. 7) 18 (24. 0) Embolic event, n (%) AV-Fistel (local), n (%) 4 (5. 3) 6 (8. 0) 1 (1. 3) 2 (2. 7) Target Vessel Perforation, n (%) 1 (1. 3) 0 Complication *p-value * For comparison Fisher‘s exact test is performed in general D. Scheinert, LINC 2017

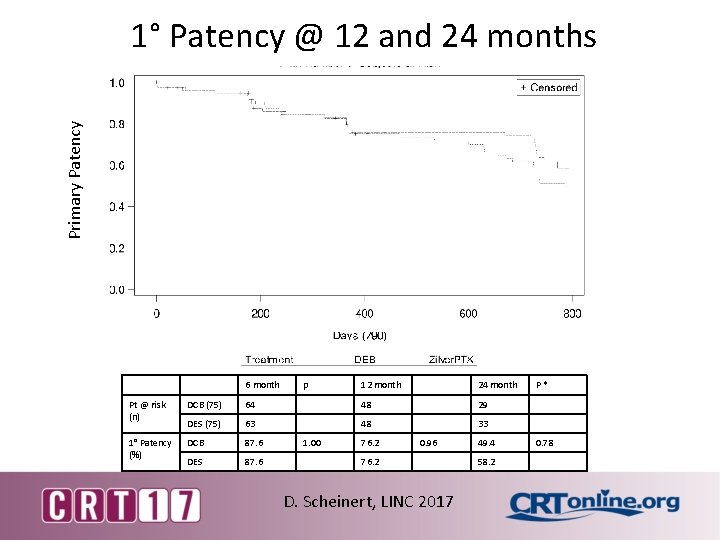
Primary Patency 1° Patency @ 12 and 24 months 6 month p 12 month 24 month Pt @ risk (n) DCB (75) 64 48 29 DES (75) 63 48 33 1° Patency (%) DCB 87. 6 DES 87. 6 1. 00 76. 2 0. 96 76. 2 D. Scheinert, LINC 2017 49. 4 58. 2 P * 0. 78

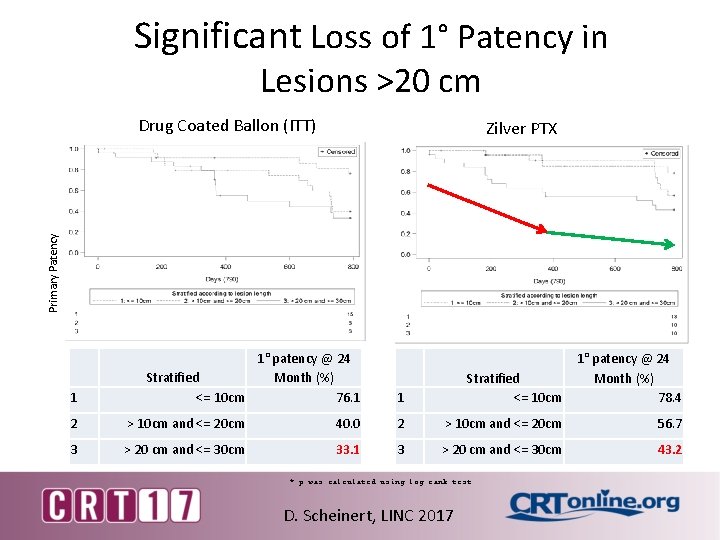
Significant Loss of 1° Patency in Lesions >20 cm Drug Coated Ballon (ITT) Primary Patency Zilver PTX 1 1° patency @ 24 Stratified Month (%) <= 10 cm 76. 1 1 Stratified <= 10 cm 1° patency @ 24 Month (%) 78. 4 2 > 10 cm and <= 20 cm 40. 0 2 > 10 cm and <= 20 cm 56. 7 3 > 20 cm and <= 30 cm 33. 1 3 > 20 cm and <= 30 cm 43. 2 * p was calculated using log rank test D. Scheinert, LINC 2017

Residual Stenosis ≥ 30% Acute Outcome Residual Stenosis • Visual estimate ≥ 30%, n (%) • Core Lab > 30%, n (%) DCB (n=75) Zilver. PTX (n=75) 2 (2. 7) 1 (1. 3) 14 (18. 7) 30 (40. 0) *p-value < 0. 001* Optimize acute outcome with consistent vessel preparation may potentially lead to better longterm patency D. Scheinert, LINC 2017

Fem-Pop Stenting: Is ZILVER PTX DES The “De Facto” Stent to Deploy? REAL PTX randomized controlled pilot trial results illustrates several points: --- All RCTs are not created equal --- “Vessel prep” and uniform deployments techniques both DCBs and Zilver PTX are essential --- Additional analysis of the ‘mode of failure’ should provide additional insights --- Cost considerations in technology choice must be assessed in future trials