Felletmdosts Korszer anyagok s technolgik MSc 2015 A








![Machined length [m] Cutting speed [m/min] Feed [mm/rev] The machined length versus the main Machined length [m] Cutting speed [m/min] Feed [mm/rev] The machined length versus the main](https://slidetodoc.com/presentation_image/42cda365f7888ed0964726fba2a5b6bc/image-19.jpg)









![Absorption degree [%] CO 2 laser Nd: : Ya. G laser Diode laser Excimer Absorption degree [%] CO 2 laser Nd: : Ya. G laser Diode laser Excimer](https://slidetodoc.com/presentation_image/42cda365f7888ed0964726fba2a5b6bc/image-37.jpg)


- Slides: 42

Felületmódosítás Korszerű anyagok és technológiák, MSc 2015

A felületi tulajdonságok tudománya: átfogó (interdiszciplináris) terület • A felület a tiszta fizikai és kémiai tulajdonságok szemszögéből. • Topográfiával kapcsolatos tulajdonságok (felületi érdesség). • Tágabb értelemben: „surface engineering”: komplex nézőpont • Kapcsolat a felületi tulajdonságok, az alkalmazás és az alkalmazott technológiák között. • (Tiszta fizikai és kémiai tulajdonságok: alapkutatás szemszöge. ) • A felületek tulajdonsága a felhasználók szempontjából: „surface engineering”.

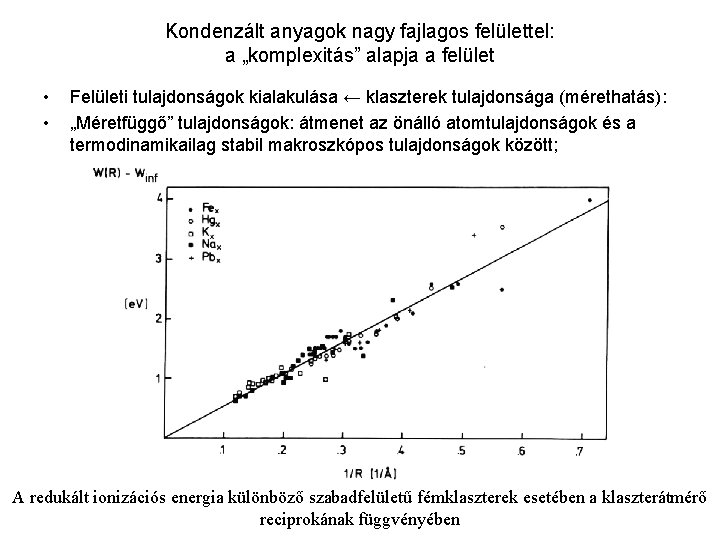
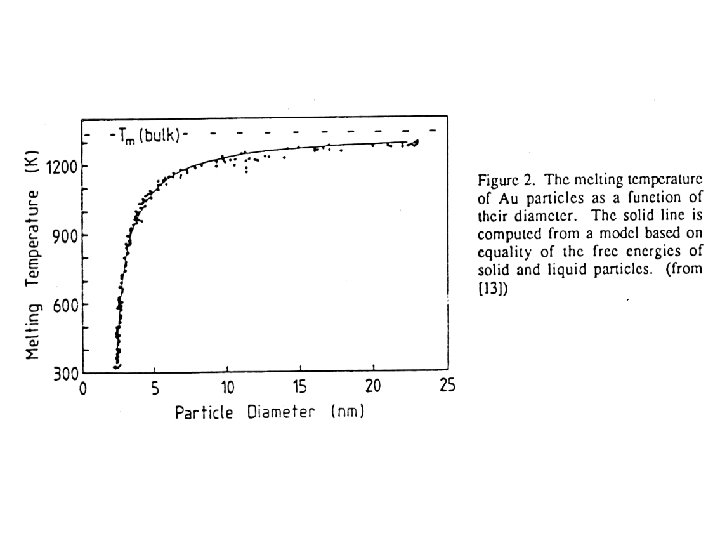
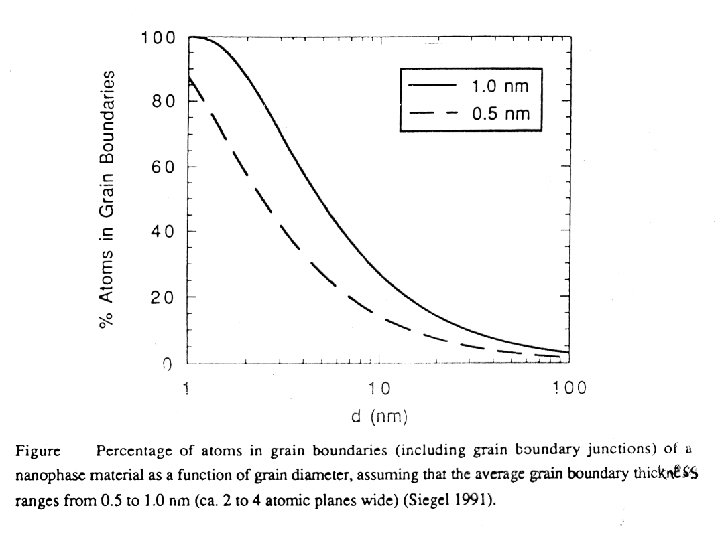
Kondenzált anyagok nagy fajlagos felülettel: a „komplexitás” alapja a felület • • Felületi tulajdonságok kialakulása ← klaszterek tulajdonsága (mérethatás): „Méretfüggő” tulajdonságok: átmenet az önálló atomtulajdonságok és a termodinamikailag stabil makroszkópos tulajdonságok között; A redukált ionizációs energia különböző szabadfelületű fémklaszterek esetében a klaszterátmérő reciprokának függvényében



Aim: the local modification of surface properties changing either the local composition or the structure or both of them. The desired surface properties are often contradict to those in the bulk! (i. e. local hardness, local corrosion resistance) examples: development of peculiar compositional relations on the surface of semiconductors The development of hard, wear- or corrosion-resistant surfaces on cutting, drilling tools Development of optical or decorative layers Two alternatives: Covering the surface with protective layer (corrosion resistant layers) Structural or compositional changes in the surface layer

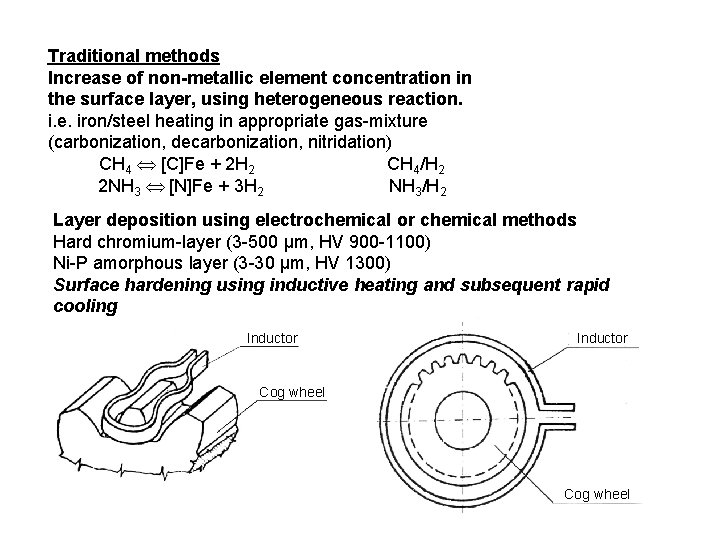
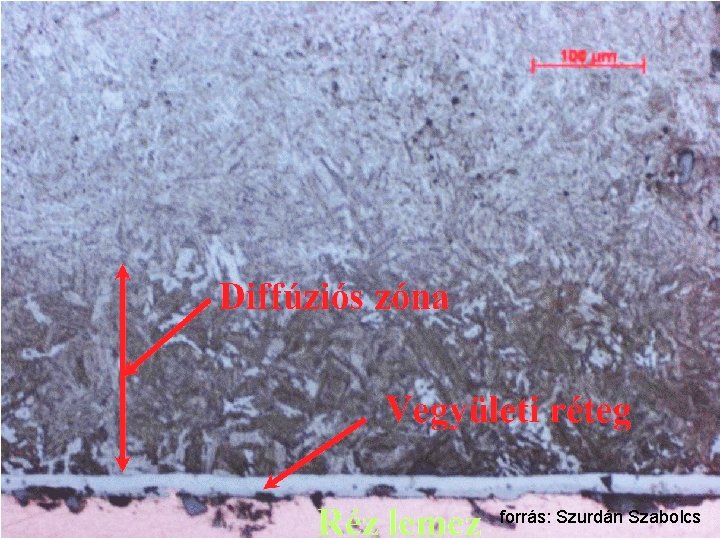
Traditional methods Increase of non-metallic element concentration in the surface layer, using heterogeneous reaction. i. e. iron/steel heating in appropriate gas-mixture (carbonization, decarbonization, nitridation) CH 4 [C]Fe + 2 H 2 CH 4/H 2 2 NH 3 [N]Fe + 3 H 2 NH 3/H 2 Layer deposition using electrochemical or chemical methods Hard chromium-layer (3 -500 μm, HV 900 -1100) Ni-P amorphous layer (3 -30 μm, HV 1300) Surface hardening using inductive heating and subsequent rapid cooling Inductor Cog wheel


forrás: Szurdán Szabolcs


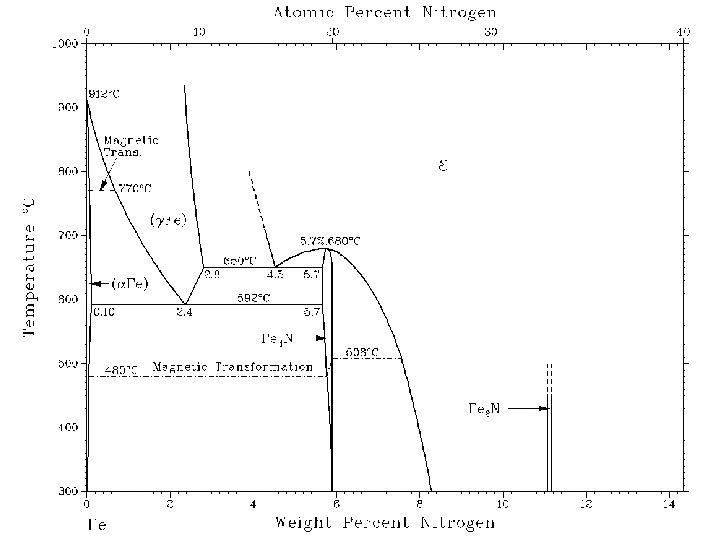
Példa: Al-Si bevonat „press hardening” technológiához ausztenitesítés: 880 -950°C - ~ 10 % Si x

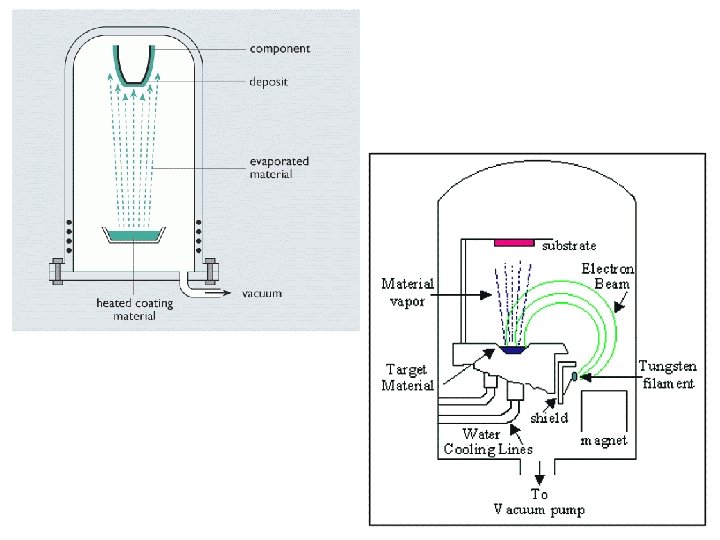
Deposition of thin layers by physical methods (Physical Vapour Deposition, PVD) • favour: • • High melting point metals can be deposited to low substrat temperature because T substrat can be low • The electrical conductivity of substrate not necessary • Complex multilayers can be deposited • Alloy deposition is also possible • „clean technology” (without the formation of products causing environmental pollution) • limits: • Expensive (vacuum circumstances)

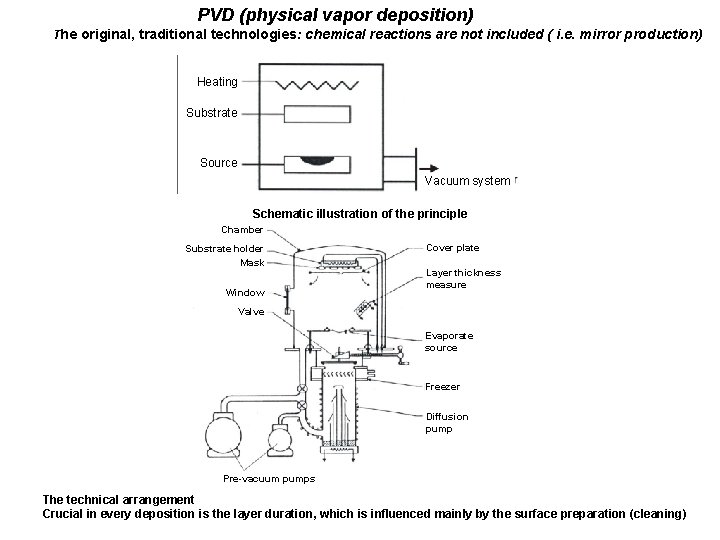
PVD (physical vapor deposition) The original, traditional technologies: chemical reactions are not included ( i. e. mirror production) Heating Substrate Source Vacuum system Schematic illustration of the principle Chamber Substrate holder Mask Window Cover plate Layer thickness measure Valve Evaporate source Freezer Diffusion pump Pre-vacuum pumps The technical arrangement Crucial in every deposition is the layer duration, which is influenced mainly by the surface preparation (cleaning)

Physical Vapour Deposition Source: Platit


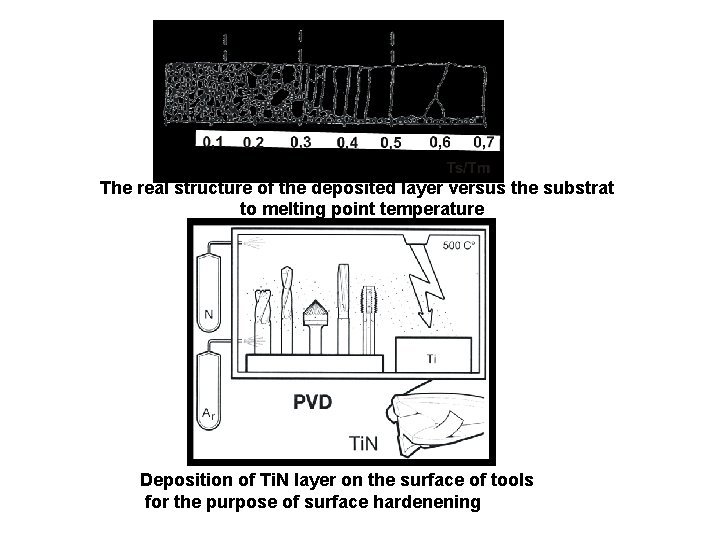
The real structure of the deposited layer versus the substrat to melting point temperature Deposition of Ti. N layer on the surface of tools for the purpose of surface hardenening

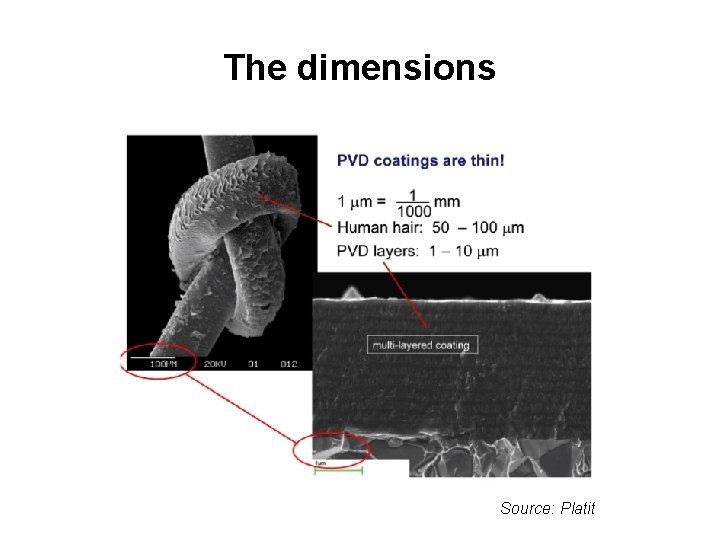
The dimensions Source: Platit

Some another exmples Source: Platit
![Machined length m Cutting speed mmin Feed mmrev The machined length versus the main Machined length [m] Cutting speed [m/min] Feed [mm/rev] The machined length versus the main](https://slidetodoc.com/presentation_image/42cda365f7888ed0964726fba2a5b6bc/image-19.jpg)
Machined length [m] Cutting speed [m/min] Feed [mm/rev] The machined length versus the main machining parameters: cutting speed (a) and feed (b) ( • Ti. N coated, o Ti. N coated and newly grinded, □nitride coating ■ without coating

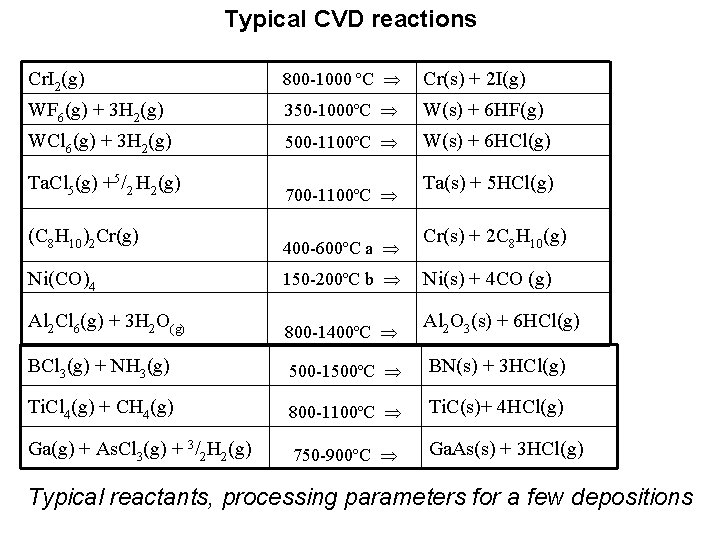
Chemical Vapour Deposition, (CVD) basic principles In the original form, the procedure consist of two isothermal reactions: a. ) at T 1 temperature M +n. X MXn (M layer forming metal , X halogen) At T 1 a volatile compound is formed b. ) at T 2 (T 2 > T 1 ) MXn M + n. X Thermal decomposition of MXn occur on the substrate surface Subsequently the decomposition process, the halogen molecule is circuated (in order to form new volatile molecules) Typical chemical reactions in the CVD procedure: Metal-halogenides are often used as precursor materials in these techniques. The reason: volatile compounds (high tension even at low temperatures!) (see tables) Besides the metal-layer depositions, the method also used for compound depositions (refractory carbides, silicides, borides)

Typical CVD reactions Cr. I 2(g) 800 -1000 ºC Cr(s) + 2 I(g) WF 6(g) + 3 H 2(g) 350 -1000ºC W(s) + 6 HF(g) WCl 6(g) + 3 H 2(g) 500 -1100ºC W(s) + 6 HCl(g) Ta. Cl 5(g) +5/2 H 2(g) 700 -1100ºC (C 8 H 10)2 Cr(g) 400 -600ºC a Cr(s) + 2 C 8 H 10(g) Ni(CO)4 150 -200ºC b Ni(s) + 4 CO (g) Al 2 Cl 6(g) + 3 H 2 O(g) 800 -1400ºC Al 2 O 3(s) + 6 HCl(g) BCl 3(g) + NH 3(g) 500 -1500ºC BN(s) + 3 HCl(g) Ti. Cl 4(g) + CH 4(g) 800 -1100ºC Ti. C(s)+ 4 HCl(g) Ga(g) + As. Cl 3(g) + 3/2 H 2(g) 750 -900ºC Ga. As(s) + 3 HCl(g) Ta(s) + 5 HCl(g) Typical reactants, processing parameters for a few depositions

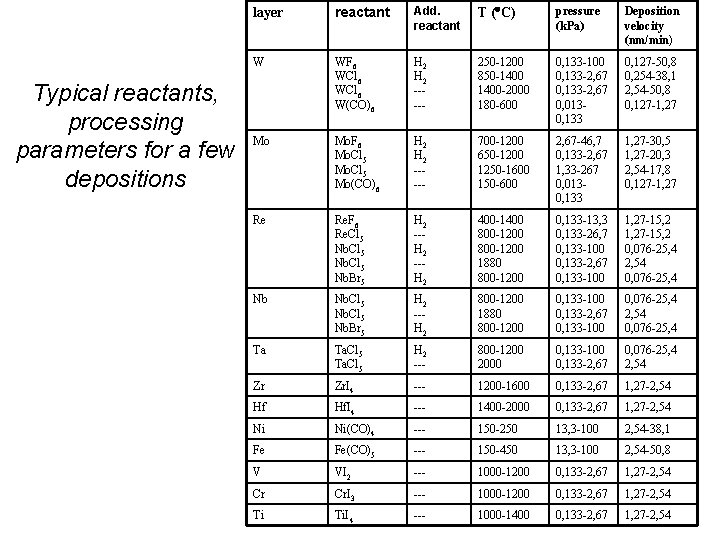
Typical reactants, processing parameters for a few depositions layer reactant Add. reactant T ( C) pressure (k. Pa) Deposition velocity (nm/min) W WF 6 WCl 6 W(CO)6 H 2 ----- 250 -1200 850 -1400 -2000 180 -600 0, 133 -100 0, 133 -2, 67 0, 0130, 133 0, 127 -50, 8 0, 254 -38, 1 2, 54 -50, 8 0, 127 -1, 27 Mo Mo. F 6 Mo. Cl 5 Mo(CO)6 H 2 ----- 700 -1200 650 -1200 1250 -1600 150 -600 2, 67 -46, 7 0, 133 -2, 67 1, 33 -267 0, 0130, 133 1, 27 -30, 5 1, 27 -20, 3 2, 54 -17, 8 0, 127 -1, 27 Re Re. F 6 Re. Cl 5 Nb. Br 5 H 2 --H 2 400 -1400 800 -1200 1880 800 -1200 0, 133 -13, 3 0, 133 -26, 7 0, 133 -100 0, 133 -2, 67 0, 133 -100 1, 27 -15, 2 0, 076 -25, 4 2, 54 0, 076 -25, 4 Nb Nb. Cl 5 Nb. Br 5 H 2 --H 2 800 -1200 1880 800 -1200 0, 133 -100 0, 133 -2, 67 0, 133 -100 0, 076 -25, 4 2, 54 0, 076 -25, 4 Ta Ta. Cl 5 H 2 --- 800 -1200 2000 0, 133 -100 0, 133 -2, 67 0, 076 -25, 4 2, 54 Zr Zr. I 4 --- 1200 -1600 0, 133 -2, 67 1, 27 -2, 54 Hf Hf. I 4 --- 1400 -2000 0, 133 -2, 67 1, 27 -2, 54 Ni Ni(CO)4 --- 150 -250 13, 3 -100 2, 54 -38, 1 Fe Fe(CO)5 --- 150 -450 13, 3 -100 2, 54 -50, 8 V VI 2 --- 1000 -1200 0, 133 -2, 67 1, 27 -2, 54 Cr Cr. I 3 --- 1000 -1200 0, 133 -2, 67 1, 27 -2, 54 Ti Ti. I 4 --- 1000 -1400 0, 133 -2, 67 1, 27 -2, 54

The important metals and ceramics produced by the CVD method Metals Cu, Be, Al, Ti, Zr, Hf, Th, Ge, Sn, Pb, V, Nb, Ta, As, Sb, Bi, Cr, Mo, W, U, Re, Fe, Co, Ni, Ru, Rh, Os, Ir, Pt Graphite carbides karbon C, B 4 C, Si. C, Ti. C, Zr. C, Hf. C, Th. C 2, VC, Nb 2 C, Ta 2 C, Cr 4 C, Cr 7 C 3, Cr 3 C 2, Mo. C, Mo 2 C, W 2 C, VC 2, V 2 C 3 Nitrides BN, Ti. N, Zr. N, VN, Nb. N, Ta. N, Si 3 N 4 Boron and borides B, Al. B 2, Ti. B 2, Zr. B 2, Th. B 4, Th. B, Nb. B, Ta. B, Mo 3 B 2, WB, Fr 2 B, Fe. B, Ni 3 B 2, Ni 2 B Silicon and silicides Si és Ti, Zr, Nb, Mo, W, Mn, Fe, Ni, Co and varies silicides Oxides Al 2 O 3, Be. O, Si. O 2, Zr. O 2, Cr 2 O 3, Sn. O 2

Examples for the compound-layer deposition • • The Ti. N layer deposition: 2 Ti. Cl 4 +4 H 2 +N 2 2 Ti. N +8 HCl Al 2 O 3 layer deposition: 2 Al. Cl 3 +3 CO 2 +H 2 Al 2 O 3 +3 CO +6 HCl

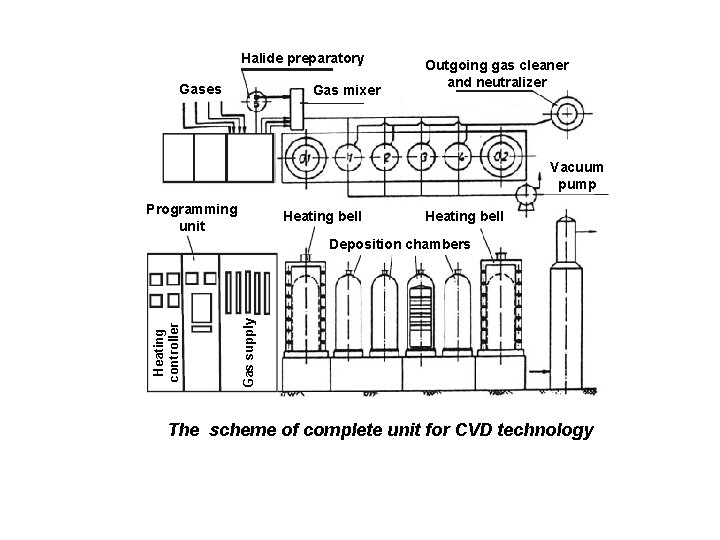
Halide preparatory Gases Gas mixer Outgoing gas cleaner and neutralizer Vacuum pump Programming unit Heating bell Gas supply Heating controller Deposition chambers The scheme of complete unit for CVD technology

The properties of deposits produced by CVD , layer-substrate: the compatibility

Favour: -high temperature: deposit accomodat even the complicated, irregular surfaces (inner surfaces can be covered)

Another example Tantállal bevont gégecső

The limits: • • The high substrat temperature (for example for structural or carbon steels is not recommended! 6 -800 o. C!) Expensive reactants

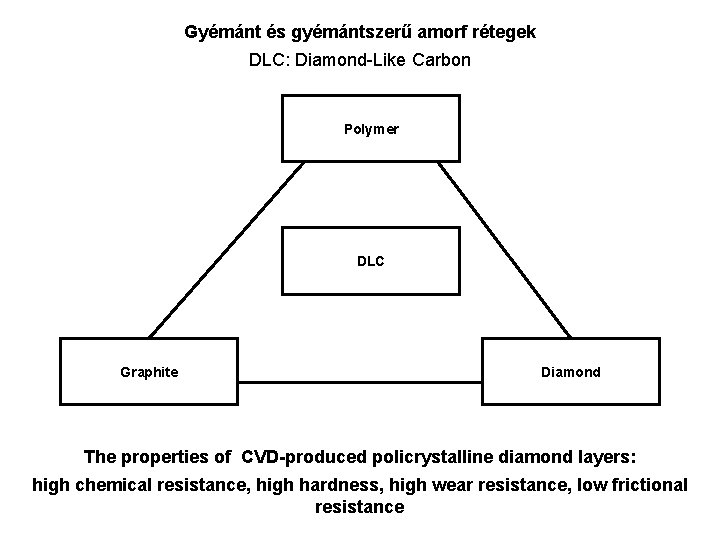
Gyémánt és gyémántszerű amorf rétegek DLC: Diamond-Like Carbon Polymer DLC Graphite Diamond The properties of CVD-produced policrystalline diamond layers: high chemical resistance, high hardness, high wear resistance, low frictional resistance

Hardness and young moduli of carbon- based coated layers Me: fém az elektromos vezetőképesség növeléséhez, fémkarbidok H: hidrogéntartalom a C 2 H 2 elbomlásából a: amorf fázis tartalmú réteg Si: szilíciumtartalom i-C: mátrix sp 3 -as kötésekkel, de amorf

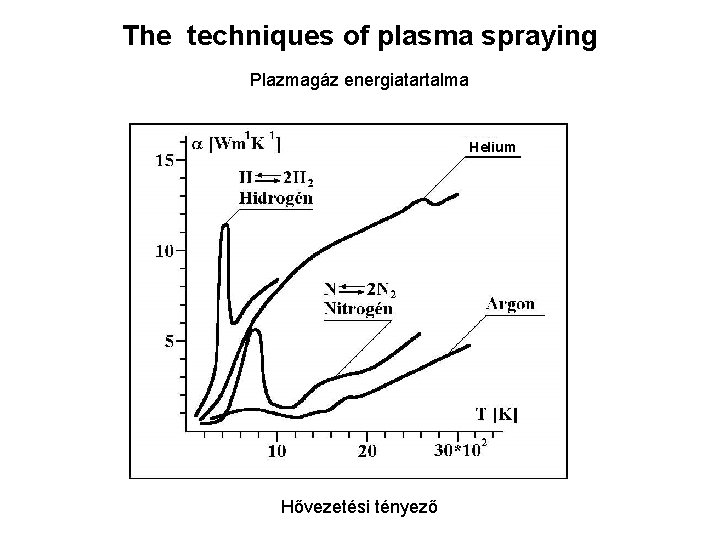
The techniques of plasma spraying Plazmagáz energiatartalma Helium Hővezetési tényező

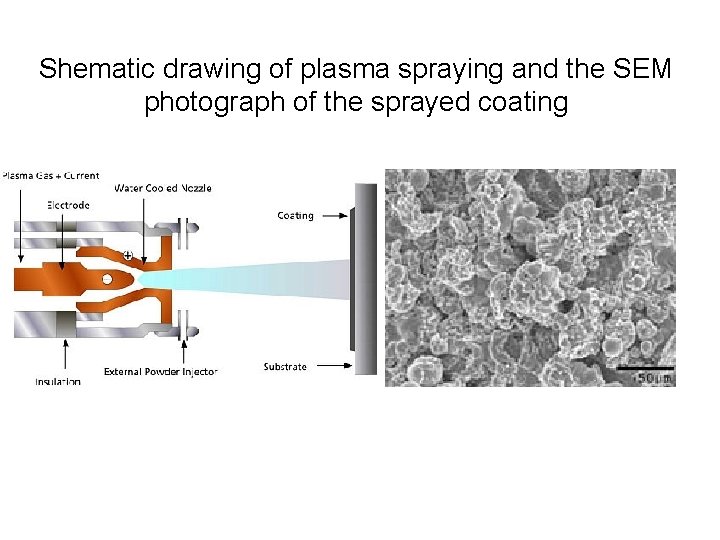
Shematic drawing of plasma spraying and the SEM photograph of the sprayed coating

Characteristics of plasma-sprayed layers Improvement of • Abrasive properties • Hardness • Corrosion resistance • (Especially in those cases, when the base material is low-cost • improvement of bio-compatibility (implanted parts) Succesfully applied for • the surface improvement of carbonsteels • surface hardness increase • corrosion resistance increase

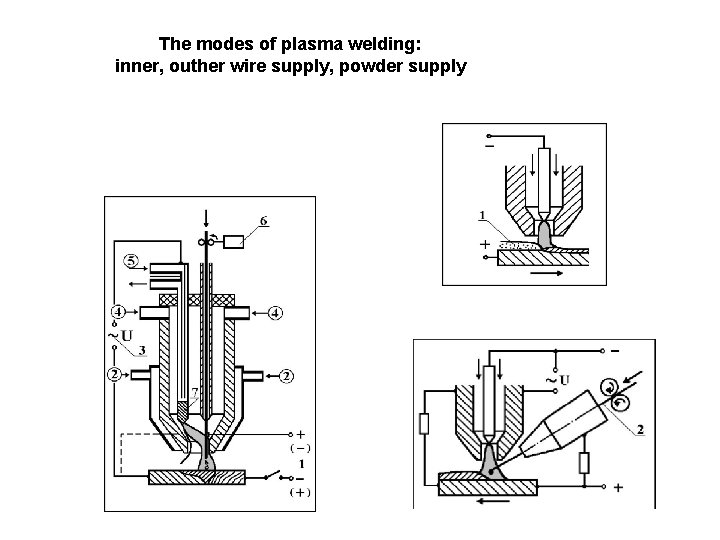
The modes of plasma welding: inner, outher wire supply, powder supply

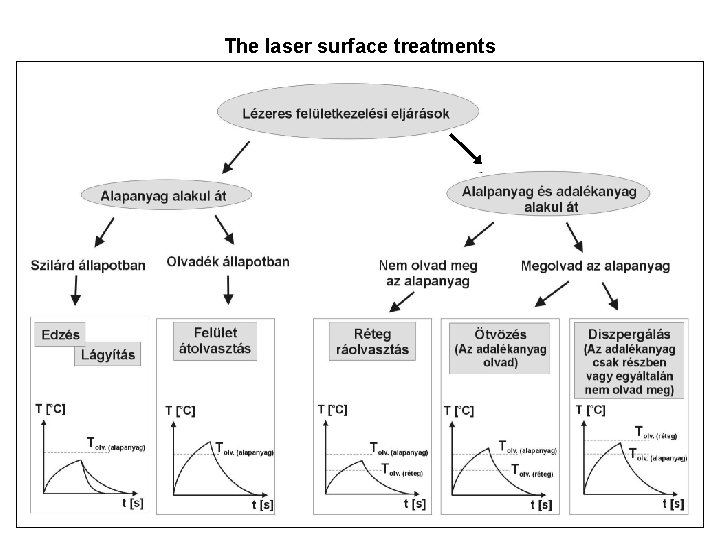
The laser surface treatments
![Absorption degree CO 2 laser Nd Ya G laser Diode laser Excimer Absorption degree [%] CO 2 laser Nd: : Ya. G laser Diode laser Excimer](https://slidetodoc.com/presentation_image/42cda365f7888ed0964726fba2a5b6bc/image-37.jpg)
Absorption degree [%] CO 2 laser Nd: : Ya. G laser Diode laser Excimer laser The effectivity highly depends on the absorption degree Iron Wavelength (mm) The absorption degree of various laser beams as a function of wavelengths

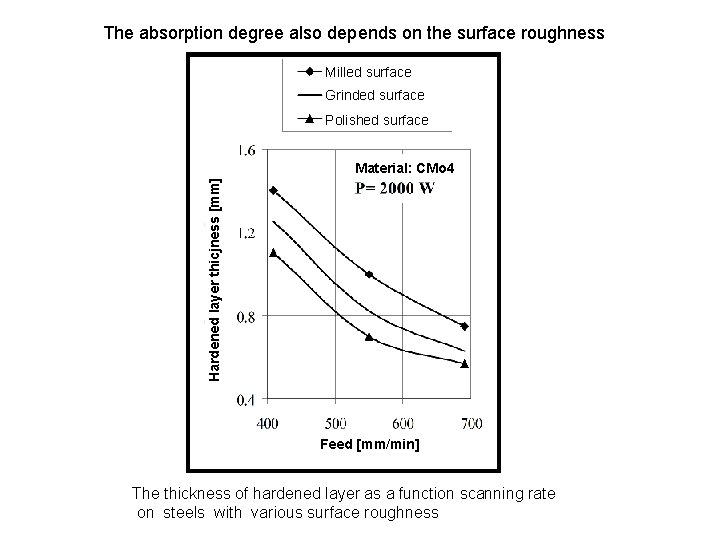
The absorption degree also depends on the surface roughness Milled surface Grinded surface Polished surface Hardened layer thicjness [mm] Material: CMo 4 Feed [mm/min] The thickness of hardened layer as a function scanning rate on steels with various surface roughness

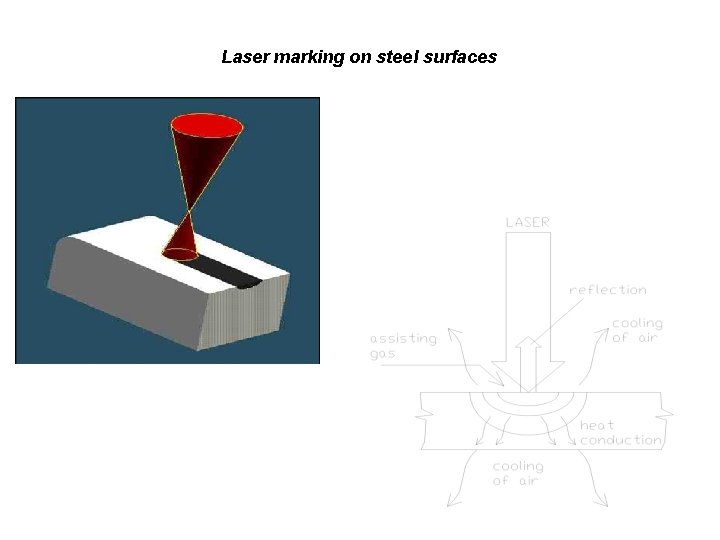
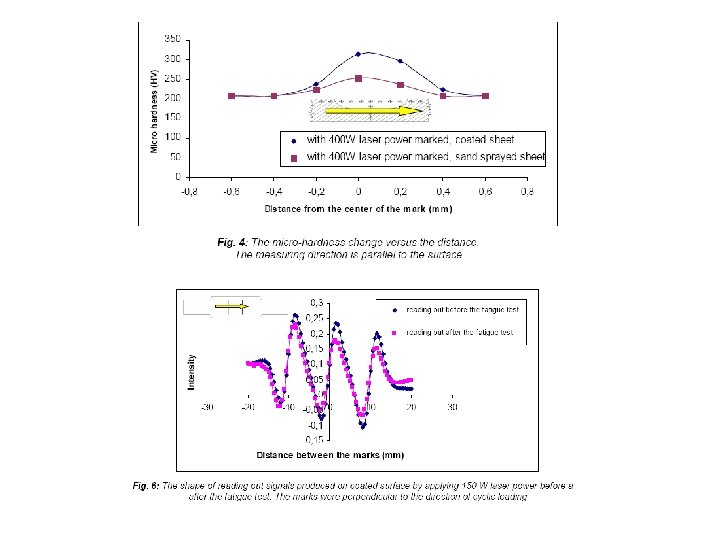
Laser marking on steel surfaces

The local structural change as the basis of code formation Depending on the applied energy density various structural changes can be induced (phase transformation, recrystallisation etc)

