Feldspar Group l l Most abundant mineral in

Feldspar Group l l Most abundant mineral in the crust 6 of 7 most common elements Defined through 3 endmembers F l Albite (Na), Anorthite (Ca), Orthoclase (K) Comprised of 2 series: Albite-anorthite (Na-Ca) F Albite-orthoclase (Na-K) F

Tectosilicates Feldspars Substitute Al 3+ for Si 4+ allows Na+ or K+ to be added Albite-Orthoclase Substitute two Al 3+ for Si 4+ allows Ca 2+ to be added Albite-Anorthite Albite: Na. Al. Si 3 O 8

Feldspar Group – Albite-Anorthite series l l l Complete solid solution Plagioclase Feldspars 6 minerals F Albite (Na) F Oligoclase F Andesine F Labradorite F Bytownite F Anorthite (Ca) Albite-Anorthite double duty F F l End-members (Pure Na or Ca) Minerals 90 -99. 99% Na or Ca Notation: F Anx. Aby An 20 Ab 80=Oligoclase

Feldspar Group – Albite-Anorthite series l Optical techniques to distinguish between plagioclase feldspars: Michel-Levy Method – uses extinction angles of twinned forms to determine An-Ab content F Combined Carlsbad-Albite Method uses Michel. Levy technique for both sides of a twin form F
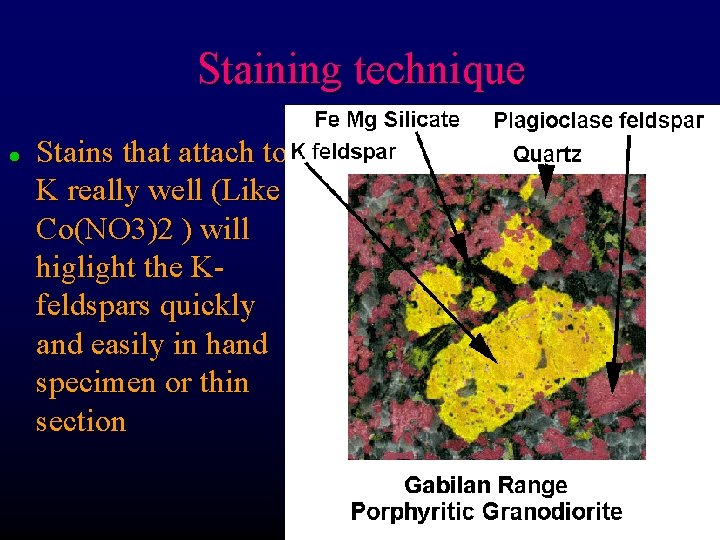
Staining technique l Stains that attach to K really well (Like Co(NO 3)2 ) will higlight the Kfeldspars quickly and easily in hand specimen or thin section

Feldspar Group – Albite-Orthoclase series Several minerals – Alkali Feldspars l l High – T minerals F Sanidine F Anorthoclase F Monalbite F High Albite Low Temperature exsolution at solvus F l Chicken soup separation Forms 2 minerals, in igneous rocks these are typically intergrowths, or exsolution lamellae – perthitic texture monalbite anorthoclase 1100 Temperature (ºC) l high albite 900 700 500 sanidine intermediate albite orthoclase low albite microcline Miscibility Gap 300 10 Orthoclase KAl. Si 3 O 8 30 50 % Na. Al. Si 3 O 8 70 90 Albite Na. Al. Si 3 O 8
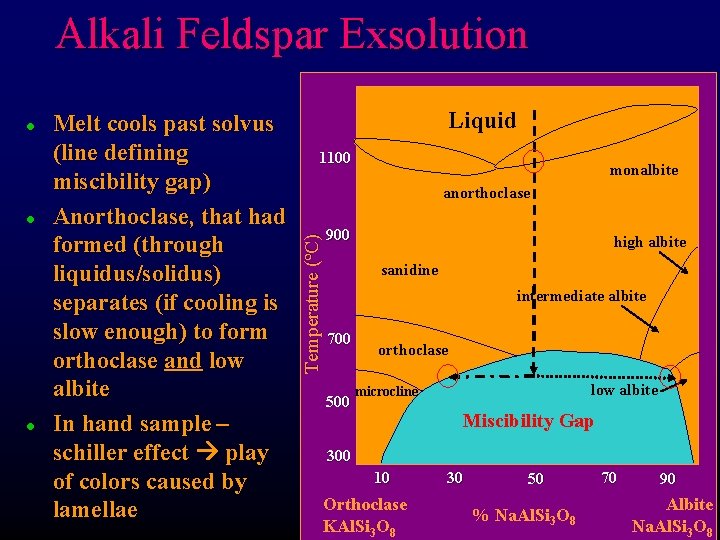
Alkali Feldspar Exsolution l l Melt cools past solvus (line defining miscibility gap) Anorthoclase, that had formed (through liquidus/solidus) separates (if cooling is slow enough) to form orthoclase and low albite In hand sample – schiller effect play of colors caused by lamellae Liquid 1100 monalbite anorthoclase Temperature (ºC) l 900 high albite sanidine intermediate albite 700 500 orthoclase low albite microcline Miscibility Gap 300 10 Orthoclase KAl. Si 3 O 8 30 50 % Na. Al. Si 3 O 8 70 90 Albite Na. Al. Si 3 O 8

Alkali Feldspar lamellae

Feldspathoid Group l l Very similar to feldspars and zeolites Include Nepheline, Analcime, and Leucite Also framework silicates, but with another Al substitution for Si Only occur in undersaturated rocks (no free Quartz, Sipoor) because they react with Si. O 2 to form feldspars
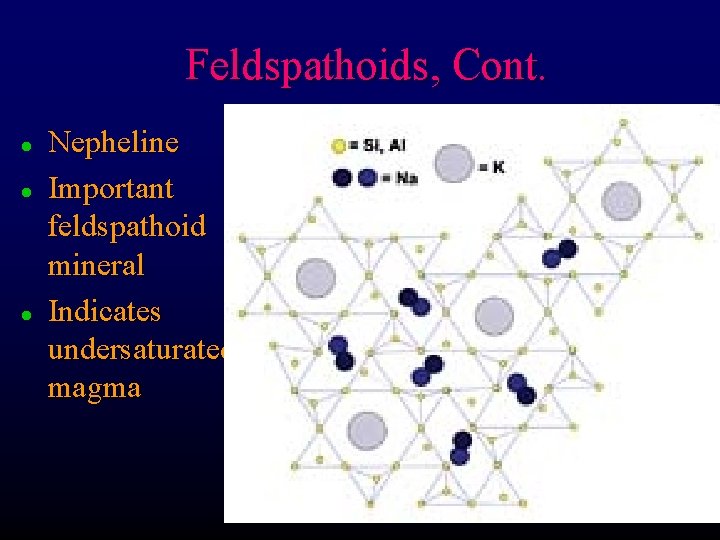
Feldspathoids, Cont. l l l Nepheline Important feldspathoid mineral Indicates undersaturated magma
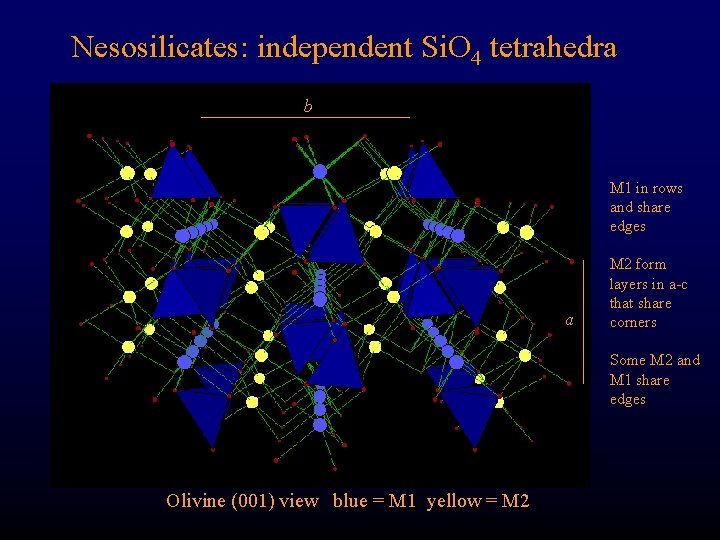
Nesosilicates: independent Si. O 4 tetrahedra b M 1 in rows and share edges a M 2 form layers in a-c that share corners Some M 2 and M 1 share edges Olivine (001) view blue = M 1 yellow = M 2

Olivine – complete solid solution l F Forsterite-Fayalite Fox. Fayalite – Fe end-member Forsterite – Mg end-member Olivine Occurrences: F F F Principally in mafic and ultramafic igneous and meta-igneous rocks Fayalite in meta-ironstones and in some alkalic granitoids Forsterite in some siliceous dolomitic marbles Monticellite Ca. Mg. Si. O 4 Ca M 2 (larger ion, larger site) High grade metamorphic siliceous carbonates

Distinguishing Forsterite-Fayalite l Petrographic Microscope F F F l l Index of refraction careful of zoning!! 2 V different in different composition ranges Pleochroism/ color slightly different Spectroscopic techniques – many ways to determine Fe vs. Mg Same space group (Pbnm), Orthorhombic, slight differences in unit cell dimensions only
![Inosilicates: single chains- pyroxenes b a sin Diopside: Ca. Mg [Si 2 O 6] Inosilicates: single chains- pyroxenes b a sin Diopside: Ca. Mg [Si 2 O 6]](http://slidetodoc.com/presentation_image_h/76e01d9186b93bfe21f435b404698ef8/image-14.jpg)
Inosilicates: single chains- pyroxenes b a sin Diopside: Ca. Mg [Si 2 O 6] Where are the Si-O-Si-O chains? ? Diopside (001) view blue = Si purple = M 1 (Mg) yellow = M 2 (Ca)
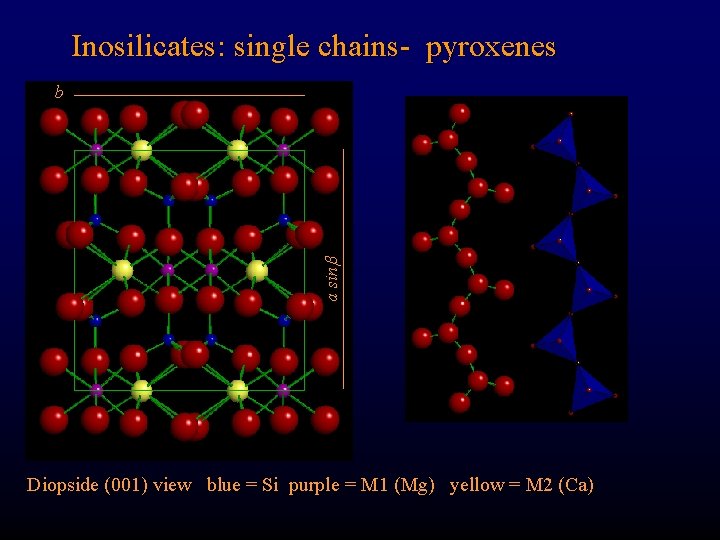
Inosilicates: single chains- pyroxenes a sin b Diopside (001) view blue = Si purple = M 1 (Mg) yellow = M 2 (Ca)
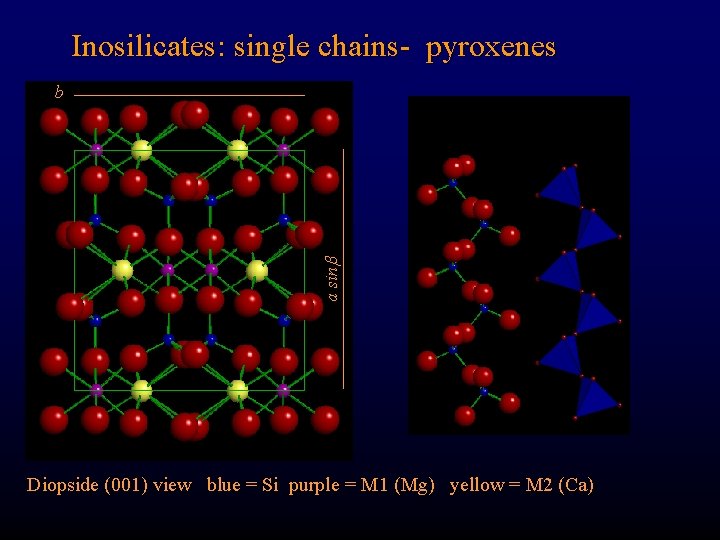
Inosilicates: single chains- pyroxenes a sin b Diopside (001) view blue = Si purple = M 1 (Mg) yellow = M 2 (Ca)
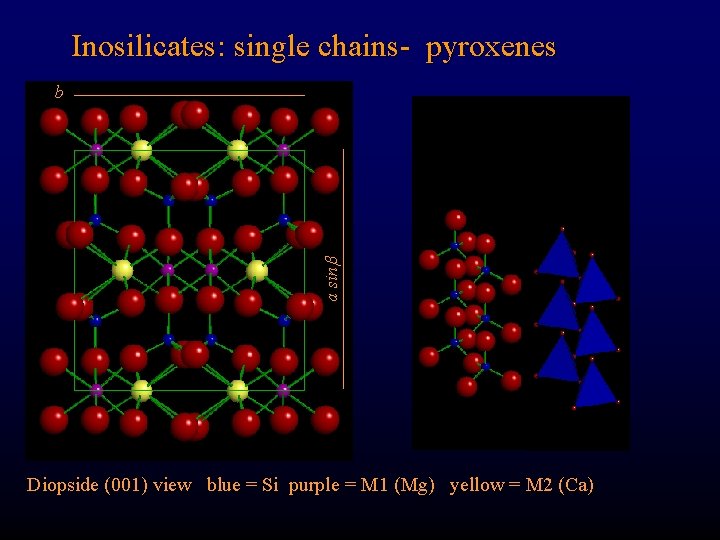
Inosilicates: single chains- pyroxenes a sin b Diopside (001) view blue = Si purple = M 1 (Mg) yellow = M 2 (Ca)
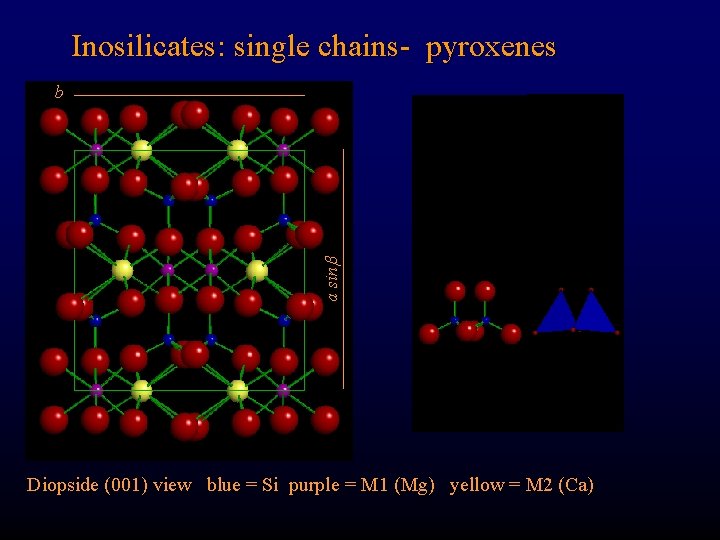
Inosilicates: single chains- pyroxenes a sin b Diopside (001) view blue = Si purple = M 1 (Mg) yellow = M 2 (Ca)
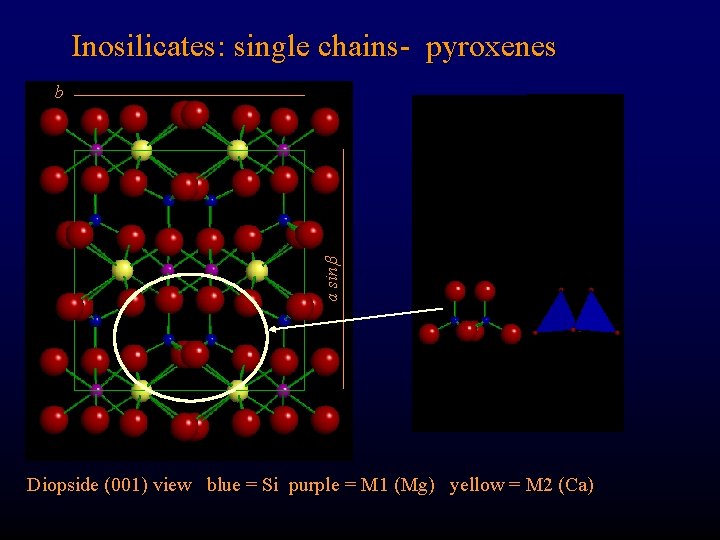
Inosilicates: single chains- pyroxenes a sin b Diopside (001) view blue = Si purple = M 1 (Mg) yellow = M 2 (Ca)
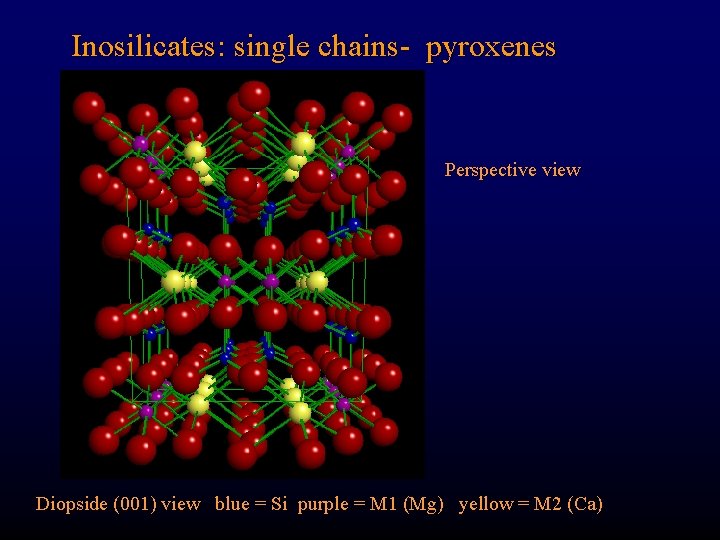
Inosilicates: single chains- pyroxenes Perspective view Diopside (001) view blue = Si purple = M 1 (Mg) yellow = M 2 (Ca)
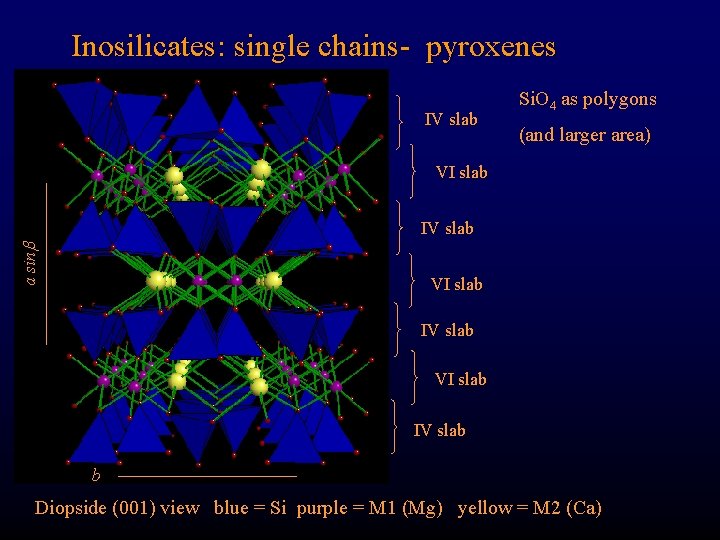
Inosilicates: single chains- pyroxenes IV slab Si. O 4 as polygons (and larger area) VI slab a sin IV slab VI slab IV slab b Diopside (001) view blue = Si purple = M 1 (Mg) yellow = M 2 (Ca)

Inosilicates: single chains- pyroxenes M 1 octahedron

Inosilicates: single chains- pyroxenes M 1 octahedron

Inosilicates: single chains- pyroxenes (+) M 1 octahedron (+) type by convention
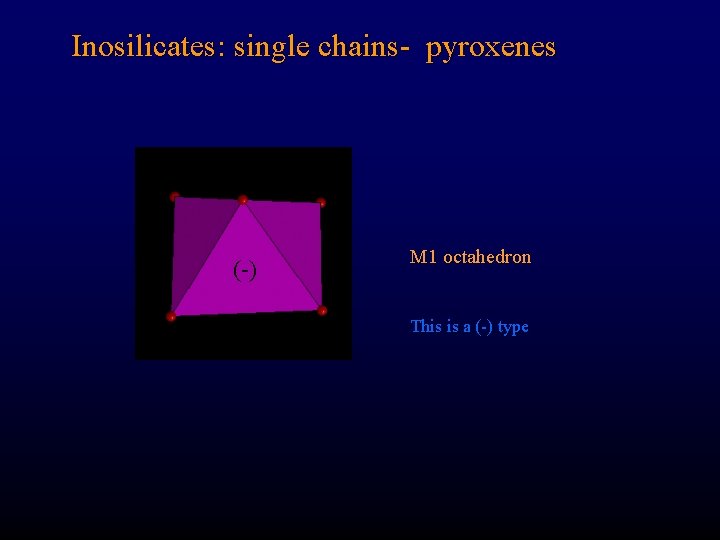
Inosilicates: single chains- pyroxenes (-) M 1 octahedron This is a (-) type
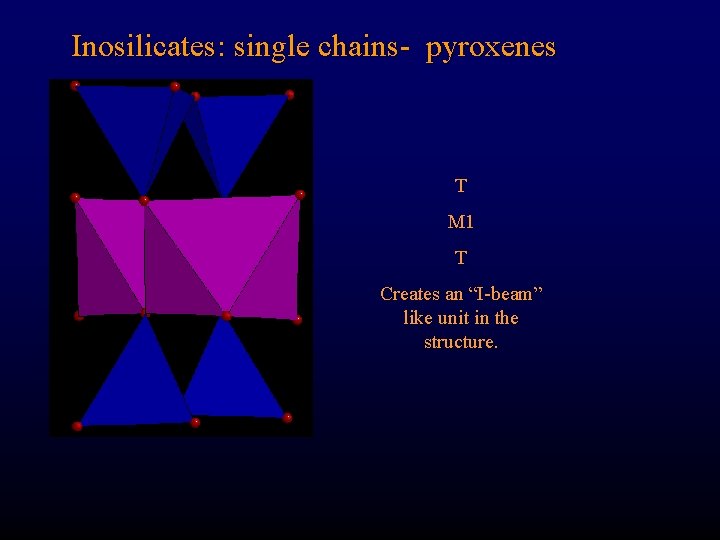
Inosilicates: single chains- pyroxenes T M 1 T Creates an “I-beam” like unit in the structure.
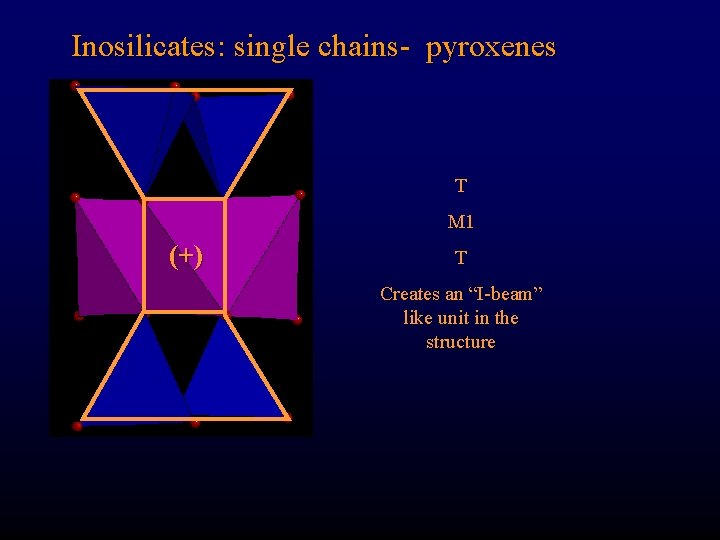
Inosilicates: single chains- pyroxenes T M 1 (+) T Creates an “I-beam” like unit in the structure
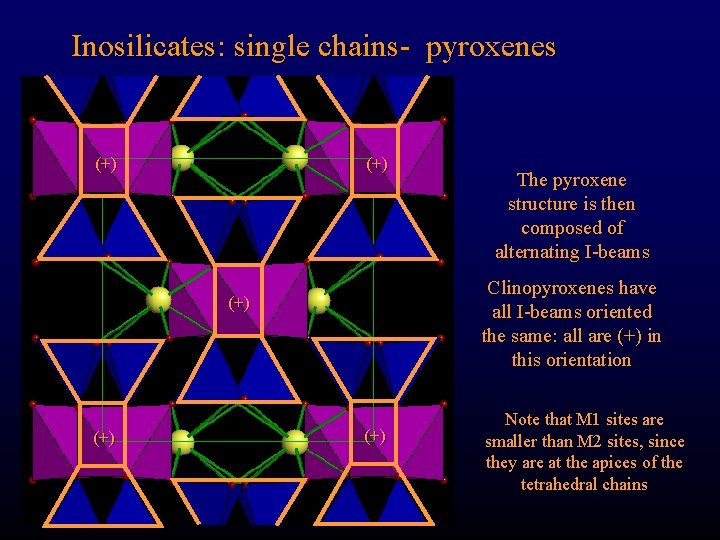
Inosilicates: single chains- pyroxenes (+) Clinopyroxenes have all I-beams oriented the same: all are (+) in this orientation (+) The pyroxene structure is then composed of alternating I-beams (+) Note that M 1 sites are smaller than M 2 sites, since they are at the apices of the tetrahedral chains
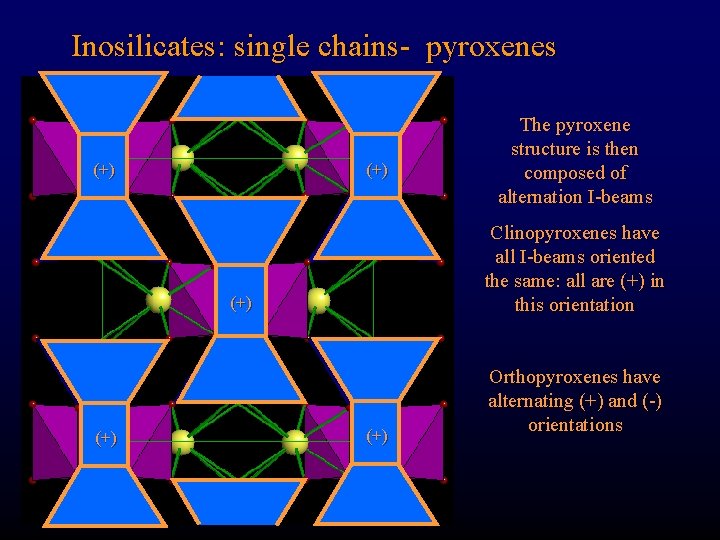
Inosilicates: single chains- pyroxenes (+) Clinopyroxenes have all I-beams oriented the same: all are (+) in this orientation (+) The pyroxene structure is then composed of alternation I-beams (+) Orthopyroxenes have alternating (+) and (-) orientations
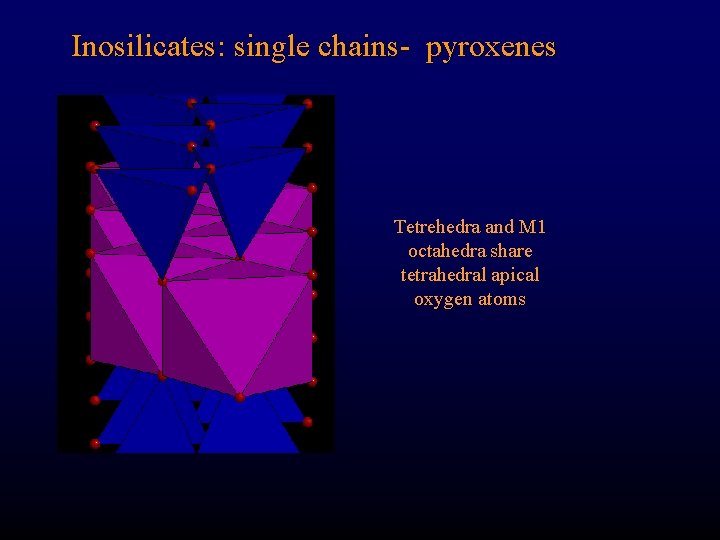
Inosilicates: single chains- pyroxenes Tetrehedra and M 1 octahedra share tetrahedral apical oxygen atoms
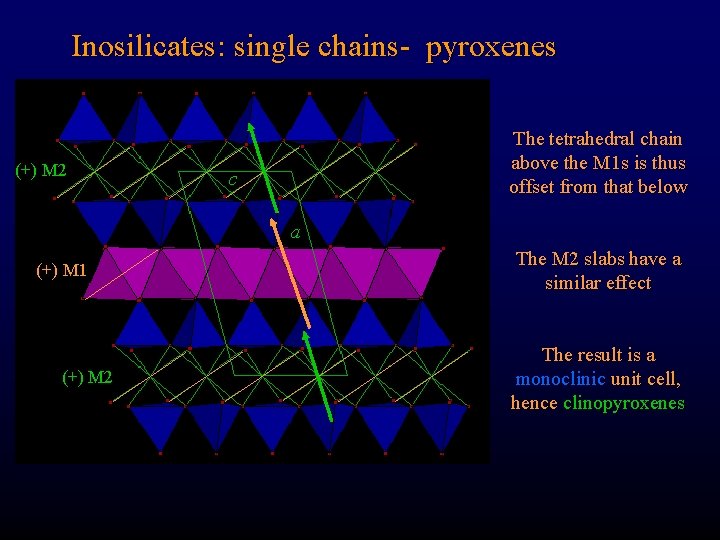
Inosilicates: single chains- pyroxenes (+) M 2 The tetrahedral chain above the M 1 s is thus offset from that below c a (+) M 1 (+) M 2 The M 2 slabs have a similar effect The result is a monoclinic unit cell, hence clinopyroxenes
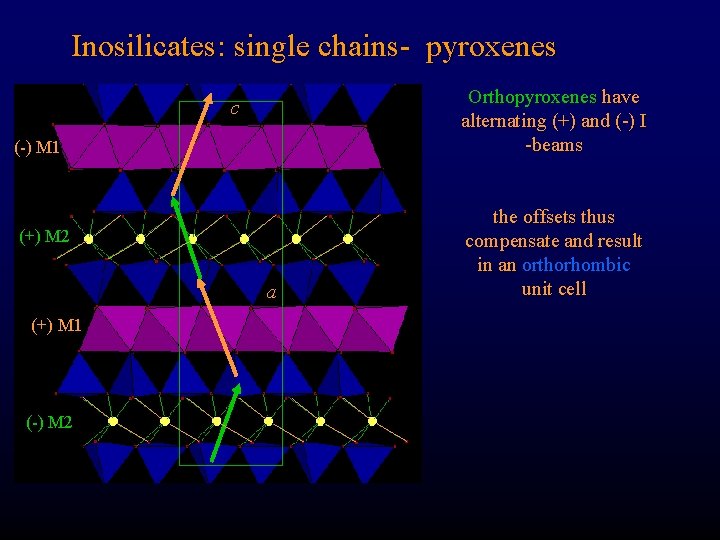
Inosilicates: single chains- pyroxenes Orthopyroxenes have alternating (+) and (-) I -beams c (-) M 1 (+) M 2 a (+) M 1 (-) M 2 the offsets thus compensate and result in an orthorhombic unit cell

Pyroxene Chemistry The general pyroxene formula: W 1 -P (X, Y)1+P Z 2 O 6 Where F W = Ca Na F X = Mg Fe 2+ Mn Ni Li F Y = Al Fe 3+ Cr Ti F Z = Si Al Anhydrous so high-temperature or dry conditions favor pyroxenes over amphiboles
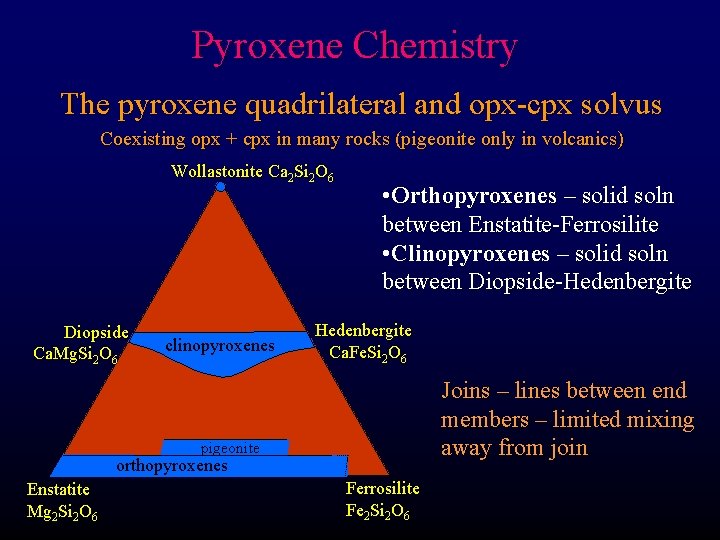
Pyroxene Chemistry The pyroxene quadrilateral and opx-cpx solvus Coexisting opx + cpx in many rocks (pigeonite only in volcanics) Wollastonite Ca 2 Si 2 O 6 Diopside Ca. Mg. Si 2 O 6 clinopyroxenes • Orthopyroxenes – solid soln between Enstatite-Ferrosilite • Clinopyroxenes – solid soln between Diopside-Hedenbergite Ca. Fe. Si 2 O 6 Joins – lines between end members – limited mixing away from join pigeonite orthopyroxenes Enstatite Mg 2 Si 2 O 6 Ferrosilite Fe 2 Si 2 O 6
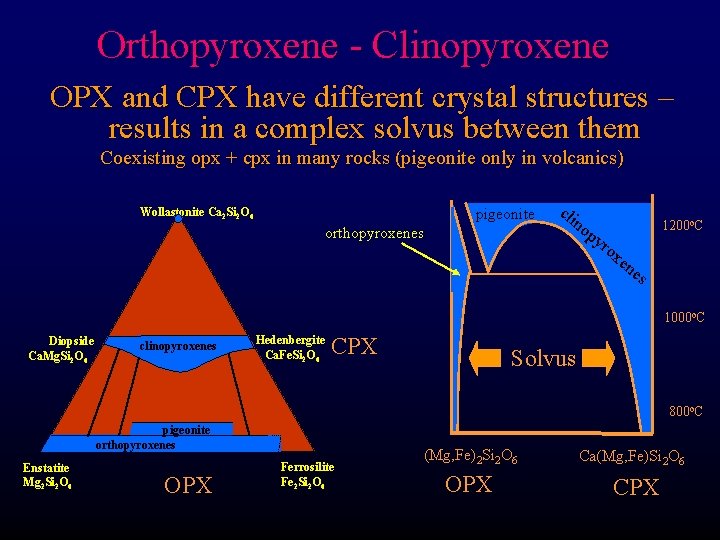
Orthopyroxene - Clinopyroxene OPX and CPX have different crystal structures – results in a complex solvus between them Coexisting opx + cpx in many rocks (pigeonite only in volcanics) pigeonite Wollastonite Ca 2 Si 2 O 6 orthopyroxenes cli no py ro 1200 o. C xe ne s 1000 o. C Diopside Ca. Mg. Si 2 O 6 clinopyroxenes Hedenbergite Ca. Fe. Si 2 O 6 CPX Solvus 800 o. C pigeonite orthopyroxenes Enstatite Mg 2 Si 2 O 6 OPX Ferrosilite Fe 2 Si 2 O 6 (Mg, Fe)2 Si 2 O 6 Ca(Mg, Fe)Si 2 O 6 OPX CPX
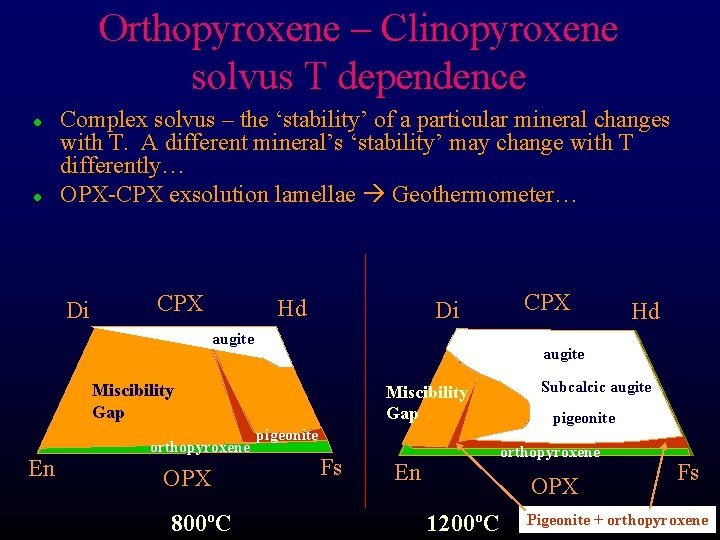
Orthopyroxene – Clinopyroxene solvus T dependence l l Complex solvus – the ‘stability’ of a particular mineral changes with T. A different mineral’s ‘stability’ may change with T differently… OPX-CPX exsolution lamellae Geothermometer… Di CPX Hd CPX Di augite Miscibility Gap En orthopyroxene OPX 800ºC Hd Subcalcic augite Miscibility Gap pigeonite Fs En pigeonite orthopyroxene OPX 1200ºC Fs Pigeonite + orthopyroxene
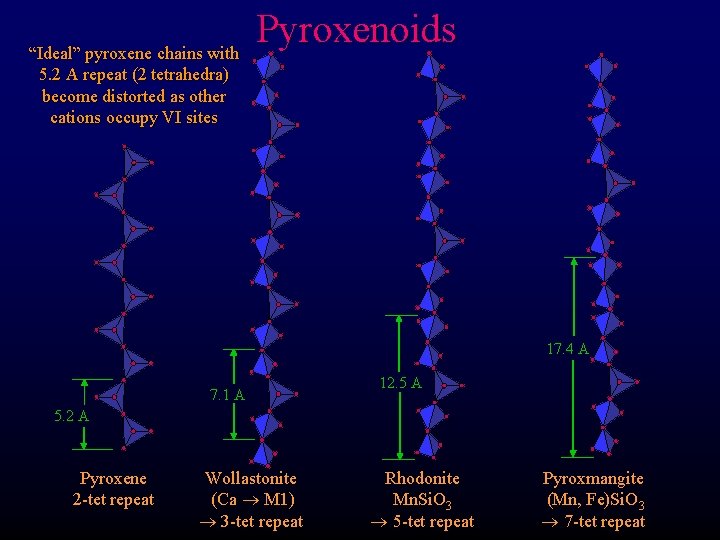
“Ideal” pyroxene chains with 5. 2 A repeat (2 tetrahedra) become distorted as other cations occupy VI sites Pyroxenoids 17. 4 A 7. 1 A 12. 5 A 5. 2 A Pyroxene 2 -tet repeat Wollastonite (Ca M 1) 3 -tet repeat Rhodonite Mn. Si. O 3 5 -tet repeat Pyroxmangite (Mn, Fe)Si. O 3 7 -tet repeat
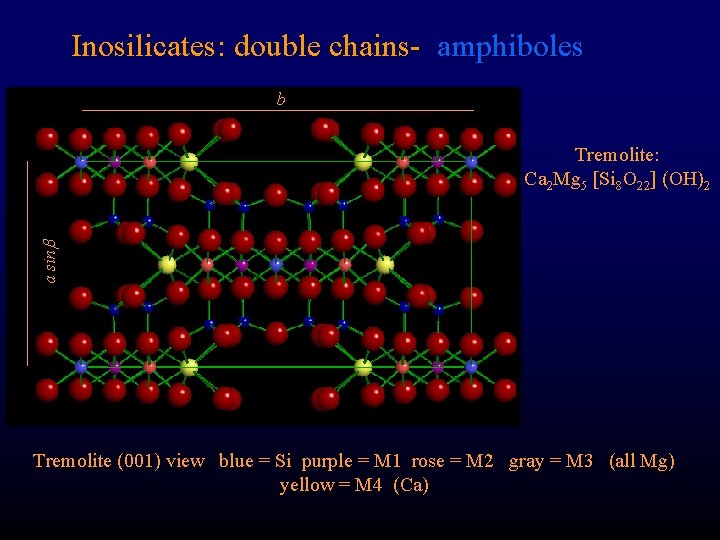
Inosilicates: double chains- amphiboles b a sin Tremolite: Ca 2 Mg 5 [Si 8 O 22] (OH)2 Tremolite (001) view blue = Si purple = M 1 rose = M 2 gray = M 3 (all Mg) yellow = M 4 (Ca)

Inosilicates: double chains- amphiboles b a sin Hornblende: (Ca, Na)2 -3 (Mg, Fe, Al)5 [(Si, Al)8 O 22] (OH)2 Hornblende (001) view dark blue = Si, Al purple = M 1 rose = M 2 light blue = M 3 (all Mg, Fe) yellow ball = M 4 (Ca) purple ball = A (Na) little turquoise ball = H

Inosilicates: double chains- amphiboles Hornblende: (Ca, Na)2 -3 (Mg, Fe, Al)5 [(Si, Al)8 O 22] (OH)2 Same I-beam architecture, but the I-beams are fatter (double chains) Hornblende (001) view dark blue = Si, Al purple = M 1 rose = M 2 light blue = M 3 (all Mg, Fe)

Inosilicates: double chains- amphiboles b (+) a sin (+) (+) Hornblende: (Ca, Na)2 -3 (Mg, Fe, Al)5 [(Si, Al)8 O 22] (OH)2 Same I-beam architecture, but the I-beams are fatter (double chains) All are (+) on clinoamphiboles and alternate in orthoamphiboles Hornblende (001) view dark blue = Si, Al purple = M 1 rose = M 2 light blue = M 3 (all Mg, Fe) yellow ball = M 4 (Ca) purple ball = A (Na) little turquoise ball = H

Inosilicates: double chains- amphiboles Hornblende: (Ca, Na)2 -3 (Mg, Fe, Al)5 [(Si, Al)8 O 22] (OH)2 M 1 -M 3 are small sites M 4 is larger (Ca) A-site is really big Variety of sites great chemical range Hornblende (001) view dark blue = Si, Al purple = M 1 rose = M 2 light blue = M 3 (all Mg, Fe) yellow ball = M 4 (Ca) purple ball = A (Na) little turquoise ball = H

Inosilicates: double chains- amphiboles Hornblende: (Ca, Na)2 -3 (Mg, Fe, Al)5 [(Si, Al)8 O 22] (OH)2 (OH) is in center of tetrahedral ring where O is a part of M 1 and M 3 octahedra (OH) Hornblende (001) view dark blue = Si, Al purple = M 1 rose = M 2 light blue = M 3 (all Mg, Fe) yellow ball = M 4 (Ca) purple ball = A (Na) little turquoise ball = H

Amphibole Chemistry See handout for more information General formula: W 0 -1 X 2 Y 5 [Z 8 O 22] (OH, F, Cl)2 W = Na K X = Ca Na Mg Fe 2+ (Mn Li) Y = Mg Fe 2+ Mn Al Fe 3+ Ti Z = Si Al Again, the great variety of sites and sizes a great chemical range, and hence a broad stability range The hydrous nature implies an upper temperature stability limit

Amphibole Chemistry Ca-Mg-Fe Amphibole “quadrilateral” (good analogy with pyroxenes) Tremolite Ca 2 Mg 5 Si 8 O 22(OH)2 Anthophyllite Mg 7 Si 8 O 22(OH)2 Actinolite Cummingtonite-grunerite Orthoamphiboles Ferroactinolite Ca 2 Fe 5 Si 8 O 22(OH)2 Clinoamphiboles Fe 7 Si 8 O 22(OH)2 Al and Na tend to stabilize the orthorhombic form in low-Ca amphiboles, so anthophyllite gedrite orthorhombic series extends to Fe-rich gedrite in more Na-Al-rich compositions

Amphibole Chemistry Hornblende has Al in the tetrahedral site Geologists traditionally use the term “hornblende” as a catch-all term for practically any dark amphibole. Now the common use of the microprobe has petrologists casting “hornblende” into end-member compositions and naming amphiboles after a well-represented end-member. Sodic amphiboles Glaucophane: Na 2 Mg 3 Al 2 [Si 8 O 22] (OH)2 Riebeckite: Na 2 Fe 2+3 Fe 3+2 [Si 8 O 22] (OH)2 Sodic amphiboles are commonly blue, and often called “blue amphiboles”
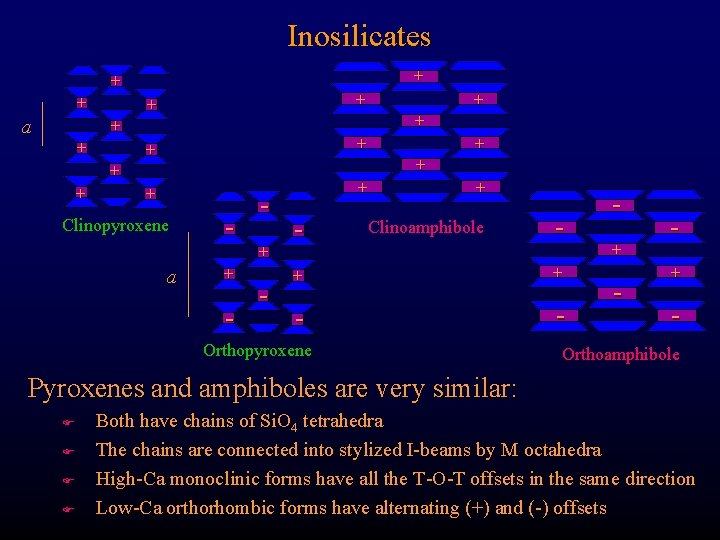
Inosilicates + + + a + + + + Clinopyroxene a + - - + Clinoamphibole - + + - - Orthopyroxene + - Orthoamphibole Pyroxenes and amphiboles are very similar: F F Both have chains of Si. O 4 tetrahedra The chains are connected into stylized I-beams by M octahedra High-Ca monoclinic forms have all the T-O-T offsets in the same direction Low-Ca orthorhombic forms have alternating (+) and (-) offsets
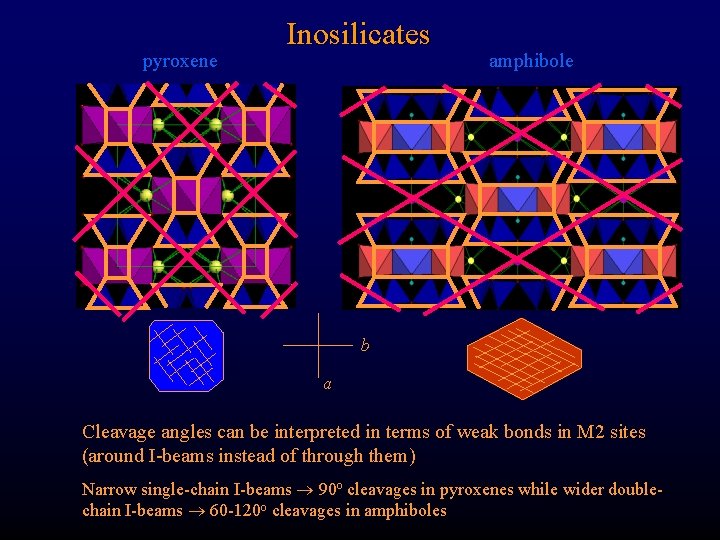
pyroxene Inosilicates amphibole b a Cleavage angles can be interpreted in terms of weak bonds in M 2 sites (around I-beams instead of through them) Narrow single-chain I-beams 90 o cleavages in pyroxenes while wider doublechain I-beams 60 -120 o cleavages in amphiboles
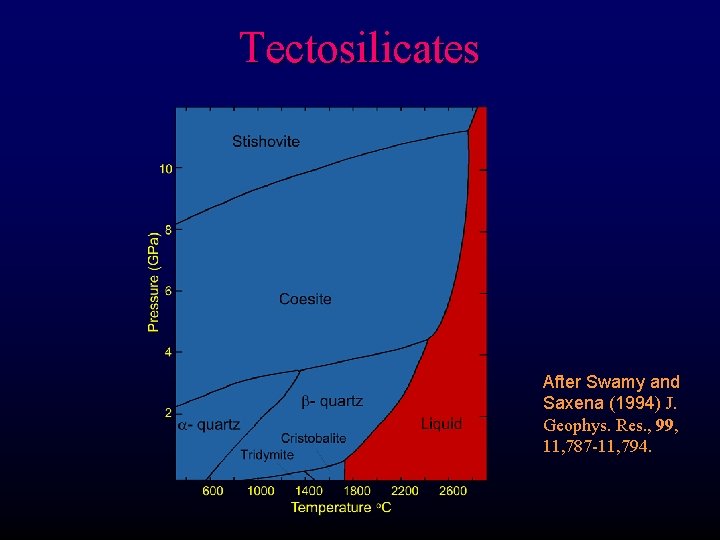
Tectosilicates After Swamy and Saxena (1994) J. Geophys. Res. , 99, 11, 787 -11, 794.

Tectosilicates Low Quartz 001 Projection Crystal Class 32

Tectosilicates High Quartz at 581 o. C 001 Projection Crystal Class 622

Tectosilicates Cristobalite 001 Projection Cubic Structure

Tectosilicates Stishovite High pressure Si. VI
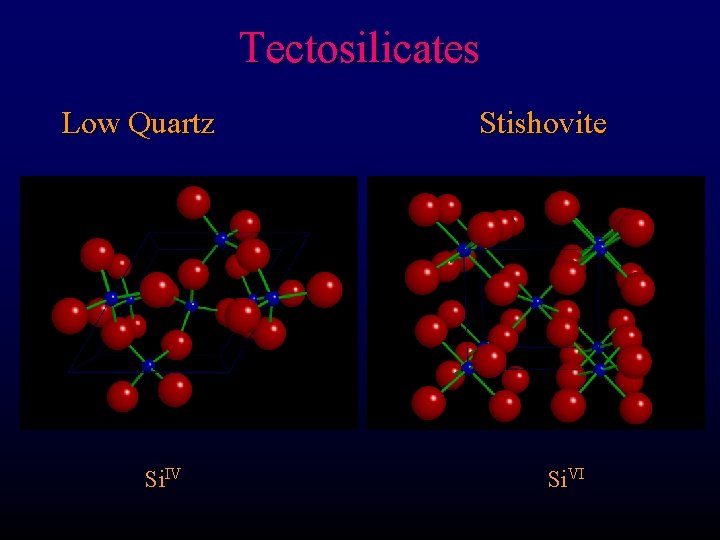
Tectosilicates Low Quartz Si. IV Stishovite Si. VI

Igneous Minerals l l Quartz, Feldspars (plagioclase and alkaline), Olivines, Pyroxenes, Amphiboles Accessory Minerals – mostly in small quantities or in ‘special’ rocks F F F F Magnetite (Fe 3 O 4) Ilmenite (Fe. Ti. O 3) Apatite (Ca 5(PO 4)3(OH, F, Cl) Zircon (Zr. Si. O 4) Sphene (a. k. a. Titanite) (Ca. Ti. Si. O 5) Pyrite (Fe. S 2) Fluorite (Ca. F 2)
- Slides: 55