Feedback on GCSE Science Double Award Chemistry GCSE

Feedback on GCSE Science (Double Award) – Chemistry
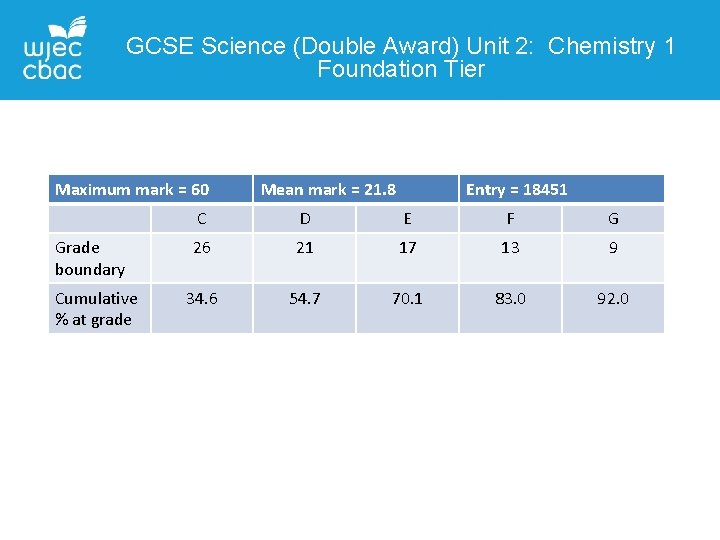
GCSE Science (Double Award) Unit 2: Chemistry 1 Foundation Tier Maximum mark = 60 Grade boundary Cumulative % at grade Mean mark = 21. 8 Entry = 18451 C D E F G 26 21 17 13 9 34. 6 54. 7 70. 1 83. 0 92. 0
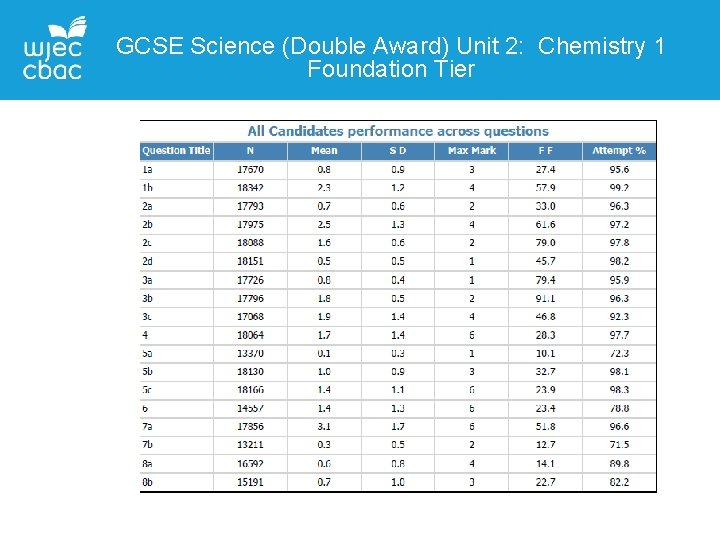
GCSE Science (Double Award) Unit 2: Chemistry 1 Foundation Tier
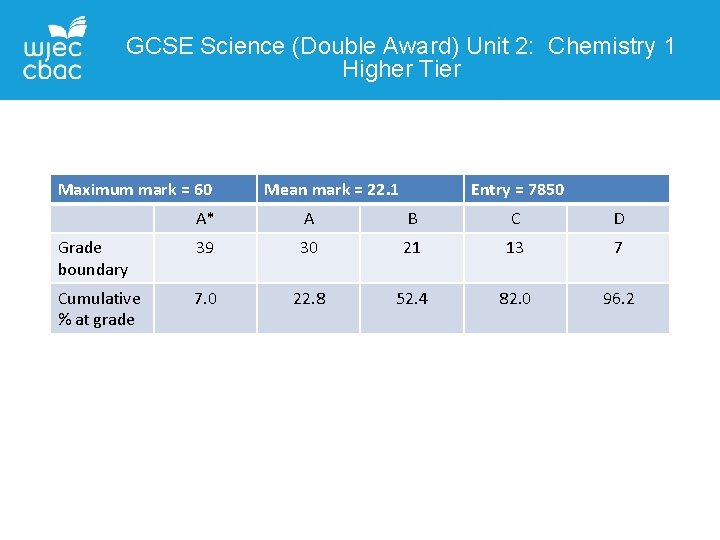
GCSE Science (Double Award) Unit 2: Chemistry 1 Higher Tier Maximum mark = 60 Mean mark = 22. 1 Entry = 7850 A* A B C D Grade boundary 39 30 21 13 7 Cumulative % at grade 7. 0 22. 8 52. 4 82. 0 96. 2
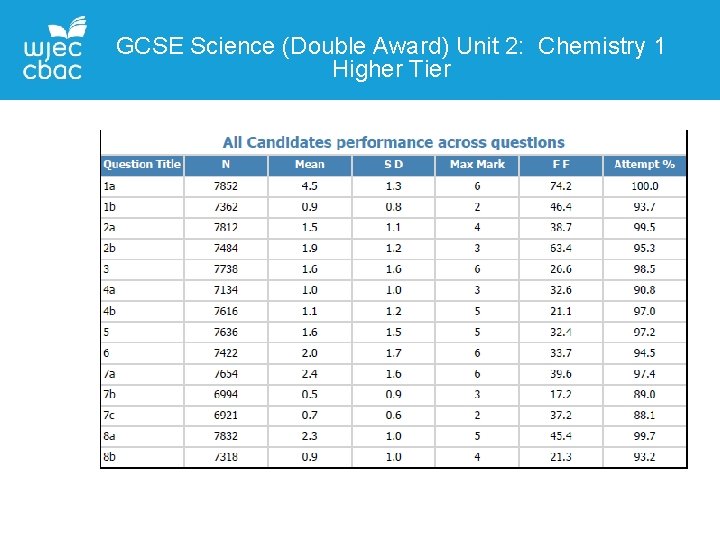
GCSE Science (Double Award) Unit 2: Chemistry 1 Higher Tier
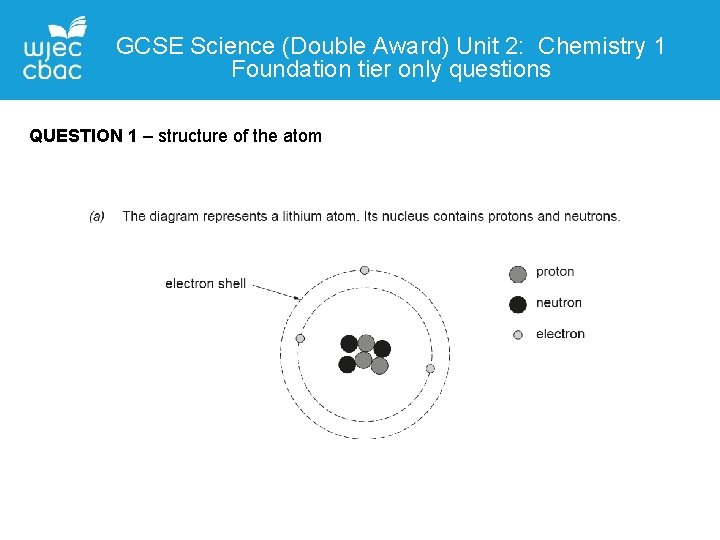
GCSE Science (Double Award) Unit 2: Chemistry 1 Foundation tier only questions QUESTION 1 – structure of the atom

GCSE Science (Double Award) Unit 2: Chemistry 1 Foundation tier only questions QUESTION 2 – fair testing
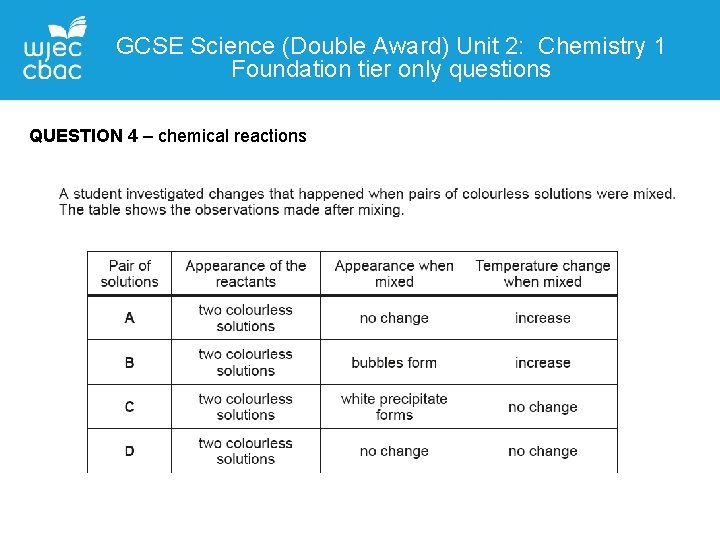
GCSE Science (Double Award) Unit 2: Chemistry 1 Foundation tier only questions QUESTION 4 – chemical reactions
GCSE Science (Double Award) Unit 2: Chemistry 1 Foundation tier only questions QUESTION 6 – carbon cycle
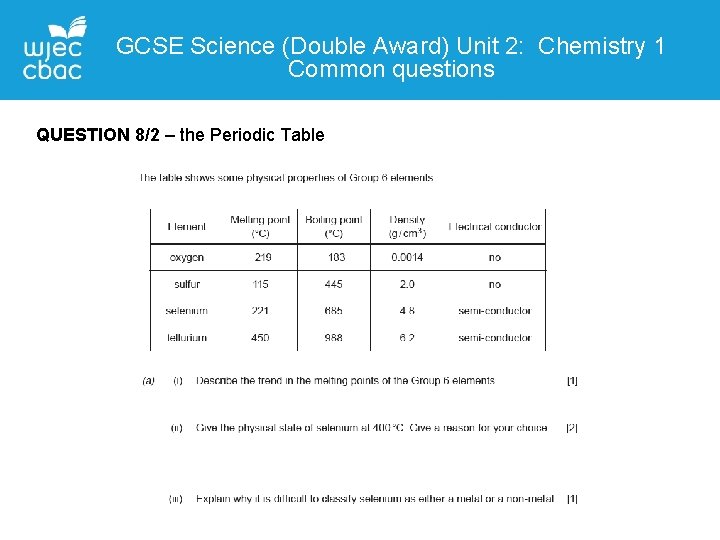
GCSE Science (Double Award) Unit 2: Chemistry 1 Common questions QUESTION 8/2 – the Periodic Table
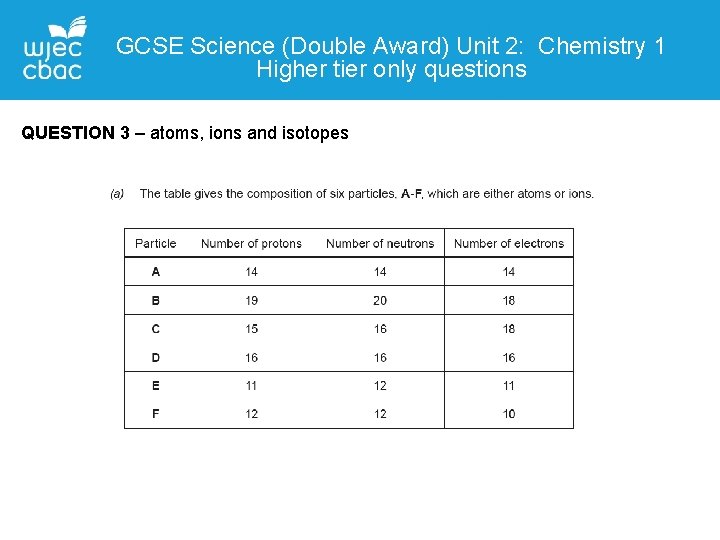
GCSE Science (Double Award) Unit 2: Chemistry 1 Higher tier only questions QUESTION 3 – atoms, ions and isotopes
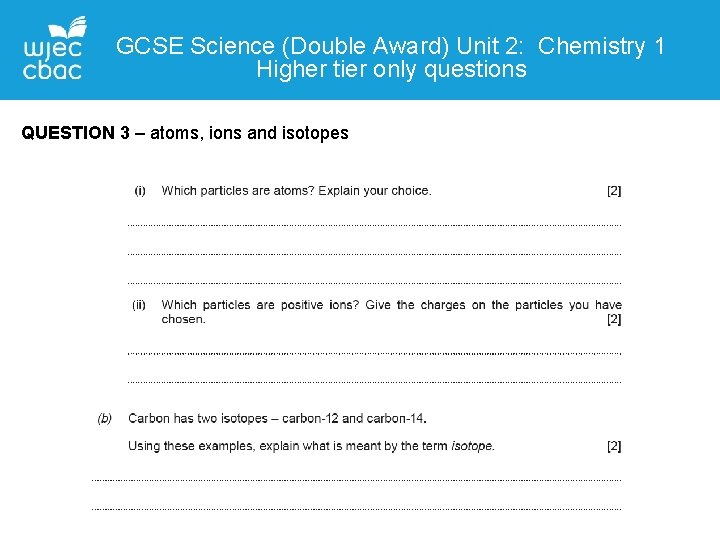
GCSE Science (Double Award) Unit 2: Chemistry 1 Higher tier only questions QUESTION 3 – atoms, ions and isotopes

GCSE Science (Double Award) Unit 2: Chemistry 1 Higher tier only questions QUESTION 4 – plate tectonics

GCSE Science (Double Award) Unit 2: Chemistry 1 Higher tier only questions QUESTION 8 – ‘simplest’ formula
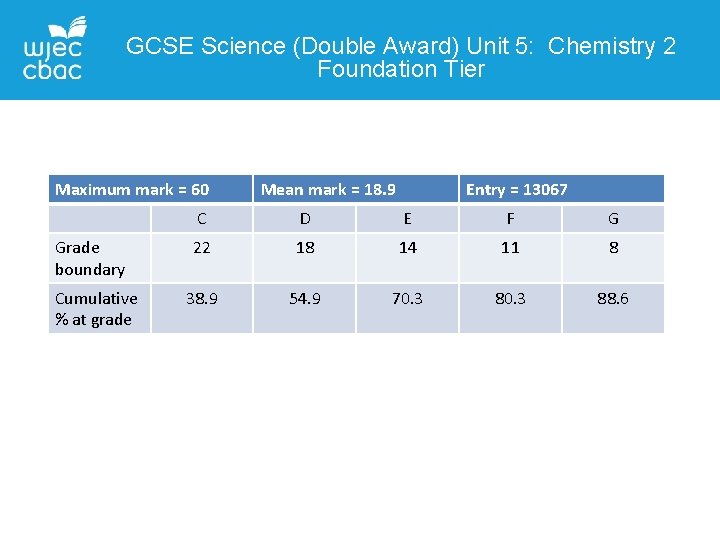
GCSE Science (Double Award) Unit 5: Chemistry 2 Foundation Tier Maximum mark = 60 Grade boundary Cumulative % at grade Mean mark = 18. 9 Entry = 13067 C D E F G 22 18 14 11 8 38. 9 54. 9 70. 3 88. 6
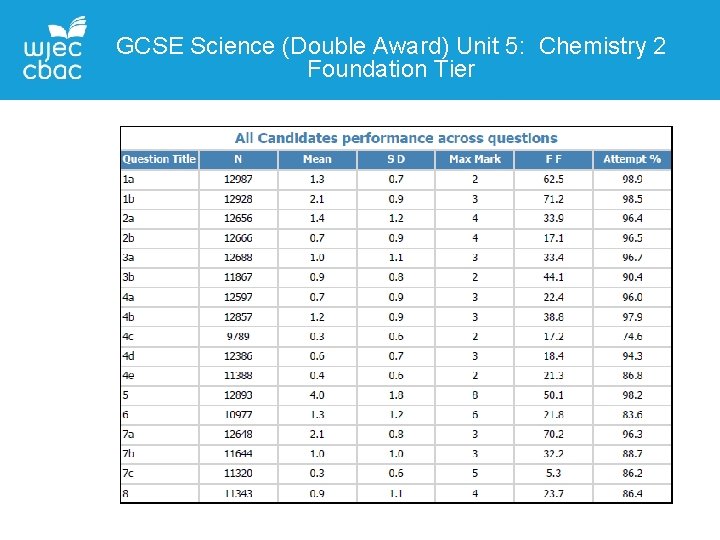
GCSE Science (Double Award) Unit 5: Chemistry 2 Foundation Tier
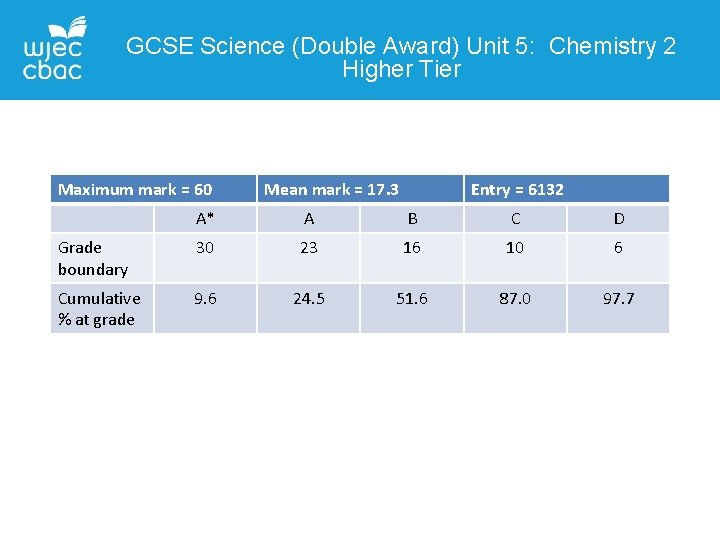
GCSE Science (Double Award) Unit 5: Chemistry 2 Higher Tier Maximum mark = 60 Mean mark = 17. 3 Entry = 6132 A* A B C D Grade boundary 30 23 16 10 6 Cumulative % at grade 9. 6 24. 5 51. 6 87. 0 97. 7
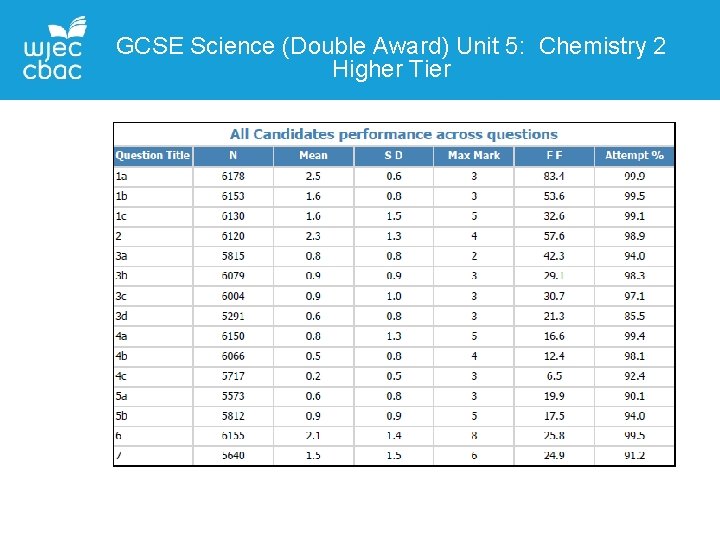
GCSE Science (Double Award) Unit 5: Chemistry 2 Higher Tier

GCSE Science (Double Award) Unit 5: Chemistry 2 Foundation tier only questions QUESTION 2 – chemical reactions
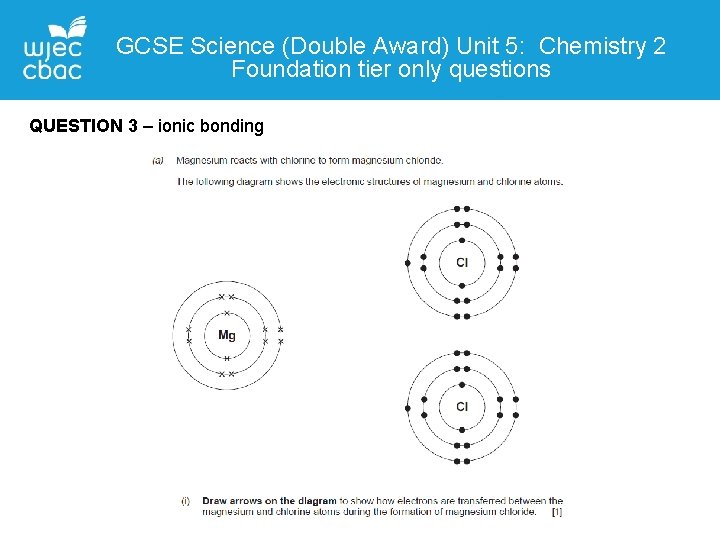
GCSE Science (Double Award) Unit 5: Chemistry 2 Foundation tier only questions QUESTION 3 – ionic bonding

GCSE Science (Double Award) Unit 5: Chemistry 2 Foundation tier only questions QUESTION 4 – crude oil

GCSE Science (Double Award) Unit 5: Chemistry 2 Foundation tier only questions QUESTION 4 – disposal of plastics
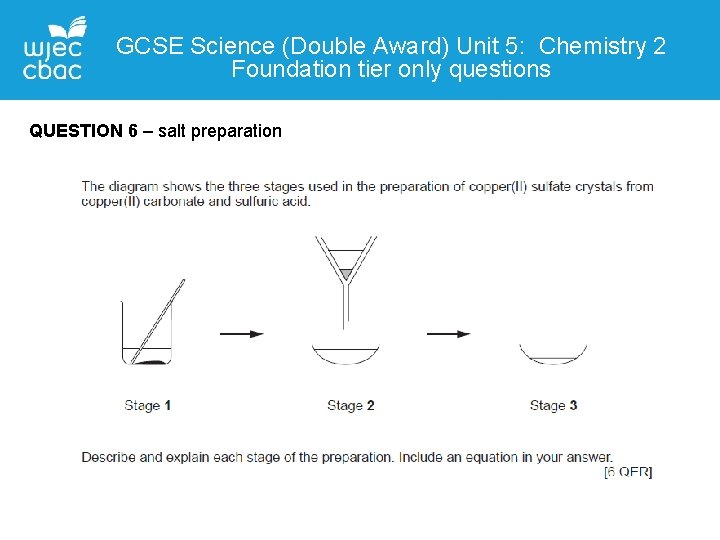
GCSE Science (Double Award) Unit 5: Chemistry 2 Foundation tier only questions QUESTION 6 – salt preparation
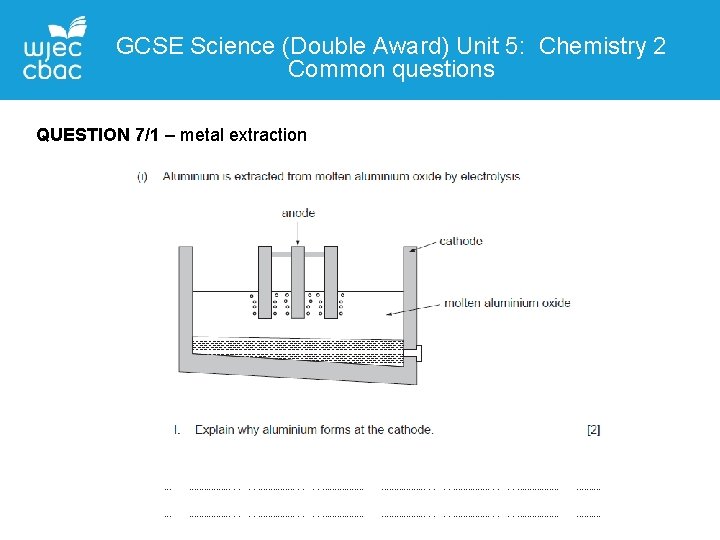
GCSE Science (Double Award) Unit 5: Chemistry 2 Common questions QUESTION 7/1 – metal extraction

GCSE Science (Double Award) Unit 5: Chemistry 2 Higher tier only questions QUESTION 4 – flame tests and testing for ions

GCSE Science (Double Award) Unit 5: Chemistry 2 Higher tier only questions QUESTION 7 – ionic bonding and structure

Teaching & Learning Chemical calculations • Minimum of 20% of marks linked to mathematical skills • Percentage of ‘calculations’ may be higher … not all marks can be counted as mathematical marks • Other than energy changes … specified in the first unit (year 10) • Double Award papers for 2018 – 10 -13 marks for calculations • Look to build re-cap of these skills into year 11 units

Teaching & Learning Chemical calculations Acids, bases and salts Cu. O powder added to a volume of HCl in a beaker Cu. O + 2 HCl → Cu. Cl 2 + H 2 O Solution contains 0. 124 mol of HCl (Double Award specification has no n=c. V) Calculate mass of Cu. O required to neutralise the acid

Teaching & Learning Chemical calculations Metals and their extraction Galena containing Pb. S mined in Gwydyr Forest, Halkyn Mountain, Clyne valley Ore roasted to make Pb. O reduced in reaction with coke 2 Pb. O + C → 2 Pb + CO 2 2500 kg of Pb. O Calculate how much Pb you would expect to form Calculate the minimum amount of carbon (coke) required to produce this amount

Teaching & Learning Chemical calculations Crude oil, fuels and carbon compounds Cracking reaction e. g. C 15 H 32 → 2 C 2 H 4 + C 3 H 6 + C 8 H 18 Calculate mass of ethene / propene / octane formed from 530 kg of C 15 H 32

Any Questions?
- Slides: 31