Feedback on GCSE Chemistry GCSE Chemistry Unit 1

Feedback on GCSE Chemistry
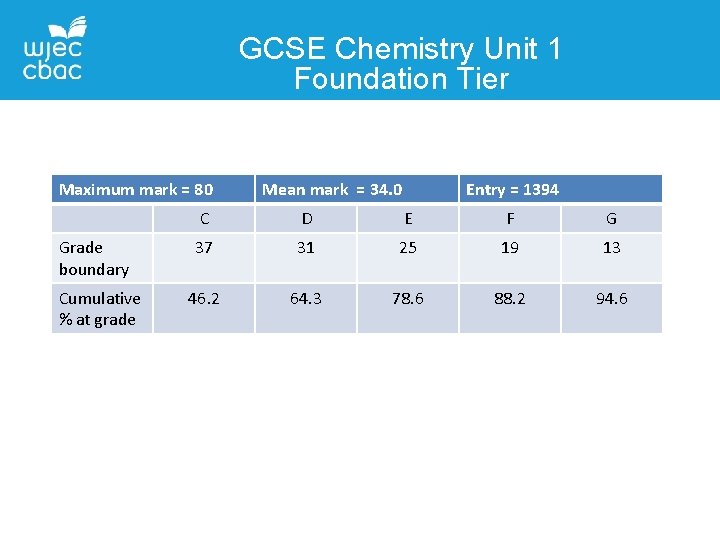
GCSE Chemistry Unit 1 Foundation Tier Maximum mark = 80 Grade boundary Cumulative % at grade Mean mark = 34. 0 Entry = 1394 C D E F G 37 31 25 19 13 46. 2 64. 3 78. 6 88. 2 94. 6
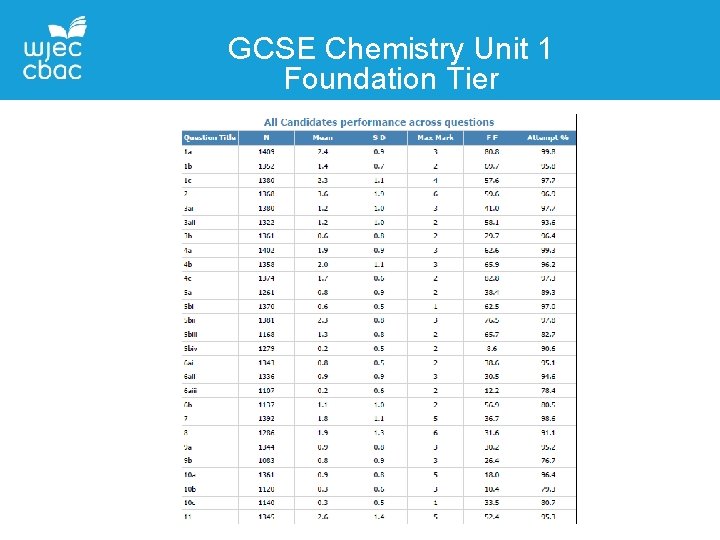
GCSE Chemistry Unit 1 Foundation Tier
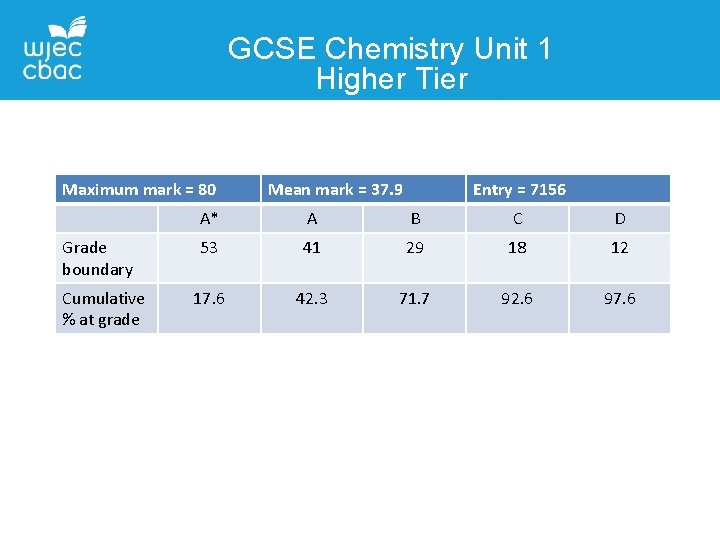
GCSE Chemistry Unit 1 Higher Tier Maximum mark = 80 Grade boundary Cumulative % at grade Mean mark = 37. 9 Entry = 7156 A* A B C D 53 41 29 18 12 17. 6 42. 3 71. 7 92. 6 97. 6
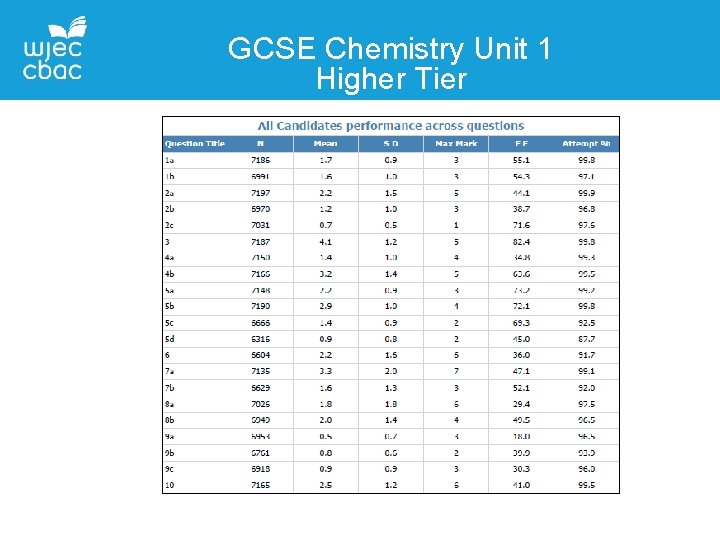
GCSE Chemistry Unit 1 Higher Tier
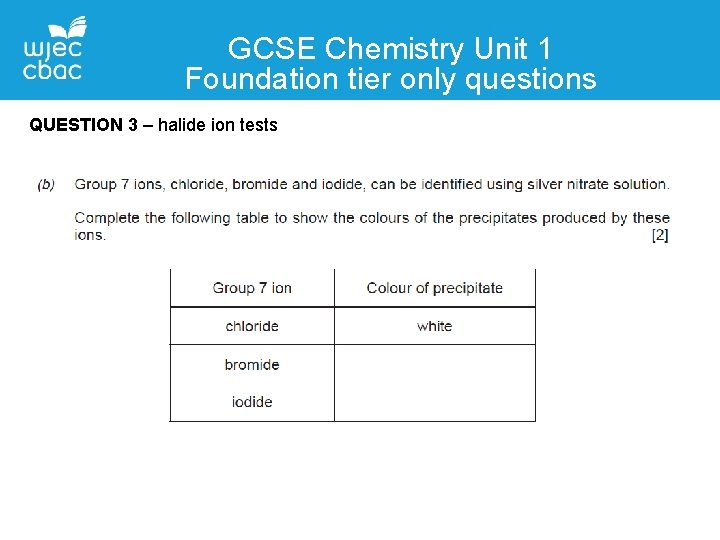
GCSE Chemistry Unit 1 Foundation tier only questions QUESTION 3 – halide ion tests

GCSE Chemistry Unit 1 Foundation tier only questions QUESTION 5 – refining practical design

GCSE Chemistry Unit 1 Foundation tier only questions QUESTION 6 – decomposition of carbonates
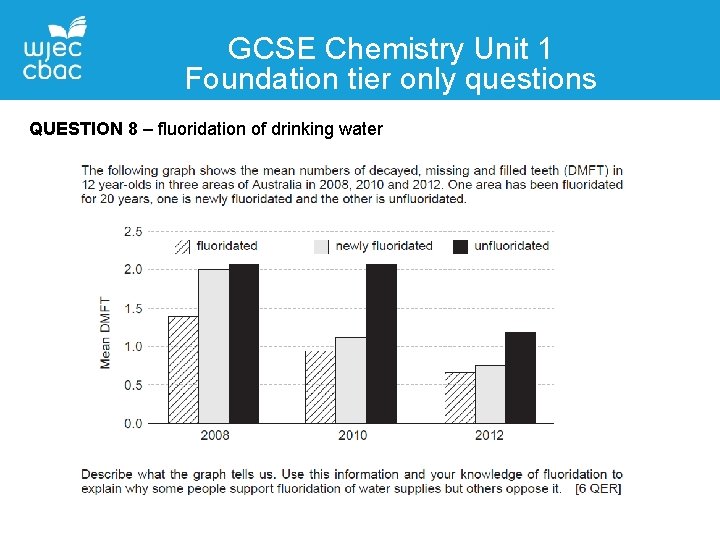
GCSE Chemistry Unit 1 Foundation tier only questions QUESTION 8 – fluoridation of drinking water

GCSE Chemistry Unit 1 Common questions QUESTION 9 – hard water

GCSE Chemistry Unit 1 Common questions QUESTION 10 – solubility curves

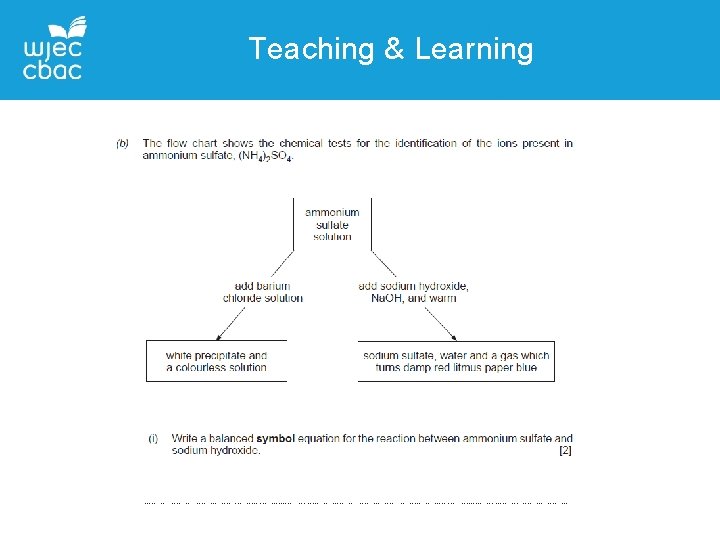
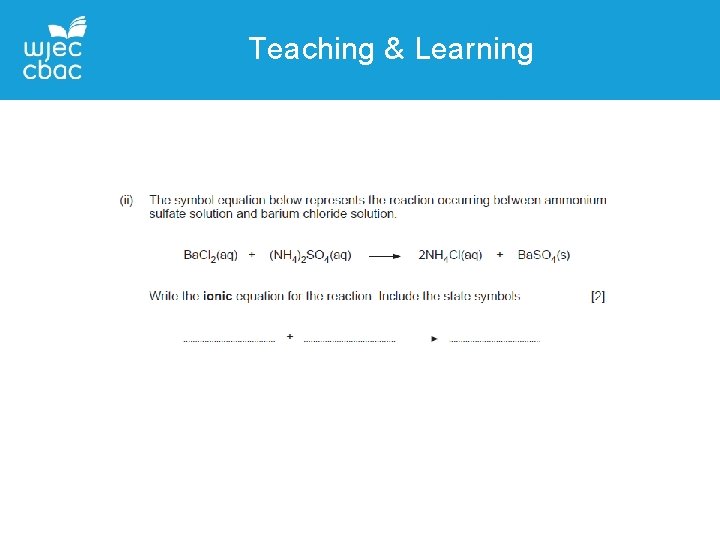
GCSE Chemistry Unit 1 Common questions QUESTION 10 – ions

GCSE Chemistry Unit 1 Higher tier only questions QUESTION 5 – noble gases

GCSE Chemistry Unit 1 Higher tier only questions QUESTION 6 – limestone cycle

GCSE Chemistry Unit 1 Higher tier only questions QUESTION 8 – reacting masses and percentage yield
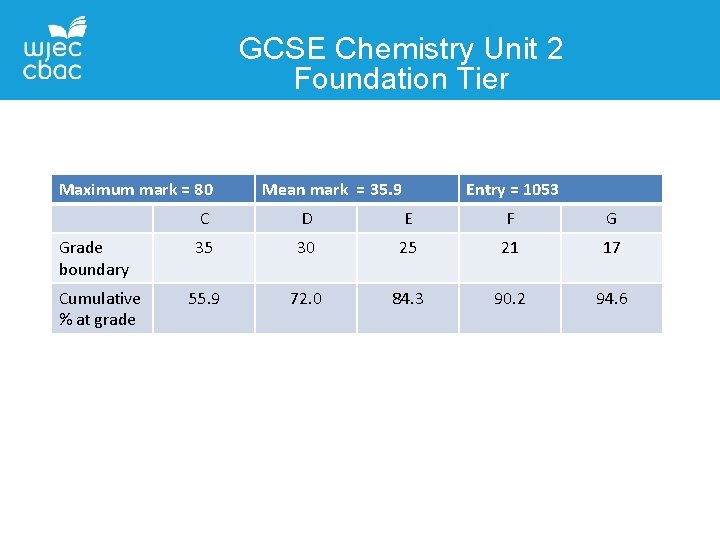
GCSE Chemistry Unit 2 Foundation Tier Maximum mark = 80 Grade boundary Cumulative % at grade Mean mark = 35. 9 Entry = 1053 C D E F G 35 30 25 21 17 55. 9 72. 0 84. 3 90. 2 94. 6
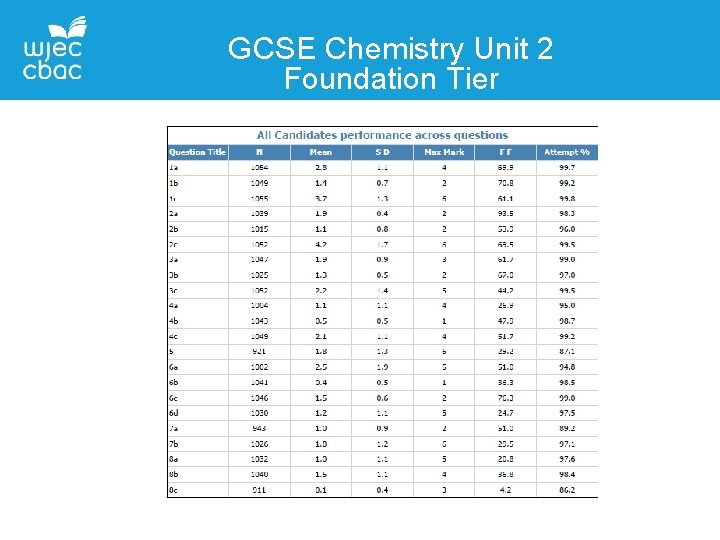
GCSE Chemistry Unit 2 Foundation Tier
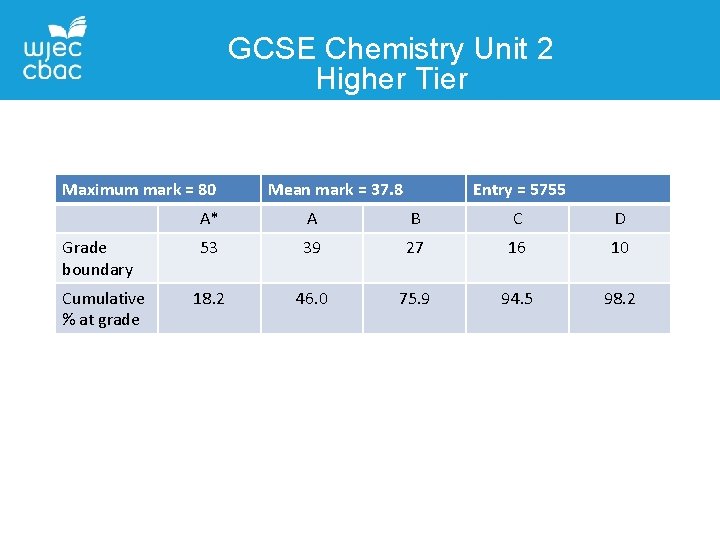
GCSE Chemistry Unit 2 Higher Tier Maximum mark = 80 Grade boundary Cumulative % at grade Mean mark = 37. 8 Entry = 5755 A* A B C D 53 39 27 16 10 18. 2 46. 0 75. 9 94. 5 98. 2
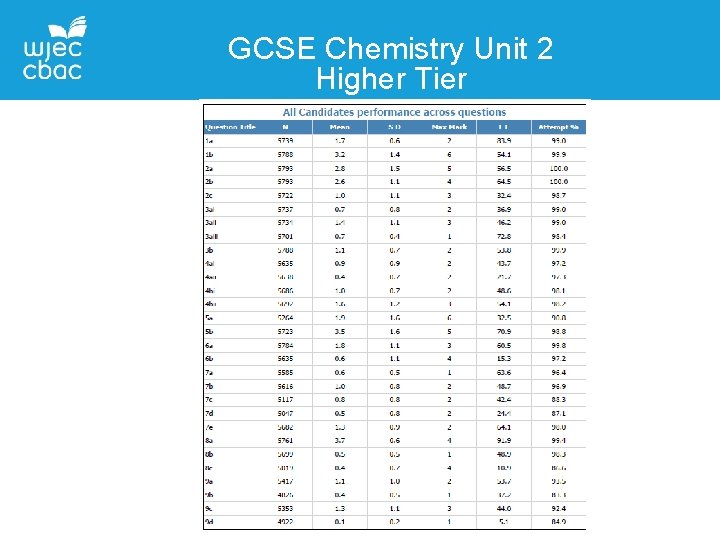
GCSE Chemistry Unit 2 Higher Tier
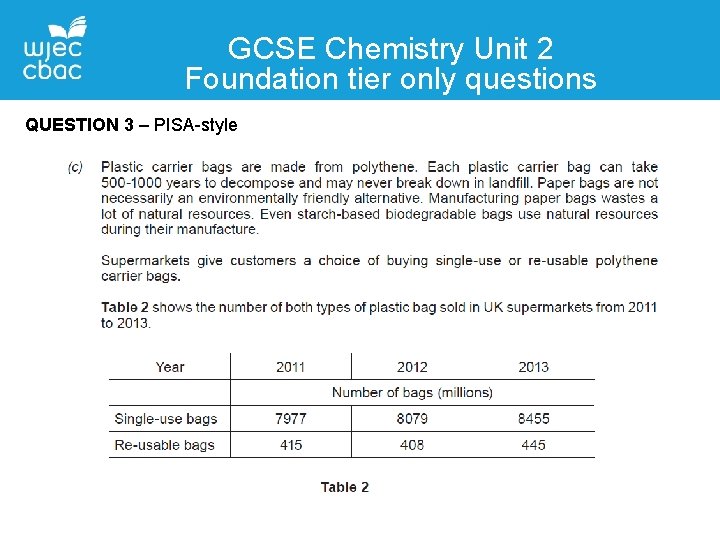
GCSE Chemistry Unit 2 Foundation tier only questions QUESTION 3 – PISA-style
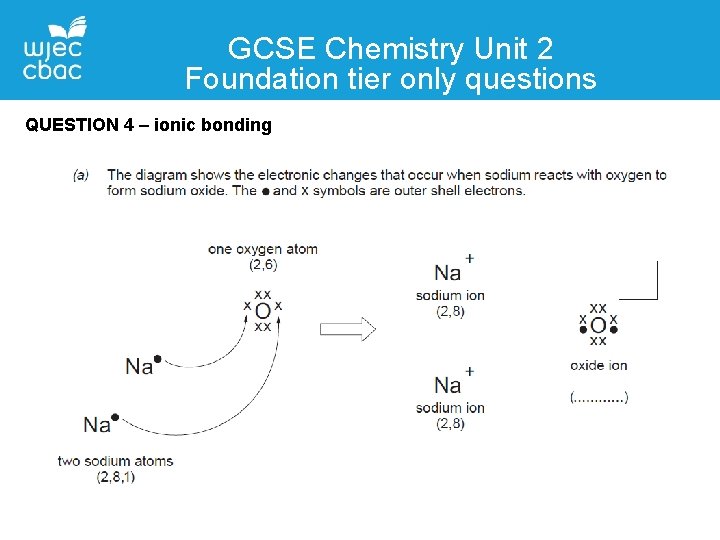
GCSE Chemistry Unit 2 Foundation tier only questions QUESTION 4 – ionic bonding

GCSE Chemistry Unit 2 Foundation tier only questions QUESTION 5 – fire triangle

GCSE Chemistry Unit 2 Common questions QUESTION 7 – neutralisation reaction

GCSE Chemistry Unit 2 Higher tier only questions QUESTION 4 – ionic bonding and structure

GCSE Chemistry Unit 2 Higher tier only questions QUESTION 5 – Contact Process

GCSE Chemistry Unit 2 Higher tier only questions QUESTION 7 – test for alcohols

GCSE Chemistry Unit 2 Higher tier only questions QUESTION 9 – evaluation of evidence

Teaching & Learning Chemical formulae and equations Study all four papers carefully – what is the total number of marks awarded formulae and equations on each one?

Teaching & Learning Chemical formulae and equations • Keep practising / testing skills English/Welsh – spelling and grammar tests Mathematics – tables tests • Introduce formulae (and word equations) in KS 3 • Read questions carefully – much of the required information is often given in questions

Teaching & Learning

Teaching & Learning

Teaching & Learning
Teaching & Learning
Teaching & Learning

Teaching & Learning Ionic bonding • Most easily shown using diagrams • Before and after – atoms and ions • Electrons should not appear in two places at once
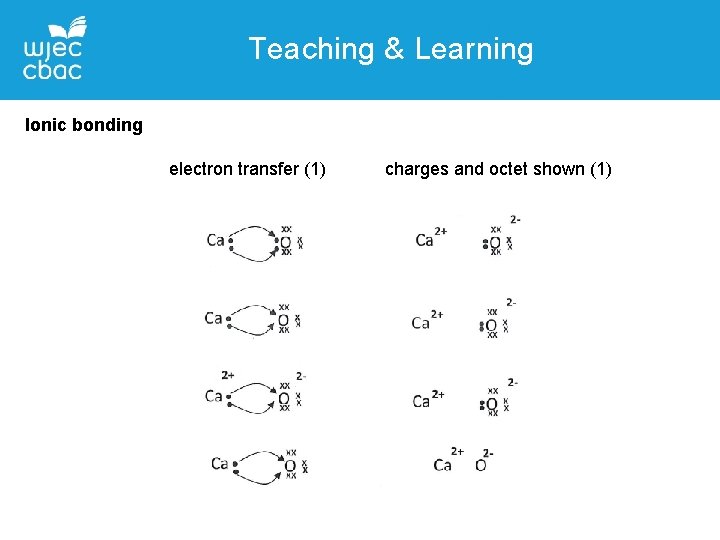
Teaching & Learning Ionic bonding electron transfer (1) charges and octet shown (1)

Any Questions?
- Slides: 37