Featured Article Efficacy and Safety of Dulaglutide Added





- Slides: 10

Featured Article: Efficacy and Safety of Dulaglutide Added Onto Pioglitazone and Metformin Versus Exenatide in Type 2 Diabetes in a Randomized Controlled Trial (AWARD-1) Carol Wysham, Thomas Blevins, Richard Arakaki, Gildred Colon, Pedro Garcia, Charles Atisso, Debra Kuhstoss, and Mark Lakshmanan Diabetes Care Volume 37: 2159 -2167 August, 2014

STUDY OBJECTIVE • To compare the efficacy and safety of dulaglutide with placebo and exenatide in type 2 diabetic patients • To determine superiority of dulaglutide 1. 5 mg versus placebo in Hb. A 1 c change at 26 weeks Wysham C. et al. Diabetes Care 2014; 37: 2159 -2167

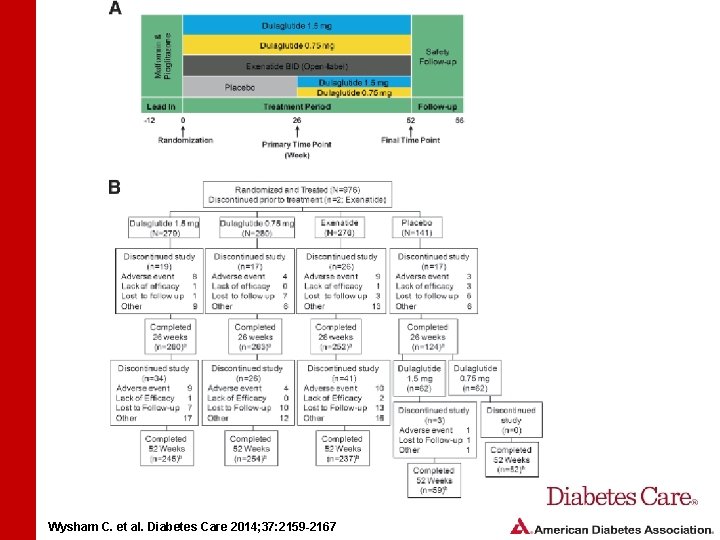
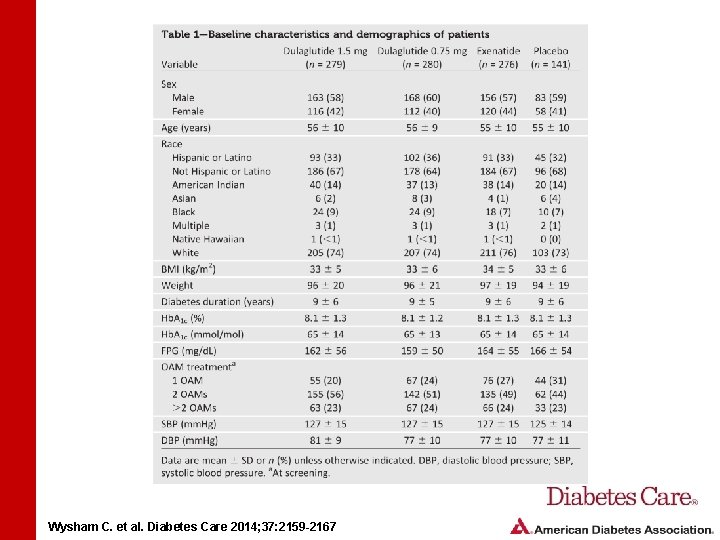
STUDY DESIGN AND METHODS • Study randomized patients to dulaglutide 1. 5 mg, dulaglutide 0. 75 mg, exenatide 10 mg, or placebo • Patients were treated with metformin (1, 500– 3, 000 mg) and pioglitazone (30– 45 mg) • Mean baseline Hb. A 1 c was 8. 1% Wysham C. et al. Diabetes Care 2014; 37: 2159 -2167

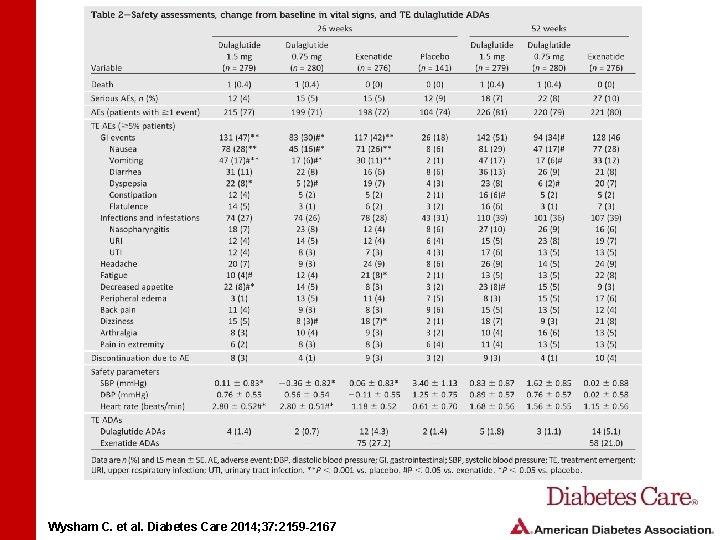
Wysham C. et al. Diabetes Care 2014; 37: 2159 -2167

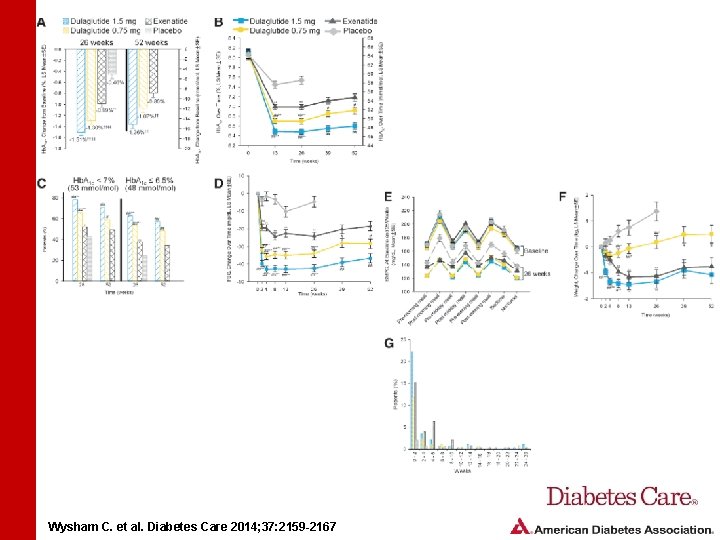
RESULTS • Least squares mean ± SE Hb. A 1 c change from baseline to primary end point was: • • • Both dulaglutide doses were superior to placebo at 26 weeks and exenatide at 26 and 52 weeks • Greater percentages of patients reached Hb. A 1 c targets with dulaglutide 1. 5 mg and 0. 75 mg than with placebo and exenatide 21. 51 ± 0. 06% for dulaglutide 1. 5 mg 21. 30 ± 0. 06% for dulaglutide 0. 75 mg 20. 99 ± 0. 06% for exenatide 20. 46 ± 0. 08% for placebo Wysham C. et al. Diabetes Care 2014; 37: 2159 -2167

RESULTS • At 26 and 52 weeks, total hypoglycemia incidence was lower in patients receiving dulaglutide 1. 5 mg than in those receiving exenatide • No dulaglutide-treated patients reported severe hypoglycemia • Most common gastrointestinal adverse events for dulaglutide were nausea, vomiting, and diarrhea • Events were mostly mild to moderate and transient Wysham C. et al. Diabetes Care 2014; 37: 2159 -2167

Wysham C. et al. Diabetes Care 2014; 37: 2159 -2167

Wysham C. et al. Diabetes Care 2014; 37: 2159 -2167

Wysham C. et al. Diabetes Care 2014; 37: 2159 -2167

CONCLUSIONS • Both once-weekly dulaglutide doses demonstrated superior glycemic control versus placebo and exenatide with an acceptable tolerability and safety profile Wysham C. et al. Diabetes Care 2014; 37: 2159 -2167