FDA Industry Workshop September 16 2005 Medical Device

FDA Industry Workshop September 16, 2005 Medical Device versus Drug Similarities and Differences Jeng Mah, David Breiter Guidant Corporation © Guidant 2004

Workshop 2005 Device vs. Drug - physical Device 2 Drug © Guidant 2004
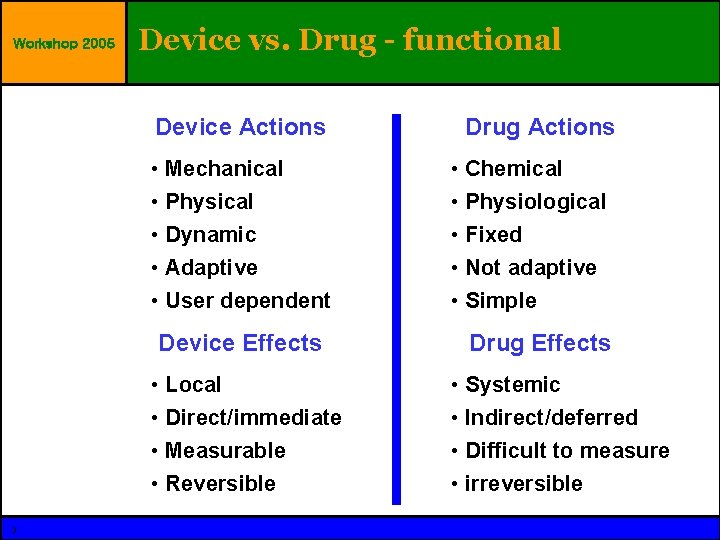
Workshop 2005 Device vs. Drug - functional Device Actions • Mechanical • Physical • Dynamic • Adaptive • User dependent Device Effects • Local • Direct/immediate • Measurable • Reversible 3 Drug Actions • Chemical • Physiological • Fixed • Not adaptive • Simple Drug Effects • Systemic • Indirect/deferred • Difficult to measure • irreversible © Guidant 2004

Workshop 2005 Device vs. Drug - statistical Device Studies • May perform multiple adaptive functions (bundled features) • Extensive & informative bench and acute tests • Non-blinded, nonplacebo pivotal studies • Automation & decision making • Subject specific optimal programming • User interface/skill affects/determines outcomes • Real time data collection generates lots of data 4 Drug Studies • One drug one desired effect (usually), or deal with drug interactions • Phase I, II studies serve different purposes • Active control, blinded pivotal studies • Subjects receive identical, fixed treatments • Data collection focuses on final, pragmatic outcomes © Guidant 2004

Workshop 2005 Device vs. Drug - statistical Device Studies • Difficult to administer • FDA requirement: single pivotal trial • Reliability and quality control issues 5 Drug Studies • Easy to administer • FDA requirement: two pivotal trials • Reliability and quality control issues are less prominent © Guidant 2004

Workshop 2005 An Example To test an ‘intelligent’ device with a new feature: the new feature include one programmable parameter; physician needs to find the optimal setting of each patient based on the responses of an acute test. This parameter can be modified anytime. Questions: how do we model a true ‘device effect’ adjusted for the physician, parameter, and patient? What is the ‘treatment’ we are interested in? Feature? Parameter? Programmability? Utility? What is the experimental unit? When do we need to do new studies? 6 © Guidant 2004
- Slides: 6