FDA Clinical Investigator Audits Insight and Practical Tips




































- Slides: 38

FDA Clinical Investigator Audits: Insight and Practical Tips Office of Research Compliance Quality Improvement Office

Overview • Provide background about FDA’s Audit Program • Describe the general process of an FDA Clinical Investigator Inspection – Initial notification – Site preparation – Site visit – Post site visit including how to respond to an FDA Form 483

FDA Audit (Inspection): Background (1 of 2) • Conducted under FDA’s Bioresearch Monitoring (BIMO) Program by ‘FDA investigators’ (auditors) • BIMO Program objectives are to help: – ensure the protection of the rights, safety, and welfare of human research subjects – verify the accuracy and reliability of clinical trial data – assess compliance with statutory requirements and FDA's regulations

FDA Audit (Inspection): Background (2 of 2) • FDA BIMO Program involves site visits to: – clinical investigators – sponsors – contract research organizations – Institutional Review Boards (IRBs) – nonclinical (animal) laboratories – bioequivalence analytical laboratories

FDA Audits of Clinical Investigators • Announced or Unannounced – Majority are announced and allow an average of 3 days notice • Types of audits: – For-cause/Directed • Reasons may include follow-up to previous FDA Audit, response to concerns raised by sponsor, IRB, employees, study subjects; submission of unrealistic data – Routine/Surveillance • As part of drug/device approval process • Usually highest enrolling sites

The Audit Process: Site Notification • Clinical Investigator (PI) is generally notified by phone • Questions to ask if the FDA calls: – – – What? ----Study to be audited & needed documentation When? ---Start date and expected duration Where? --Location(s) for the site audit Who? ----FDA Inspector name(s) & contact information Why------Rationale for audit selection • FDA is authorized at reasonable times to access, inspect, and copy any required records related to the clinical investigation.

The Audit Process: Site Preparation (1 of 2) • Notify applicable parties of the impending audit – Study team members • sub-investigators, coordinators, regulatory personnel, etc. – IU SOPs require you to inform IU QIO who will: – provide assistance throughout, including consultation & advice regarding preparation, execution, & follow-up – attend the initial & exit meetings with FDA along with any another related meetings; relay pertinent updates to ORC & other University oversight – coordinate and review any written response to FDA Form 483 • Others to notify include: study sponsor, pharmacy, lab, ICRC, department chair, University Counsel, etc.

The Audit Process: Site Preparation (2 of 2) • Ensure all records are available & organized – Complete subject records • Source documents & case report forms • Paper or electronic; if electronic, you must use appropriate methods: limited access or plan to sit with the auditor(s) – Regulatory documents • delegation of authority log, enrollment log, IRB approvals, 1572, CV’s, training records, adverse event reports, etc. – QIO will offer an FDA Audit Checklist • List of studies performed by the PI – protocol number, protocol title, product name, IND/IDE number, name of sponsor & study dates.

The Audit Process: Site Visit (1 of 3) • Auditor conducts an initial meeting with the study team – generally, include the PI, main coordinator(s), & the IRB auditor but may vary depending on the situation • Auditor is required to: – display his/her FDA credentials – issue a Form FDA 482 (Notice of Inspection) to the PI • Auditor generally explains the process, expectations, & asks the study team questions about conduct of the study.

The Audit Process: Site Visit (2 of 3) • Study team should request a meeting at the end of each day with the auditor(s). The PI doesn’t have to attend this meeting but it is helpful if they check in daily. • Auditor reviews the research records and asks questions as they arise in order to verify compliance with the regulations, 21 CFR parts 50, 56, 312, and/or 812. • Records inspected generally include regulatory files, drug/device accountability logs, informed consent documents, other source documents, & CRFs.

The Audit Process: Site Visit (3 of 3) • At the end of an inspection, the auditor conducts an exit interview with the PI, study team, and IRB Auditor. • The auditor discusses the findings from the inspection. • If deficiencies in meeting the regulations were found, a written Form FDA 483 (Inspectional Observations) will be issued to the PI. • The 483 describes any inspectional observations that, in the opinion of the auditor conducting the inspection, represent deviations from applicable statutes and regulations.

Common FDA Findings • Failure to follow the investigational plan and signed investigator statement/agreement (e. g. , failure to conduct or supervise the study in accordance with the relevant, current protocol(s)). See 21 CFR 312. 60 and 812. 110(b). • Protocol deviations (e. g. , failure to appropriately document and report any medically necessary protocol deviations). See 21 CFR 312. 66 and 812. 150(a)(4). • Inadequate recordkeeping. See 21 CFR 312. 62 and 812. 140(a). • Inadequate accountability for the investigational product. See 21 CFR 312. 62(a) and 812. 140(a)(2). • Inadequate subject protection, including informed consent issues. See 21 CFR part 50, 312. 60, and 812. 100.

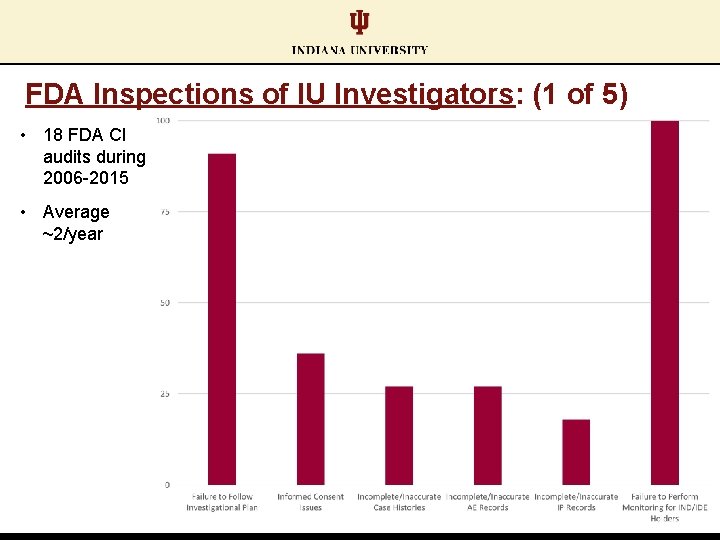
FDA Inspections of IU Investigators: (1 of 5) • 18 FDA CI audits during 2006 -2015 • Average ~2/year

FDA Inspections of IU Investigators: (2 of 5) Failure to Follow the Investigational Plan: • Out of window visits • “Complete” PE not performed; “Complete” vital signs not obtained; temperatures not recorded • Not contacting LARs/family members according to protocol plan • Allowing “untrained” staff to complete study procedures • Failure to dose reduce or hold dosing according to protocol • Failure to perform safety procedures (EKGs, observation after dosing) • Samples not processed according to protocol • Enrolling subjects who did not meet all inclusion/exclusion criteria

FDA Inspections of IU Investigators: (3 of 5) Informed Consent Issues: • Failure to obtain informed consent from subjects despite being conscious and oriented • Reading consent to subjects who were without reading glasses, rather than rescheduling for time when glasses were available • Allowing a significant other (whose status as an LAR was questionable) to provide LAR consent • Missing ICS • Not having LARs provide consent prior to study participation beginning

FDA Inspections of IU Investigators: (4 of 5) Incomplete/Inaccurate Case Histories: • Inconsistency between various source & CRFs • Incomplete documentation of data submission to sponsor Incomplete/Inaccurate AE Records: • Discovery of additional AEs not assessed by PI • Failure to report AEs annually • Failure to report AEs “immediately”

FDA Inspections of IU Investigators: (5 of 5) Investigational Product Issues: • Receipt/Return dates not logged • Serial #s not recorded on data sheets • Failure to keep IP dispensing product (IV bags) for sponsor reconciliation Study Monitoring Issues (when PI holds IND/IDE): • No written monitoring plan • Not following the written monitoring plan

The Audit Process: Post Site Visit (1 of 4) • Auditor prepares a written Establishment Inspection Report (EIR). The EIR, Form FDA 483 (if issued), copies of any materials collected during the inspection, & any PI response are forwarded to the appropriate FDA Center for further evaluation & final classification. • Classifications: – No Action Indicated (NAI) • No objectionable conditions found or the significance of the documented objectionable conditions found does not justify further action. – Voluntary Action Indicated (VAI) • Objectionable conditions found, but Center is not prepared to take any further regulatory action. – Official Action Indicated (OAI) • Objectionable conditions found & one of the actions will be recommended: Notice of disqualification & opportunity to explain, a Warning Letter, reinspections to verify corrective actions, rejection of data from site, or civil/criminal actions

The Audit Process: Post Site Visit (2 of 4) Form Inspectional Observations Form (FDA-483) WHAT is it? • Describes the auditor’s observations of deviations from statutes and regulations. WHO responds? WHEN do you respond? PI • Verbally during the exit interview; and/or • In writing after the inspection • A 483 is issued ONLY IF deviations are observed. • The PI should respond to the form in writing within 15 days to the District office • Issued, and discussed, at the audit close-out meeting. WHAT is it NOT? Inspectional Observations Form (FDA-483) PI • It is NOT the final determination regarding the PI/site compliance. • Verbally during the exit interview; and/or • In writing after the inspection • The PI should respond to the form in writing within 15 days to the District office

The Audit Process: Post Site Visit (3 of 4) Form WHAT is it? Establishment Inspection Report (EIR) • Contains: • Brief history of prior inspectional findings and FDA/CI actions in response to previous findings (if applicable) • Auditor’s narrative report • Any refusals, voluntary corrections or promises made by PI/site • Written by auditor within 30 days. • Provided to PI when audit is “closed”. You may have to ask your auditor for this! WHO responds? WHEN do you respond? N/A

The Audit Process: Post Site Visit (4 of 4) Your inspection classification determines what type of FDA Audit follow-up letter you receive: (1) A letter that generally states that FDA observed basic compliance with pertinent regulations. (Note that a letter is not always sent when FDA observes no significant deviations. ) (2) An Informational or Untitled Letter that identifies deviations from statutes and regulations that do not meet the threshold of regulatory significance for a Warning Letter. (3) A Warning Letter that identifies serious deviations from applicable statutes and regulations. A Warning Letter is issued for violations of regulatory significance. (4) A Notice of Initiation of Disqualification Proceedings and Opportunity to Explain (NIDPOE).

How do we avoid getting a Warning Letter?

PI’s Response to the FDA-483: • Not required, but EXPECTED and encouraged • Why respond? § Demonstrates an understanding and acknowledgement of the observations § Demonstrates a commitment to correct (i. e. the intent to voluntarily comply) § Establishes credibility § Could possibly mitigate an FDA compliance decision for further action (e. g. Warning Letter) –FAILURE TO RESPOND TO THE 483 CAN BE SEEN AS INDIFFERENCE BY THE FDA

FDA-483: How to respond: (1 of 2) Your response should tell the whole story and stand on its own! • Assess each observation § Focus on specifics § Focus on system-wide/global implications § Consider root-cause analysis § Focus on the regulatory requirement(s) associated with the observation • Develop action plan to achieve immediate, short-term, and longterm corrections • Develop preventive action plans • Know when to seek outside resources • QIO reviews your 483 Response prior to FDA submission

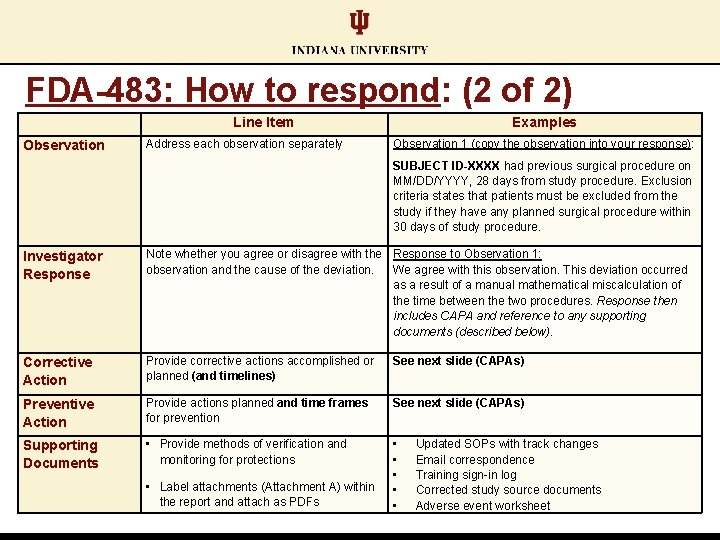
FDA-483: How to respond: (2 of 2) Observation Line Item Address each observation separately Examples Observation 1 (copy the observation into your response): SUBJECT ID-XXXX had previous surgical procedure on MM/DD/YYYY, 28 days from study procedure. Exclusion criteria states that patients must be excluded from the study if they have any planned surgical procedure within 30 days of study procedure. Investigator Response Note whether you agree or disagree with the Response to Observation 1: observation and the cause of the deviation. We agree with this observation. This deviation occurred as a result of a manual mathematical miscalculation of the time between the two procedures. Response then includes CAPA and reference to any supporting documents (described below). Corrective Action Provide corrective actions accomplished or planned (and timelines) See next slide (CAPAs) Preventive Action Provide actions planned and time frames for prevention See next slide (CAPAs) Supporting Documents • Provide methods of verification and monitoring for protections • • • Label attachments (Attachment A) within the report and attach as PDFs Updated SOPs with track changes Email correspondence Training sign-in log Corrected study source documents Adverse event worksheet

Corrective and Preventive Action Plans (CAPAs): (1 of 2) Examples include, but are not limited to: • Correct anything that can be “fixed” (missing data transcription, incorrect data transcription, missed submissions to the IRB, sponsor notification, etc. ) • Create or update written SOPs • Provide training (or retraining) to study team members/clinic staff involved in research procedures • Changes in personnel • Continuing education • Develop an audit system to detect problems • Develop electronic systems to track work and dates

Corrective and Preventive Action Plans (CAPAs): (2 of 2) • Be complete • include specific details and timelines of the steps taken to correct the deficiency and prevent the recurrence • Be realistic • Implementing new programs/mechanisms takes time • Be able to deliver what you promise • Provide documentation of implemented corrections and preventions where feasible.

Response/CAPA Supporting Documentation: Provide copies of: • corrections: • Corrected source or corrected CRF (screenshot or audit trail of corrected e. CRF) • Documentation of IRB submission and approval • Documentation of Sponsor notification and instructions • training/re-training materials: • Agendas or Materials Used • Sign-in sheets • Correspondence to study team members • preventive measures: • Updated (tracked)/New SOPs • New compliance tools (checklists, etc. )

Examples of Poor 483 Responses

FDA Warning Letter Examples (CI): (1 of 6) July 13, 2015 Warning Letter to a CI (B. Michlin): You failed to ensure that the investigation was conducted according to the investigational plan [21 CFR 312. 60]. The investigational plan requires that the screening visit include safety laboratory tests. . . You failed to adhere to these requirements. Specifically: Protocol XXXX requires the screening visit to include safety laboratory tests (including virology, hematology, and clinical chemistry) of the subjects’ blood. Screening blood samples were not collected for the following subjects iv. Subject 614016 i. Subject 614005 vii. Subject 614022 v. Subject 614017 ii. Subject 614009 viii. Subject 614023 vi. Subject 614019 iii. Subject 614015 We acknowledge your February 10, 2015 written response in which you state that a Quality Assurance Internal Chart Review Tool will be used periodically to ensure that protocol-required procedures are being completed. We also acknowledge your February 10, 2015, written response to Item a. vi. above, in which you state that source documentation standard operating procedures (SOPs) were updated to include specific documentation of all laboratory tests performed. However, although you promised certain corrective measures in your response, you did not specifically address your failure to collect the protocol-specified screening blood samples. Your response is inadequate because you did not provide documentation of the SOPs you will use for oversight of the studies you conduct. In addition, you did not provide any details of the in-service protocol and specimen-handling training for you and your staff. Without this information, we are unable to determine whether your corrective action plan is adequate to prevent similar violations in the future.

FDA Warning Letter Examples (CI): (2 of 6) June 29, 2015 Warning Letter to a CI (H. Gross, Part 1): You failed to ensure that the investigation was conducted according to the investigational plan [21 CFR 312. 60]. The investigational plan for Protocol XXXX required you to ensure that study subjects met the protocol inclusion and exclusion criteria before their enrollment. You failed to adhere to these requirements. Specifically: i. Protocol XXXX specified that for subjects with proteinuria > 2+ or urine protein creatinine ratio > 0. 5, a 24 -hour urine protein should be obtained and the level must be <1 gram of protein in 24 -hour urine to be eligible for enrollment. Subject 2102 had a protein level of 37. 1 mg/dl, which is clinically considered 2+ protein (<100 mg/dl). However, a 24 -hour urine test was not performed. This subject was randomized into the clinical investigation on May 17, 2012. ii. Protocol XXXX specified that subjects receiving chronic therapy with nonsteroidal anti-inflammatory agents or other antiplatelet agents, including clopidogrel, will not be enrolled into the clinical investigation. Subject 2100 was taking Plavix (clopidogrel) between January 3, 2012, and March 2, 2012. However, this subject was randomized into this clinical investigation on February 6, 2012. In your January 28, 2015, written response to the violations noted in Item 1. a. , you agreed that Subjects 2102 and 2100 did not meet the eligibility criteria. You indicated that as part of your corrective action plan, you developed a standard operating procedure (SOP), “Second Eligibility Check, ” which specifies that study staff will verify that eligibility criteria are met prior to enrollment. Your response is inadequate because, although you stated that your SOP “Second Eligibility Check” has been put into effect and that your research coordinators are well aware of this requirement, you failed to provide documentation that your research staff have been adequately trained in this SOP. Without such documentation, we are unable to determine whether your corrective action plan appears sufficient to prevent similar violations in the future.

FDA Warning Letter Examples (CI): (3 of 6) June 29, 2015 Warning Letter to a CI (H. Gross, Part 2): You failed to ensure that the investigation was conducted according to the investigational plan [21 CFR 312. 60]. (continued) Protocol XXXX specified that XXXX will be administered over 90 (± 10) minutes, and that subjects will be observed postadministration for at least 30 minutes. After this observation period, XXXX/placebo will be administered. Any infusion-related symptoms must have resolved before the administration of XXXX/placebo. i. You administered the study drugs in reverse order for one subject. For Subject 006, XXXX/placebo infusion was followed by XXXX infusion at the following study visits: Cycles 4, 7, and 10 -12. ii. You failed to adhere to the required 30 -minute observation period before administering XXXX/placebo for all subjects enrolled in this clinical investigation at the following study visits: 1. For Subject 001: Cycles 1 and 2 2. For Subject 003: a. Cycles 2 -7 and 9 -14 b. Cycle 8: No observation period was performed; XXXX/placebo was administered one minute before the XXXX infusion was stopped… In your January 28, 2015, written response you confirmed that the order of administration of study drugs was reversed for five of Subject XX’s treatment cycles. You also confirmed that observation periods were not followed for multiple treatment cycles for four subjects. You indicated that as part of your corrective action plan, you trained study staff to review protocol orders and infusion requirements with infusion nurses before starting study drug administration. Your response is inadequate because it does not contain sufficient detail. Specifically, it is unclear whether study staff and infusion nurses would be adequately trained on protocol requirements before initiating the study, or only before study drug administration for each subject. As a result, we are unable to determine whether your corrective action appears sufficient to prevent similar violations in the future.

FDA Warning Letter Examples (CI): (4 of 6) June 29, 2015 Warning Letter to a CI (H. Gross, Part 3): You failed to ensure that the investigation was conducted according to the investigational plan [21 CFR 312. 60]. (continued) Protocol XXXX required coagulation profile laboratory tests to be performed at baseline; at Cycles 4, 8, and 9 -x; at the summary visit; and at the 30 -day follow-up visit. For Protocol XXXX, the coagulation profile included prothrombin time (PT or INR) and partial thromboplastin time (PTT). You failed to perform coagulation profile laboratory tests for four of the five subjects that received study drug at the following specified intervals: i. For Subject 2100: Cycles 4 and 8 -17 ii. For Subject 2102: Cycle 4 iii. For Subject 2104: Cycles 4, 8, and 13 -17… In your January 28, 2015, written response to the violations noted in [the items]. above, you confirmed that protocol-required laboratory tests were missed. . . You indicated that as a part of your corrective action plan, you have added a “clinical trials link” to your site’s electronic medical record (EMR) to provide access to study information for study staff. You further indicated that you will review with study staff the process of accessing study information and the importance of following the study calendar. Your response is inadequate because you did not provide sufficient information to enable us to evaluate the adequacy of your corrective action plan for use in any future clinical research that you may conduct. It is unclear how adding a “clinical trials link” to your site’s EMR will ensure that protocol requirements will be met for studies conducted at your site. You did not provide any details of a corrective action plan to prevent similar violations from occurring in the future, nor have you provided sufficient details regarding your plan to implement additional measures and procedures to address the inspection findings. Without these details, we are unable to determine whether your corrective action plan appears sufficient to prevent similar violations in the future.

FDA Warning Letter Examples (CI): (5 of 6) May 4, 2015 Warning Letter to a CI (B. Nguyen, Part 1): You failed to ensure that the investigation was conducted according to the investigational plan [21 CFR 312. 60]. Protocol XXXX requires that TSH be performed at Baseline, Week 24, and every 24 weeks thereafter, up to and including the End of Study/Final Visit. TSH is a safety assessment in the study. Specifically: a. b. c. Subject 033 -800 -02 missed TSH collection at Weeks 48 and 72. Subject 033 -008 -03 missed TSH collection at Week 24. Subject 033 -008 -04 missed TSH collection at End of Study. In your October 2, 2014, written response to the Form FDA 483, you indicated that missed laboratory tests were mainly due to subject noncompliance. You stated that orders for laboratory tests were given to the subjects at each visit. You noted that corrective measures taken at the time to address subject noncompliance included phone calls to subjects and oral reminders during the following clinical visit. Your response is inadequate because you have not provided sufficient details about your corrective action plan. You have not provided adequate documentation of your efforts to address subject noncompliance (e. g. , phone calls, oral reminders during the following clinical visit) in the source records. Without those details in the records, we are unable to determine whether your corrective action plan is adequate to prevent similar violations in the future.

FDA Warning Letter Examples (CI): (6 of 6) May 4, 2015 Warning Letter to a CI (B. Nguyen, Part 2): You failed to ensure that the investigation was conducted according to the investigational plan [21 CFR 312. 60]. (continued) Protocol XXXX requires that XXXX treatment be altered if one of the following three events occurs: Protein ≥ 30 mg/d. L on the urine dipstick, confirmed by 24 -hour protein >1. 5 g/24 hours. Creatinine clearance <50 m. L/min (Cockcroft and Gault formula). Urea >2 times the upper limit of normal. If one of the three events noted above occurs, the study protocol requires that XXXX be interrupted until the noted level returns to baseline, and XXXX will then be restarted at the same dose. If one of the three events noted above occurs a second time, XXXX will be interrupted until the noted level returns to baseline, and XXXX will then restarted with a dose reduction of 1. 5 mg/kg/day (i. e. , the new dose will be 6 mg/kg/day). If one of the three events noted above occurs a third time, XXXX will be permanently discontinued. On January 16, 2013, Subject 1313 in Protocol XXXX had a baseline creatinine level of 0. 5 mg/d. L. This subject’s creatinine clearance on February 27, 2013, was 49. 7 m. L/min (Cockcroft and Gault formula); therefore, the protocol required that XXXX treatment be interrupted until a return to baseline renal function. This action was not taken, however. On March 13, 2013, the subject’s creatinine level had increased to 1. 6 mg/d. L, indicating worsening renal function. The subject’s final dose of XXXX was given on the morning of March 18, 2013, and the subject was discontinued from the study on the same day due to progressive disease. In your October 2, 2014, written response to the Form FDA 483, you indicated that the subject’s grade 2 renal insufficiency was “considered related to a major fluid retention, mainly with ascites and inferior vena cava compression, related to tumor evolution. ” You also indicated that “this functional renal deficiency was considered non-threatening, ” and that “continuation despite renal functional dysfunction was a medical choice” in the subject’s best interest. Your response is inadequate because the cause of renal insufficiency is not a factor in adhering to the protocol’s treatment modification rules for renal disorders, and you have not provided a corrective action plan to address this issue. Without a corrective action plan describing how you will adhere to protocol-specified treatment modification rules for renal disorders, we are unable to determine whether similar violations will be prevented in the future.

FDA Guidance Documents: • FDA Information Sheet for IRBs, Clinical Investigators, and Sponsors: FDA Inspections of Clinical Investigators: http: //www. fda. gov/downloads/Regulatory. Information/Guidances/UC M 126553. pdf • FDA Presentation “Writing an Effective 483 Response”: http: //www. fda. gov/downloads/Biologics. Blood. Vaccines/News. Events/ Workshops. Meetings. Conferences/UCM 102921. pdf • FDA Regulatory Procedures Manual: http: //www. fda. gov/ICECI/Compliance. Manuals/Regulatory. Procedure s. Manual/default. htm • FDA BIMO Clinical Investigators and Sponsor-Investigators Manual (Guidance for FDA Staff): http: //www. fda. gov/downloads/ICECI/Enforcement. Actions/Bioresearc h. Monitoring/ucm 133773. pdf

Additional Information from FDA • Inspections, Compliance, Enforcement, and Criminal Investigations Posted Warning Letters http: //www. fda. gov/ICECI/Enforcement. Actions/Warning. Letters/default. htm • Clinical Investigator Inspection List (CLIIL) The Clinical Investigator Inspection List (CLIIL) contains names, addresses, and other pertinent information gathered from inspections of clinical investigators who have performed studies with investigational new drugs. The list contains information on inspections that have been closed since July 1977. http: //www. fda. gov/Drugs/Information. On. Drugs/ucm 135198. htm

QUESTIONS? http: //researchcompliance. iu. edu/qio/index. html