FDA c GMP Inspections Peking University 2005 Robert

FDA c. GMP Inspections Peking University 2005 Robert C. Horan, Ph. D FDA Pharmaceutical Inspectorate New York District

FDA Inspections n n n Periodic (biennial) comprehensive c. GMP Pre-Approval Inspection(PAI) “For cause” Inspection may involve more than one assignment and will verify corrections to previous inspections. All inspections cover GMPs

Foreign Inspections by Country in FY 2004
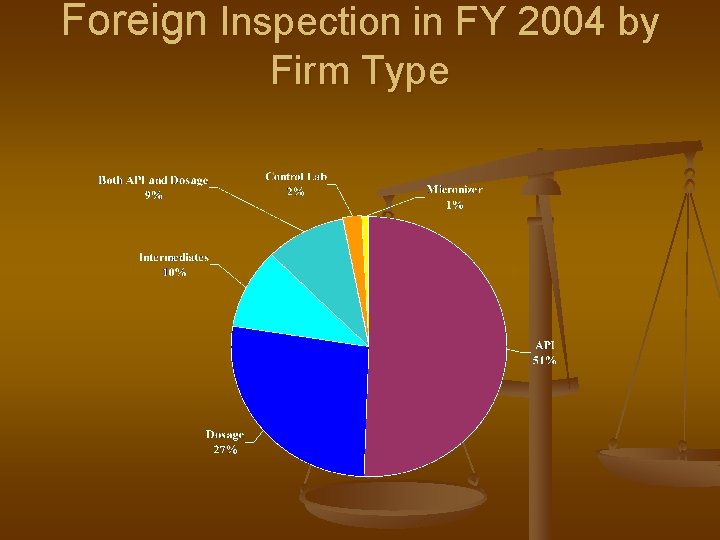
Foreign Inspection in FY 2004 by Firm Type

FDA c. GMPs for 21 st Century Initiative Announced 8/2002; objectives include: n Encourage adoption of new technologies n Promote industry use of modern quality system approaches n Encourage risk-based approaches which focus on critical elements n Ensure FDA review, compliance and inspection policies based on state-of-art pharmaceutical science

FDA c. GMPs for 21 st Century Initiative n n n n Systems Based Inspections Risk-Based Approach to Manufacturing and Regulation Pharmaceutical Inspectorate PAT Guidance document/ PAT Team Quality Systems Guidance document Process Validation (Compliance Policy Guide revised; Guidance being revised) 21 CFR Part 11 Electronic Records Guidance (riskbased; geared toward GMP documents)

Pharmaceutical Inspectorate n n n Cadre of most experienced investigators who are dedicated to drug inspections Intensively trained along with quality reviewers and compliance staff in FDA headquarters (HQ) Overall goal is to have PI work closely with HQ personnel – more efficiently integrate review and inspection functions

Pharmaceutical Inspectorate n n FDA Review staff, Compliance Officers and PI candidates attended training modules which focused on: Current Regulatory Programs Advanced Quality Systems PAT and Modern Pharmaceutical Technology Risk Management

Pharmaceutical Inspectorate Field Investigators (18) from across U. S. make up the Pharmaceutical Inspectorate n Screening process with certification board n Completed training with HQ personnel n One month detail working with HQ staff n Level III certification (highest level) n Conduct PAIs, complex drug inspections

Process Analytical Technology n PAT is a system for designing, analyzing and controlling manufacturing through “real time” measurements of critical quality attributes of raw and in-process materials and processes, with the goal of ensuring final product quality. See FDA Guidance document on PAT

Process Analytical Technology Examples of PAT applications: n Continuous real time measurements of content uniformity of tablets during production (using near Infra-Red) n Near IR measurement of moisture level during API drying process to determine actual end of operation for each batch

Process Analytical Technology A process is generally considered well understood when: n All critical sources of variability are identified and explained n Variability is managed by the process “Quality cannot be tested into products; it should be built-in or should be by design. ”

Process Validation Life Cycle Approach n n n Process validation begins with process development and continues beyond the initial “validation” batches for as long as product is manufactured/ marketed Sources of critical variability identified and controlled Quality System role in maintaining validated state (quality built in; not tested into product)

Process Validation n n FDA Compliance Policy Guide “Process Validation Requirements for Drug Products and Active Pharmaceutical Ingredients CPG 7132 c. 08”; revision date 12 March 2004 FDA Industry Guideline on Process Validation – currently being revised

System Inspections n n n Quality Facilities and Equipment Materials Production Packaging/Labeling Laboratory Controls

Most Common GMP Deficiencies by System – API/ Dosage Inspections for 2004/5

“State of Control” n n n Detailed inspection of a system so that the findings reflect the state of control in that system for every product (profile) class If one of the six systems is out of control, the firm is considered out of control A system is considered out of control based on GMP deficiencies which suggest lack of assurance of quality

Quality System n n n Quality must be built into the process Quality is not tested into the product Assurance of Quality comes from - Design of robust process based on thorough knowledge of that process and the sources of variability - Effective Quality System in place

Role of Management in QS Management is responsible for: n n Organizational structure All Processes All Procedures Facilities & Resources In short, everything to insure product quality, customer satisfaction and continuous improvement

Quality System Responsibilities n n 1) 2) 3) 4) 5) Assures overall compliance with c. GMPs Review and approval duties for: Product Quality Reviews (at least annually) Complaint reviews Discrepancy/ failure investigations Change Control CAPA (Corrective And Preventive Action)

Quality system (continued) 6) Reprocess/ Rework 7) Validation/ Revalidation 8) Rejects 9) Stability Failures/ Out of trend data 10) Quarantine products 11) Documented GMP & Job Related Training

Laboratory Control System n n n Adequate lab facilities under the Quality Unit which is independent from Production Adequately staffed laboratories (supervisory and bench personnel) Written specifications for raw materials, intermediates, APIs, labels & packaging Written procedures for sampling, testing, approval or rejection of materials and for the recording and storage of data Change control for written procedures Method validation/ revalidation

Laboratory Control System n n n Reference Standards (primary; secondary) Equipment Qualification Calibration: written procedures, schedule, documentation Validation and Security for computerized handling of test results and related data; system for assuring integrity of all lab data Laboratory controls followed and documented

Laboratory Control System n n n Written procedure (SOP) covering out of specification “oos” results Investigation of “oos” results conducted in a timely manner as per SOP and documented (complete records maintained). Conclusions from “oos” investigations documented and corrective actions/ need for addition investigation identified and implemented. “oos” review included in Product Quality Reviews

Laboratory Control Records n n n n Description of samples Identification of method used Raw data for sample/ standard preparation, reagents Complete record of all data from testing Record of all calculations Statement of the test results; how compare with established acceptance criteria Signature of the person who performed each test; dates tests performed Date/ signature of second qualified person who reviewed original test records for accuracy, completeness and compliance with established standards

Production System n n n Training (documented; job-related) Master production and control records Batch production and control records Change control procedure Contemporaneous, accurate and complete batch production documentation Implementation and documentation of inprocess controls, tests, and examinations

Production system (continued) n n Adequate written procedures & practice for charge-in of materials Identification of equipment with contents, stage of manufacturing, status Equipment cleaning records Established time limits for completion of production steps/ stages

Production system (continued) n n Deviations investigated and documented contemporaneously with investigation Process validation based on knowledge of process (scientific basis for identifying critical steps/ critical process parameters/control points) Justification and consistency of in-process specifications and final product specifications Data/ information documented and available to Quality Unit for review (trending, investigations etc. )

Facilities & Equipment System n n n FACILITIES Location, design, construction appropriate to facilitate cleaning, maintenance, operations Layout and air handling designed and constructed to prevent crosscontamination Flow of materials & personnel designed to prevent mix-ups or contamination

Facilities & Equipment System n n n Defined areas or other control systems to prevent mix-ups or contamination Incoming materials (id, quarantine) Sampling area (prevent contamination) Quarantine (intermediates, APIs) Released materials Rejection

Facilities & Equipment System EQUIPMENT n n n n Appropriate design, size, location, non-reactive product contact surfaces Identification clearly marked Qualification (DQ, IQ, OQ, PQ) Calibration Preventive Maintenance schedule and procedures Cleaning procedures and validation Records of use, cleaning, maintenance

Facilities & Equipment System Lubricants, heating fluids or coolants (not contact/alter product quality) n Closed or contained equipment n Inspection prior to use ****************** n Separate facilities or containment where needed (penicillins, highly potent compounds etc. ) n

Utilities n n n Qualified and appropriately monitored; drawings should be available Designed and constructed to prevent contamination or cross-contamination Recirculated air to production (same concern) Permanently installed pipework should be appropriately identified Drains of adequate size with air break

Water n n Process water at minimum meeting WHO guidelines for potable water Justify quality of water used to achieve stated API quality and establish specifications Water treatment facilities validated API to be used for incorporation into sterile dosage form – water used in later stages should be monitored and controlled for total microbial counts, objectionable organisms and endotoxins

Materials System n n n n Written procedures for receipt, identification, quarantine, storage, handling, sampling, testing and approval or rejection of materials System to evaluate suppliers (critical materials) Purchased against agreed specification Change control process for changing suppliers Upon receipt check for correct labeling, seals Before co-mingling bulk material, id/test Assurances obtained from non-dedicated tankers

Materials System n n n Identification on large storage containers and associated manifolds, filling and discharge lines Code given to received batches; status identity At minimum, a specific identity test on incoming batches; COA Supplier evaluation should include three fully tested batches; one fully tested batch/year Written sampling plan with justification Prevent contamination of sampled containers

Materials System n n n Stored in manner to prevent degradation, contamination, no adverse effect on quality Drums, bags, boxes off the floor First in, first out Rejected materials identified and controlled under a quarantine system Established re-test/ re-evaluation periods

Packaging & Labeling System n n Written procedures for receipt, identification, quarantine, sampling, examination and/or testing P&L should conform to specifications Records maintained for each shipment (showing receipt, examination & result) Containers protective, clean, not alter product quality; if re-used, cleaned & labeling defaced

Labeling n n n Access to label storage area limited Written procedures for reconciliation; investigation if discrepancy All excess labels with batch #, destroyed Obsolete labels destroyed Printing devices controlled to insure accuracy of label (against batch record) Print labels checked against master and a copy placed into the batch record

Packaging/ Labeling Operations n n n n Documented procedures to assure correct packaging materials/ labels used Operations designed to prevent mix-ups Labels: API name, batch #, storage conditions Shipped API: Name/ address manufacturer; special transport conditions; expiry/ retest date Documented clearance before operations Packaged/ labeled intermediates or APIs examined as part of packaging (documented) Seal employed to assure package integrity

APIs are Drug Substances n n n FDA Food, Drug and Cosmetic Act definition of drug includes “articles intended for use in the diagnosis, cure, mitigation, treatment or prevention of disease in man or other animals” (no distinction between APIs & dosage forms) Before ICH Q 7 A, FDA used dosage drug regulations as guidance for API inspection Still true (see next slide) , however, ICH Q 7 A provides guidance on the application of those c. GMPs to APIs)

From current FDA Compliance Program 56002 for Drug Manufacturing Inspections: (This is the compliance program for FDA Investigators; this revision introduced Systems Inspections) “The c. GMP regulations are not direct requirements for manufacture of APIs……. but they are guidance for c. GMP in API manufacture. ”

Current FDA Compliance Guide on Process validation From FDA Compliance Policy Guide “Process Validation Requirements for Drug Products and Active Pharmaceutical Ingredients CPG 7132 c. 08”; revision date 12 March 2004: Validation of manufacturing processes is a requirement of the Current Good Manufacturing Practice (c. GMP) regulations for finished pharmaceuticals, and is considered an enforceable element of current good manufacturing practice for active pharmaceutical ingredients (APIs) under the broader statutory c. GMP provisions of the Federal Food, Drug, and Cosmetic Act”.

Differences API/ Dosage Form n n APIs involve purification steps GMP controls tighter for later API steps API impurity profile is critical focus and steps which produce or remove impurities require greater control and validation Dosage forms do not involve purification

Similarities APIs/Dosage Forms n n n Require demonstrated knowledge of process and application of appropriate GMP controls to assure safety, identity, strength, quality and purity. Systems in control to be in compliance Life Cycle Approach to Validation (beyond the initial “conformance batches”)

Similarities include…. n n n Processes for specific products vary in complexity (either API or dosage can involve complex or simple processes) In-Process Controls Finished Product Controls Critical Steps/ Critical Process Parameters Process Validation Quality Assurance for consumer is based on understanding & Control of Sources of Process/ Product Variability

More Similarities…. . n n n Science based approach for the establishment of processes Knowledge of process based on Process Development work Design Of Experiments (DOE) Quality System (review/ trending) Continuous Improvement possible within well characterized process
- Slides: 47