FCA LIABILITY FOR OFFLABEL MARKETING Serostim Update THOMAS

FCA LIABILITY FOR OFF-LABEL MARKETING Serostim Update THOMAS M. GREENE Greene & Hoffman November 8, 2005 1

MAJOR OFF-LABEL CASES Serostim Neurontin $ 430 M Global $ 704 M $ 240 M Criminal $ 137 M $ 190 M Civil $ 567 M THOMAS M. GREENE & HOFFMAN - BOSTON, MA 2

FCA LIABILITY n “Court holds that Relator has presented evidence showing that it was foreseeable that Parke-Davis's conduct (including nonfraudulent promotion of off-label Neurontin uses) would ineluctably result in false Medicaid claims. ” United States ex rel. Franklin v. Pfizer et al. , No. 96 -11651 -PBS, 2003 U. S. Dist. LEXIS 15754, *15 (D. Mass. Aug. 22, 2003) THOMAS M. GREENE & HOFFMAN - BOSTON, MA 3

SEROSTIM n APPROVED INDICATION Indicated for the treatment of HIV patients with wasting or cachexia to increase lean body mass and body weight, and improve physical endurance. Concomitant antiretroviral therapy is necessary. THOMAS M. GREENE & HOFFMAN - BOSTON, MA 4

Erosion of Market n Almost immediately after approval, market for Serostim began to decline steeply due to development of protease inhibitor drugs and combination therapy, which greatly curtailed AIDS wasting syndrome THOMAS M. GREENE & HOFFMAN - BOSTON, MA 5

SEROSTIM n OFF-LABEL USE q Treatment of wasting even when there is no weight loss q Using BIA devices and unapproved software to compare body cell mass to a theoretical “ideal” reading THOMAS M. GREENE & HOFFMAN - BOSTON, MA 6

MAIN OFF-LABEL MESSAGE n Wasting was being masked by weight gain caused by protease inhibitors n Body cell mass was the true measure of wasting – a patient could have no weight loss or even weight gain, and still be wasting, which would only be detectable by measuring body cell mass THOMAS M. GREENE & HOFFMAN - BOSTON, MA 7

KICKBACKS n Target “thought-leaders” n Offer all-expense-paid trip to Cannes in exchange for 30 new scripts ($630, 000) THOMAS M. GREENE & HOFFMAN - BOSTON, MA 8
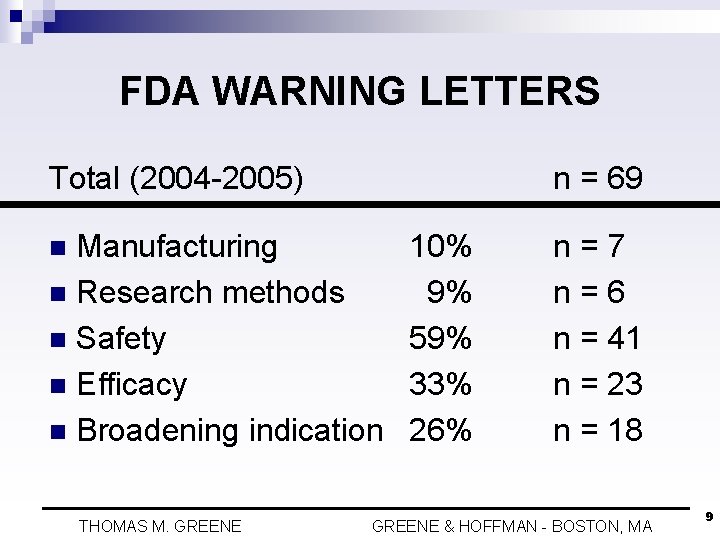
FDA WARNING LETTERS Total (2004 -2005) n = 69 Manufacturing n Research methods n Safety n Efficacy n Broadening indication n THOMAS M. GREENE 10% 9% 59% 33% 26% n=7 n=6 n = 41 n = 23 n = 18 GREENE & HOFFMAN - BOSTON, MA 9

FDA WARNING LETTERS n Only one warning letter relates to off-label statements made at a CME conference n Only one warning letter relates to off-label statements made in a “Dear Doctor” letter n No warning letters relate to off-label statements made in detailing THOMAS M. GREENE & HOFFMAN - BOSTON, MA 10

MARKETING TACTICS WITH POTENTIAL FOR MISUSE n n n n Internet Clinical Study Results Database CMEs Promotional Events Detailing Journal Articles Studies of Off-label Uses Redefining Product Rationale THOMAS M. GREENE & HOFFMAN - BOSTON, MA 11

CONCLUSIONS n FDA will not be able to monitor what is said directly to doctors n Proof of off-label marketing and kickbacks will continue to hinge on proof of intent n Qui tam cases will continue to be the primary enforcement tool THOMAS M. GREENE & HOFFMAN - BOSTON, MA 12

CONCLUSIONS n Government will continue to seek out whistleblowers by offering substantial rewards THOMAS M. GREENE & HOFFMAN - BOSTON, MA 13

RELATOR SHARE n NEURONTIN: ¨ 1 Relator, $27. 3 Million * n SEROSTIM: ¨ 5 Relators, $51. 8 Million * Includes statutory award of attorneys fees THOMAS M. GREENE & HOFFMAN - BOSTON, MA 14

THOMAS M. GREENE, ESQ 125 Summer Street, Suite 1410 - Boston, MA 02110 www. greenehoffman. com Phone: (617) 261 -0040 Fax: (617) 261 -3558 tgreene@greenehoffman. com 15
- Slides: 15