Fatty acid synthesis Prof S Kajuna Synthesis of














- Slides: 16

Fatty acid synthesis Prof. S. Kajuna Synthesis of fatty acids takes place in the cytoplasm and involves initiation of synthesis by the formation of acetoacetyl-ACP and then an elongation cycle where 2 carbon units are successively added to the growing chain. Fatty acid synthesis is rather the creation of fatty acids from acetyl-Co. A and malonyl-Co. A precursors through action of enzymes called fatty acid synthases. It is an important part of the lipogenesis process, which – together with glycolysis – functions to create fats from blood sugar in living organisms.

Acyl carrier protein (ACP) serves as a chaperone for the synthesis of fatty acids. The growing fatty acid chain is covalently bound to ACP during the entire synthesis of the fatty acid and only leaves the protein when it is attached to the glycerol backbone of the forming lipid. ACP is one of the most abundant proteins in the bacterial cell (60, 000 molecules per E. coli cell) which makes sense given the amount of lipid that must be synthesized to make an entire cell membrane. The formation of acetoacetyl-ACP can be catalyzed by a number of enzymes, but in all cases the starting substrate is acetyl-Co. A.

Once formed, acetoacetyl-ACP enters the elongation cycle for fatty acid synthesis. This cycle is the reverse of the β-oxidation of fatty acids discussed earlier. The first step in the elongation cycle is condensation of malonyl-Co. A with a growing acetoacetyl-ACP chain. This adds two carbons to the chain. The next three reactions use 2 NADPH to reduce the β-ketone and generate an acyl-ACP molecule two carbons longer than the original substrate. The acyl-ACP molecule continues through the cycle until the appropriate chain length is reached.

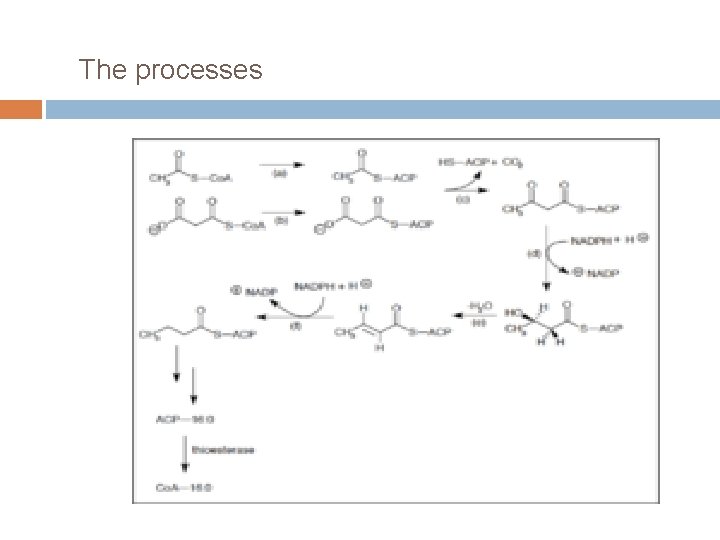
The processes

Much like β-oxidation, straight-chain fatty acid synthesis occurs via seven recurring reactions until the 16 -carbon palmitic acid is produced.

(a) Acetyl Co. A: ACP transacylase Activates acetyl Co. A for reaction with malonyl-ACP (b) Malonyl Co. A: ACP transacylase Activates malonyl Co. A for reaction with acetyl-ACP (c) 3 -ketoacyl-ACP synthetase Reacts priming acetyl-ACP with chain-extending malonyl-ACP. (d)3 -ketoacyl-ACP reductase Reduces the carbon 3 ketone to a hydroxyl group (e)3 -Hydroxyacyl ACP dehydrase Removes water (f)Enoyl-ACP reductase Reduces the C 2 -C 3 double bond.

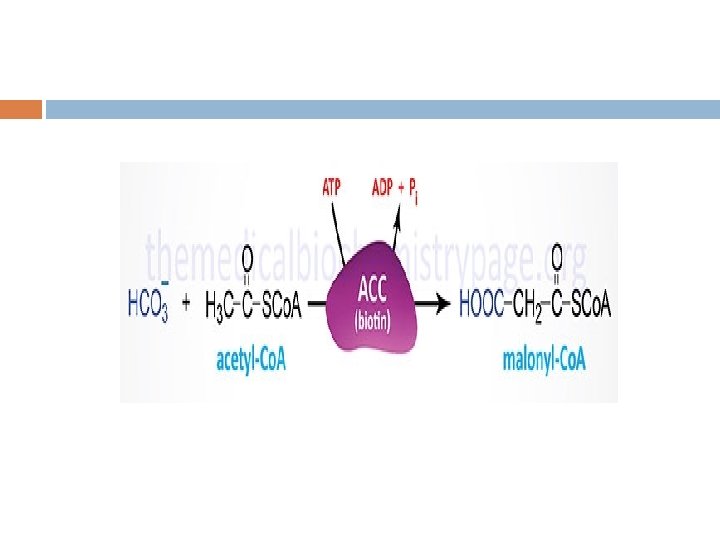
Regulation Acetyl-Co. A is formed into malonyl-Co. A by acetyl-Co. A carboxylase, at which point malonyl-Co. A is destined to feed into the fatty acid synthesis pathway. Acetyl-Co. A carboxylase is the point of regulation in saturated straight-chain fatty acid synthesis, and is subject to both phosphorylation and allosteric regulation. Regulation by phosphorylation occurs mostly in mammals, while allosteric regulation occurs in most organisms. Allosteric control occurs as feedback inhibition by palmitoyl-Co. A and activation by citrate. When there are high levels of palmitoyl-Co. A, the final product of saturated fatty acid synthesis, it allosterically inactivates acetyl-Co. A carboxylase to prevent a build-up of fatty acids in cells. Citrate acts to activate acetyl-Co. A carboxylase under high levels, because high levels indicate that there is enough acetyl-Co. A to feed into the Krebs cycle and produce energy.

The synthesis of malonyl-Co. A is the first committed step of fatty acid synthesis and the enzyme that catalyzes this reaction, acetyl. Co. A carboxylase (ACC), is the major site of regulation of fatty acid synthesis. Like other enzymes that transfer CO 2 to substrates, ACC requires a biotin co-factor. Acetyl-Co. A carboxylase is called an ABC enzyme due to the requirements for ATP, Biotin, and CO 2 for the reaction.


All of the reactions of fatty acid synthesis are carried out by the multiple enzymatic activities of FAS. Like fat oxidation, fat synthesis involves four primary enzymatic activities. These are (in order of reaction), β-ketoacyl. ACP synthase, β-ketoacyl-ACP reductase, 3 hydroxyacyl-ACP dehydratase and enoyl-Co. A reductase. The two reduction reactions require NADPH oxidation to NADP+.

The primary fatty acid synthesized by FAS is palmitic acid (palmitate). Palmitate is then released from the enzyme via the thioesterase activity of FAS. Once released, palmitate can then undergo separate elongation and/or unsaturation to yield other fatty acid molecules.

Elongation and Desaturation The fatty acid product released from FAS is palmitate (via the action of palmitoyl thioesterase) which is a 16: 0 fatty acid, i. e. 16 carbons and no sites of unsaturation. Elongation and unsaturation of fatty acids occurs in both the mitochondria and endoplasmic reticulum (microsomal membranes). The predominant site of these processes is in the ER membranes. Elongation involves condensation of acyl-Co. A groups with malonyl-Co. A. The resultant product is two carbons longer (CO 2 is released from malonyl-Co. A as in the FAS reaction) which undergoes reduction, dehydration and reduction yielding a saturated fatty acid extended in length by two carbon atoms. The reduction reactions of elongation require NADPH as co-factor just as for the similar reactions catalyzed by FAS. Mitochondrial elongation involves acetyl-Co. A units and is a reversal of oxidation except that the final reduction utilizes NADPH instead of FADH 2 as co-factor.

The desaturation of fatty acids occurs in the ER membranes as well. In mammalian cells fatty acid desaturation involves three broad specificity fatty acyl-Co. A desaturases (nonheme iron containing enzymes). These enzymes introduce unsaturation at C 5, C 6 or C 9. The names of these enzymes are Δ 5 eicosatrienoyl-Co. A desaturase (D 5 D), Δ 6 oleoyl(linolenoyl)-Co. A desaturase (D 6 D), and Δ 9 -stearoyl-Co. A desaturase (SCD).

Since these enzymes cannot introduce sites of unsaturation beyond C 9 they cannot synthesize either linoleate (18: 2Δ 9, 12) or linolenate (18: 3Δ 9, 12, 15). These fatty acids must be acquired from the diet and are, therefore, referred to as essential fatty acids. Linoleic is especially important in that it required for the synthesis of arachidonic acid. Arachindonate is a precursor for the eicosanoids (the prostaglandins, thromboxanes, and leukotrienes). It is this role of fatty acids in eicosanoid synthesis that leads to poor growth, wound healing and dermatitis in persons on fat free diets. Also, linoleic acid is a constituent of epidermal cell sphingolipids that function as the skins' water permeability barrier.

In humans, fatty acids are formed predominantly in the liver and lactating mammary glands, and, to a lesser extent, the adipose tissue. Most acetyl. Co. A is formed from pyruvate by pyruvate dehydrogenase in the mitochondria. Acetyl-Co. A produced in the mitochondria is condensed with oxaloacetate by citrate synthase to form citrate, which is then transported into the cytosol and broken down to yield acetyl-Co. A and oxaloacetate by ATP citrate lyase. Oxaloacetate in the cytosol is reduced to malate by cytoplasmic malate dehydrogenase, and malate is transported back into the mitochondria to participate in the Citric acid cycle. [

Desaturation of fatty acids involves a process that requires molecular oxygen (O 2), NADH, and cytochrome b 5. The reaction, which occurs in the endoplasmic reticulum, results in the oxidation of both the fatty acid and NADH. The most common desaturation reactions involve the placement of a double bond between carbons 9 and 10 (as in the conversion of palmitic acid to palmitoleic acid and the conversion of stearic acid to oleic acid, facilitated by the action of Δ 9 -desaturase). Other positions that can be desaturated in humans include carbon 4, 5, and 6, via Δ 4 -, Δ 5 -, and Δ 6 desaturases, respectively.