Fatal Attraction Ionic Bonding Attractions from protons and

Fatal Attraction -Ionic Bonding


• Attractions from protons and e- cause the e- to transfer
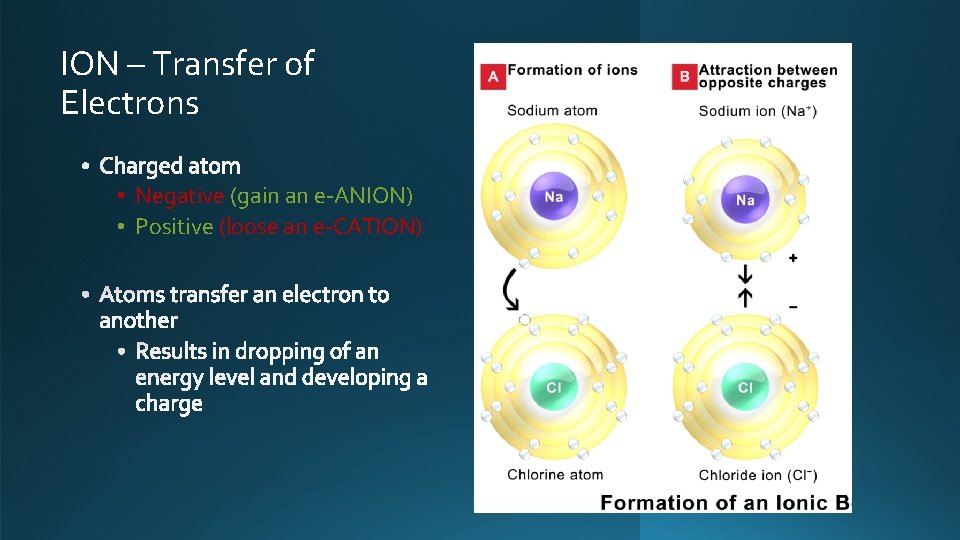
ION – Transfer of Electrons • Negative (gain an e-ANION) • Positive (loose an e-CATION)

• Most common COMPOUNDS are formed by the transferred e- from GROUP 1. • Which group would these elements most likely join with? • GROUP 7 because their valence shell is nearly complete reaching STABILITY! • Draw and example • Draw electron dot diagrams for the following compounds include the charges in the chemical formula • Na. Cl • KI • P 558 -559
Practice • Na. Cl • KI 1. Identify the number of valence e- for each element in the chemical formula 2. Which element will transfer its e 3. Create an electron dot diagram for each compound 4. Include the charges of the ions

• Most common COMPOUNDS are formed by the transferred e- from GROUP 1. • Which group would these elements most likely join with? • GROUP 7 because their valence shell is nearly complete reaching STABILITY! • Draw electron dot diagrams for the following compounds include the charges in the chemical formula • Na+Cl • K+I- • Circle and highlight positive and negative ions • What is the total charge (net charge) of the compound? • P 558 -559


• Negative (gain an e-ANION) • Positive (loose an e-CATION) • Ionic bonding
- Slides: 9