Fat Soluble Vitamins Water Soluble Vitamins THE VITAMINS

Fat Soluble Vitamins Water Soluble Vitamins THE VITAMINS Prepared by: Mona Abo Hasera

CHARACTERISTICS OF VITAMINS Vitamins are micronutrients Vitamins are essential. The roles they play in the body are very important. Most vitamins are obtained from the foods we eat. Some are made by bacteria in the intestine There is no perfect food that contains all the vitamins in the right amount. Vitamins are non-energy producing Very small amounts are needed by the body (>1 gm) Very small amounts are contained in foods. They do not contain kcalories. Vitamins are classified according to how soluble they are in fat or water.
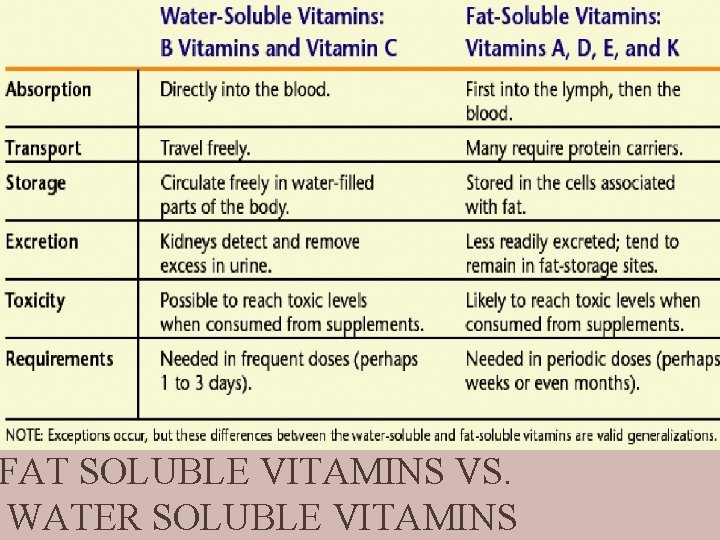
FAT SOLUBLE VITAMINS VS. WATER SOLUBLE VITAMINS

A, D, E, K FAT-SOLUBLE VITAMINS

FAT-SOLUBLE VITAMINS found in fats and oils require bile for absorption enter the lymph, then the blood held and stored in fatty tissues Needed in small amounts may reach toxic levels not readily excreted
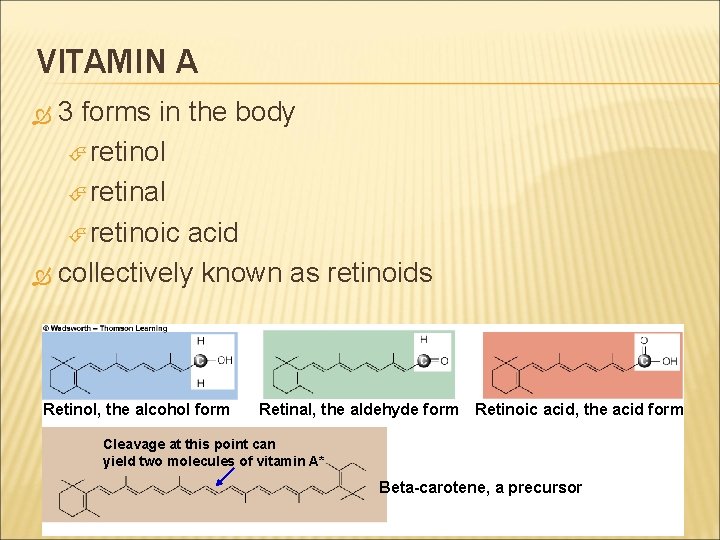
VITAMIN A 3 forms in the body retinol retinal retinoic acid collectively known as retinoids Retinol, the alcohol form Retinal, the aldehyde form Retinoic acid, the acid form Cleavage at this point can yield two molecules of vitamin A* Beta-carotene, a precursor

VITAMIN A precursor: beta- carotene derived from plant foods can split and form retinol in intestine and liver

VITAMIN A FUNCTION vision maintain epithelial tissue and skin support reproduction and growth Immune system Bone development
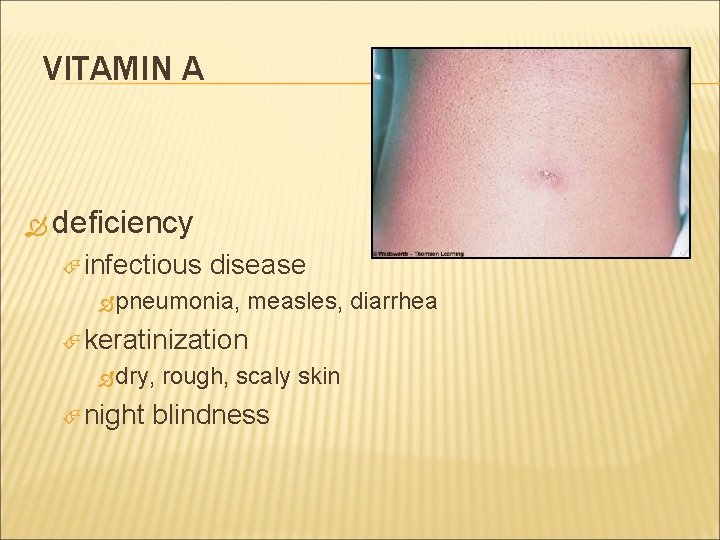
VITAMIN A deficiency infectious disease pneumonia, measles, diarrhea keratinization dry, night rough, scaly skin blindness

Vitamin A Sources Beta-carotene Dark leafy green vegetables, spinach, broccoli Deep orange veggies Carrots, pumpkin, squash, sweet potato Deep orange fruits Apricots, cantaloupe

Retinol Fortified milk, butter cheese, cream Fortified margarine Eggs Liver
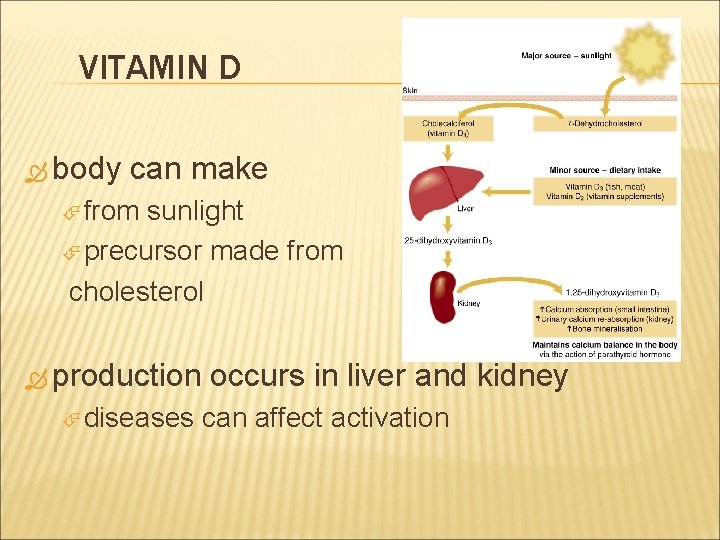
VITAMIN D body can make from sunlight precursor made from cholesterol production diseases occurs in liver and kidney can affect activation

VITAMIN D FUNCTION part of the bone-making/maintenance team maintains blood concentrations of Ca & P Mineralization of bones raises blood calcium and phosphorus by increasing absorption from digestive tract withdrawing calcium from bones stimulating retention by kidneys deficiencies ultimately creates a calcium deficiency rickets, osteomalacia

VITAMIN D sources fortified food: milk, margarine, cereals, beef, eggs sun storage from the summer does not last the winter

VITAMIN E antioxidant defender against free radicals polyunsaturated fatty acids may reduce the risk of heart disease deficiencies rare erythrocyte hemolysis

VITAMIN E widespread easily in food destroyed by heat processing
VITAMIN K aids in blood clotting and bone mineralization deficiency causes hemorrhagic disease sources made by bacteria in GI tract absorbed and stored in liver

liver is also high in vitamin K
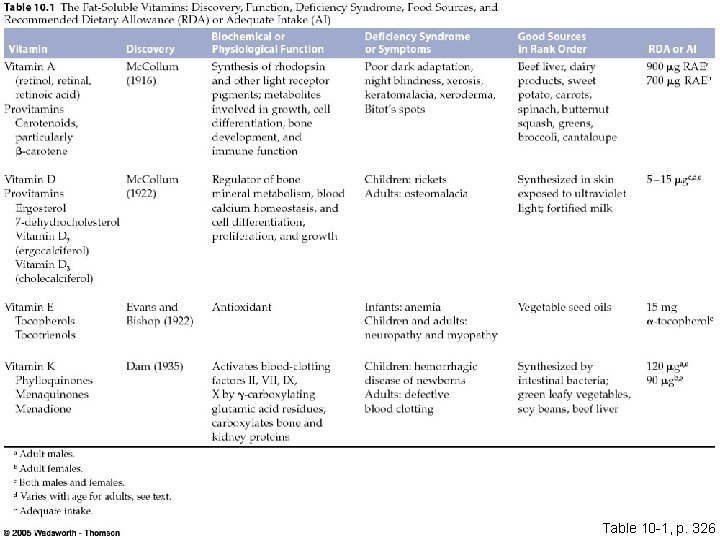
Table 10 -1, p. 326

B complex , c WATER-SOLUBLE VITAMINS

WATER SOLUBLE VITAMINS The B-complex vitamins are often associated with giving a person more energy. This is due to the fact that these vitamins each play different roles with energy metabolism in the body. When they are present in the body, they allow energy to be used more readily by the body. Since these vitamins are water soluble, they are not stored in the body like fat soluble vitamins. They dissolve in water and are excreted from the body in urine. Therefore, it is important to consume foods rich in these vitamins each day in order to fulfill the body’s need.

B COMPLEX VITAMINS Co-enzymes (activate enzymes) Found in the same foods Single Act deficiency rare together in metabolism Metabolic pathways used by protein, carbohydrate, and fat

B COMPLEX VITAMINS Thiamin (B 1) Riboflavin (B 2) Niacin (B 3) Pantothenic Acid Biotin Pyridoxine (B 6) Folate Vitamin B-12

B COMPLEX PRIMARY FUNCTIONS Energy metabolism Thiamin (B-1), Riboflavin (B-2), Niacin (B-3), Pyridoxine (B-6), Biotin, Pantothenic Acid Red blood cell synthesis Folate, B 12 Homocysteine Folate, metabolism B 12, B 6

VITAMIN C Synthesized by most animals (not by humans) Decrease absorption with high intakes Excess excreted

FUNCTIONS OF VITAMIN C Reducing agent (antioxidant) Iron absorption (enhances) Synthesis of collagen Immune functions Does not prevent colds, but may reduce duration of symptoms by a day Wound healing

VITAMIN C DEFICIENCY: HISTORY OF SCURVY Vitamin C (ascorbic acid) deficiency leads to scurvy, a disease characterized by weakness, small hemorrhages throughout the body that cause gums and skin to bleed, and loosening of the teeth. Sailors that were out at sea for months on end would often develop scurvy unless the captain had the foresight to pack limes and other citrus fruits.

DEFICIENCY OF VITAMIN C Scurvy Deficient diet for 20 -40 days Fatigue, pinpoint hemorrhages Bleeding gums and joints. Hemorrhages Associated with poverty; macrobiotic diet
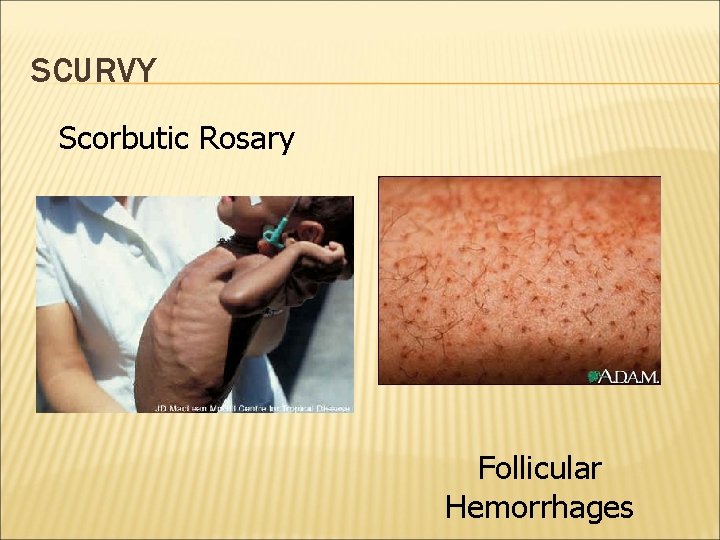
SCURVY Scorbutic Rosary Follicular Hemorrhages

FOOD SOURCES OF VITAMIN C Citrus fruit Potato Green pepper Cauliflower Broccoli Strawberry Romaine lettuce Spinach Easily lost through cooking Sensitive to heat Sensitive to iron, copper, oxygen

VITAMIN C EXCESS Hemochromatosis Vitamin Oxalate C enhances iron absorption kidney stones Erodes tooth enamel

VITAMIN C DEFICIENCY In the U. S. , deficiency is seen mostly in alcoholic persons with poor diets and older persons who eat poorly (no fresh fruits and vegetables)

DETERMINATION OF VITAMIN C (ASCORBIC ACID) CONCENTRATION BY A REDOX TITRATION WITH POTASSIUM IODATE (KIO 3) 33

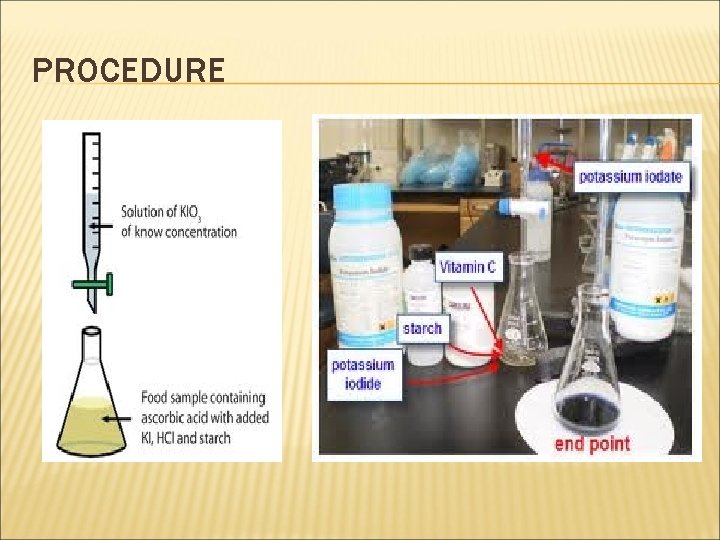
EXPERIMENT Principle 1. KIO 3 is used as a titrant and it is added to an ascorbic acid solution that contains a strong acid and potassium iodide (KI). 2. KIO 3 reacts with KI, liberating molecular iodine (I 2): KIO 3 + 5 KI + 6 H+ → 3 I 2 + 6 K+ + 3 H 2 O (1) C 6 H 8 O 6 + I 2 → C 6 H 6 O 6 + 2 I- + 2 H+ (2) 34

PROCEDURE 1. Pipette 25 ml of the provided ascorbic acid solution into a 250 ml conical flask, 2. Add 4 ml of 2 M HCl, 3. Add 5 ml of potassium iodide (KI) solution and 3 ml starch solution. 4. Then titrate with the standard potassium iodate (KIO 3) solution until the solution turns intense blue. Write down the standard potassium iodate (KIO 3) solution volume. 5. Pipette 25 ml of an unknown ascorbic acid sample, a kind of juice, into a 250 ml conical flask, then follow the same procedure of steps 1 -4 and write down the volume of the standard KIO 3 solution determine the concentration (mol/ml) of ascorbic acid in the selected sample. 35
PROCEDURE
- Slides: 36