Faster Better Biotech with Ellyn and FSEBS 4

Faster, Better Biotech with Ellyn and FSE/BS 4 NM Lab Skills and Implementation Support – Electrophoresis and Spectrophotometry Ellyn Daugherty, Ellyn@Biotech. Ed. com, 650 -400 -9424 Terri Postlethwait, FSE terri. postlethwait@thermofisher. com Vijay Varma, FSE vijay. varma@thermofisher. com Jessica Haverkock, FSE jessica. haverkock@thermofisher. com

Faster, Better Biotech Electrophoresis and Spectrophotometry Friday 10/3, 5 -8 pm • Meet and Greet in the Cafeteria, quick snack and refreshment • DNA Electrophoresis Tricks and Tips • Review of DNA electrophoresis using Restriction Digestion samples • Prepare Restriction Digestion samples • Load, run, visualize samples on horizontal gel Saturday 10/4, 9 -4 pm • Protein Electrophoresis Tricks and Tips • Review of Sample Prep and Dilutions • Prepare PAGE samples and Set-up PAGE gel in gel box • Load, run, visualize samples on vertical gel • Lunch • Spectrophotometry, Spectronic® 200 Features and Use • Prepare diluted samples for spec and collect absorbance data

Friday = DNA Electrophoresis Tricks and Tips Horizontal Gel Electrophoresis Components: • Gel box - how many trays, how gels are poured? • Power Supply – maximum current? • Gel Preparation – concentration of agarose and what kind of buffer? – DNA stain included? • Buffer – 1 X TAE or 1 X LB? • Agarose Concentration – 0. 8%, 1. 0%, or 2%? • Sample Prep and Loading – amount of DNA in sample, amount to load? • Gel Running – voltage or current ? • Sample Visualization – staining method? In the BS 4 NM lab manual, there at least 3 introductory DNA gels to run: Lab 4 j, Lab 4 k, and Lab 8 b*

Agarose Gel Preparation Need 2 equations, one for the agarose and one for the buffer: • % mass/volume for 0. 8% agarose gel in 1 X buffer ________g/m. L ______________g Conc. desired (decimal) X volume desired = mass of solute (agarose) needed • Dilution of buffer to 1 X is the C 1 V 1=C 2 V 2 equation _________ __________ Starting conc. X Volume needed of stock = Final Conc desired X Final Volume desired (X) (m. L)

Agarose Gel Preparation Need 2 equations, one for the agarose and one for the buffer: • % mass/volume for 0. 8% agarose gel in 1 X buffer For 100 m. L of 0. 8% agarose gel: 0. 008 g/m. L X 100 m. L = ? g Conc. desired (decimal) volume desired mass of solute (agarose) needed • Dilution of buffer to 1 X is the C 1 V 1=C 2 V 2 equation To make 300 m. L of 1 X buffer from 40 X buffer concentrate: 40 X X (? m. L) = Starting conc. Volume needed of stock 1 X 300 m. L Final Conc desired X Final Volume desired

Lab 8 b – Restriction Digestion of p. Amy 2014 Plasmid In the Lab manual (page 173): The p. Amylase 2014 plasmid has a size of approximately 4300 bp. It contains a single restriction site for the Hind. III restriction enzyme. It also contains a single restriction site for the Eco. RI restriction enzyme. The products of the restriction enzyme digestion can be seen on an agarose gel.

Kit Lab 8 b (p. 4) Restriction Digestion of p. Amy 2014 Plasmid

Kit Lab 8 b (p. 5) Restriction Digestion of p. Amy 2014 Plasmid

Kit Lab 8 b (p. 6)
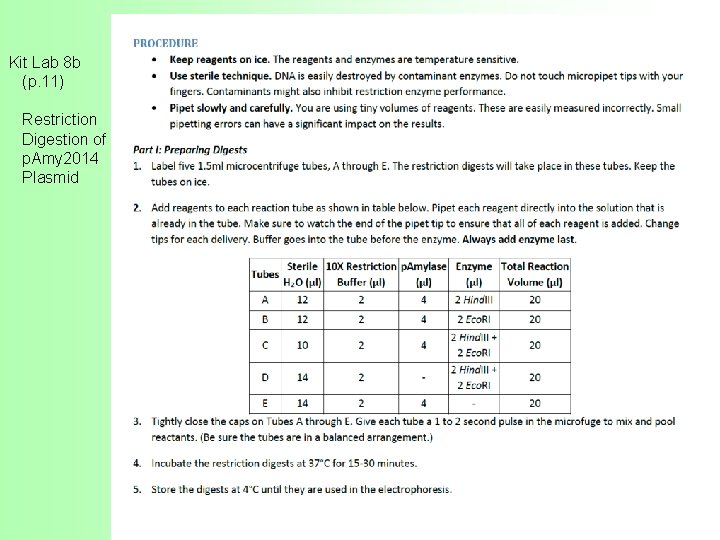
Kit Lab 8 b (p. 11) Restriction Digestion of p. Amy 2014 Plasmid

Kit Lab 8 b (p. 12) Restriction Digestion of p. Amy 2014 Plasmid

Saturday AM = PAGE Electrophoresis Tricks & Tips PAGE Gel Electrophoresis Components: • PAGE = Poly. Acrylamide Gel Electrophoresis • Gel box - Use 1 or 2 premade (purchased gels)/gel box • Power Supply – maximum current? Gel running voltage or current? • Gel concentration of PAGE is important and what kind of buffer? – protein stain in gel? • Buffer – 1 X PAGE/SDS Running Buffer or Laemmli Buffer • Sample Prep & Loading – amount of protein in sample, amount to load, how to dilute? • Sample Visualization – UV stain in gel &/or Coomassie Blue Staining and white light box In the BS 4 NM lab manual, there at least 3 introductory PAGE gels to run: Lab 5 f*, Lab 5 g, and Lab 6 f
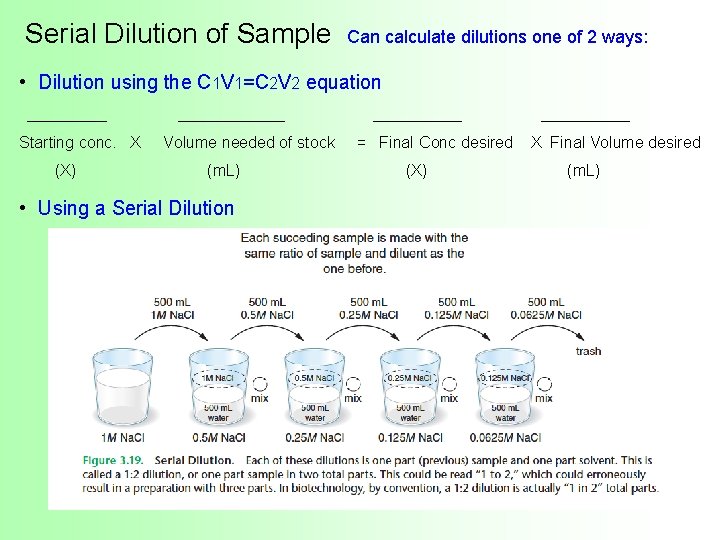
Serial Dilution of Sample Can calculate dilutions one of 2 ways: • Dilution using the C 1 V 1=C 2 V 2 equation _________ __________ Starting conc. X Volume needed of stock = Final Conc desired X Final Volume desired (X) (m. L) • Using a Serial Dilution (X) (m. L)

Lab 5 f – Characterizing Proteins with PAGE (page 114 ) : What structural characteristics of BSA and cellulase can be determined from running samples on a SDS-PAGE gel? How is the resolution of the polypeptide bands on the gel affected by the concentration of the sample loaded?

Kit Lab 5 f (p. 4)

Kit Lab 5 f (p. 5) Kit Lab 5 f (p. 7)

Kit Lab 5 f (p. 10) Draw >>> a picture of the dilution

Kit Lab 5 f (p. 11)

Kit Lab 5 f (p. 12)

Saturday PM = Spectrophotometry Tricks & Tips Spectrophotometry Basics • Must pick a wavelength where the sample can be “seen” by the spec. • Concentration must be within the range of minimum and maximum A. In the BS 4 NM lab manual, there at several spec labs in Chapter 3 and 7: Lab 7 d/7 g*

Spectronic® 200
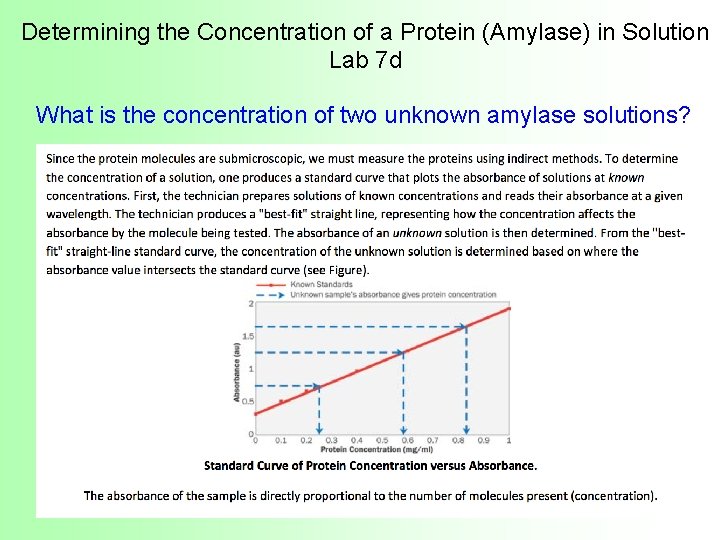
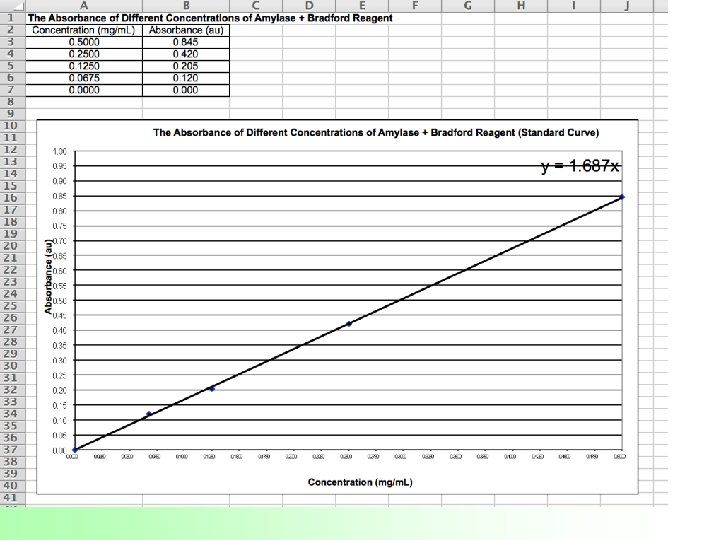
Determining the Concentration of a Protein (Amylase) in Solution Lab 7 d What is the concentration of two unknown amylase solutions?

Kit Lab 7 d (p. 9)

Kit Lab 7 d (p. 10)

Kit Lab 7 d (p. 11)
- Slides: 26