FARMAK a s CZECH REPUBLIC INTRODUCTION FARMAK a

FARMAK, a. s. CZECH REPUBLIC

INTRODUCTION FARMAK, a. s. ü Czech, privately owned, independent middle-sized company with the tradition of industrial chemical production dates back to the year 1934. ü Globally active ü Flexible, reliable and customer oriented partner ü Focused on R&D and manufacturing of: ü niche API´s ü advanced intermediates ü Supportive in dossier development ü Customer and employees oriented – The Essential is Inside Farmak, a. s. 2
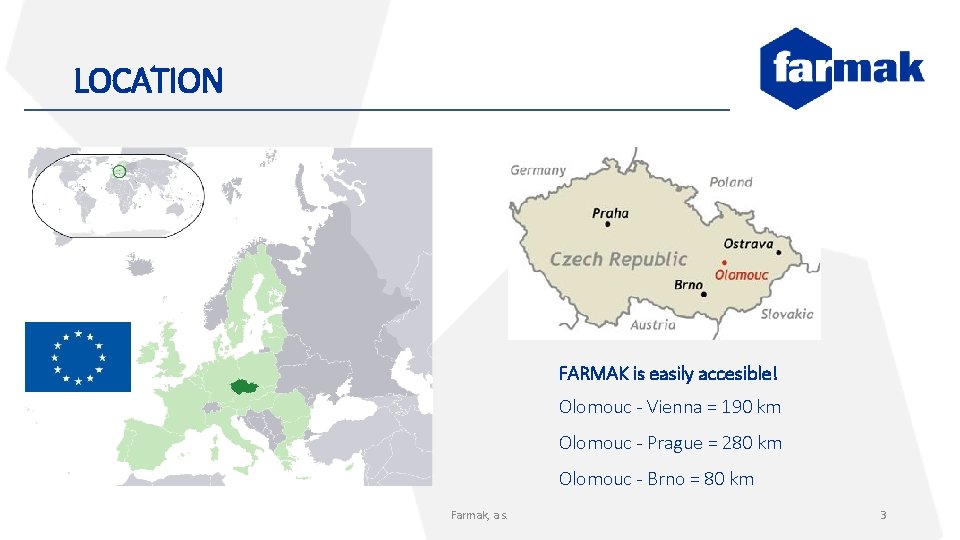
LOCATION FARMAK is easily accesible! Olomouc - Vienna = 190 km Olomouc - Prague = 280 km Olomouc - Brno = 80 km Farmak, a. s. 3

HISTORY ü 1934 - start of industrial chemical production ü 1952 - establishment of state-owned company Farmakon (member of Spofa) ü 1992 - registration of joint-stock company FARMAK ü 1996 - Farmakon acquired by FARMAK (privatisation) ü 2004 - new R&D and QA/QC centre construction ü 2008 - installation of new c. GMP kilolab ü 2014 - new Hall of Final Operations (HAFO) construction ü 2015 - new multipurpose production line installed ü 2016 - climatic chambers with water cooling system - particle size distribution measurement on Malvern instrument ü 2017 - comprehensive Drying and Homogenization Technology Line Farmak, a. s. 4
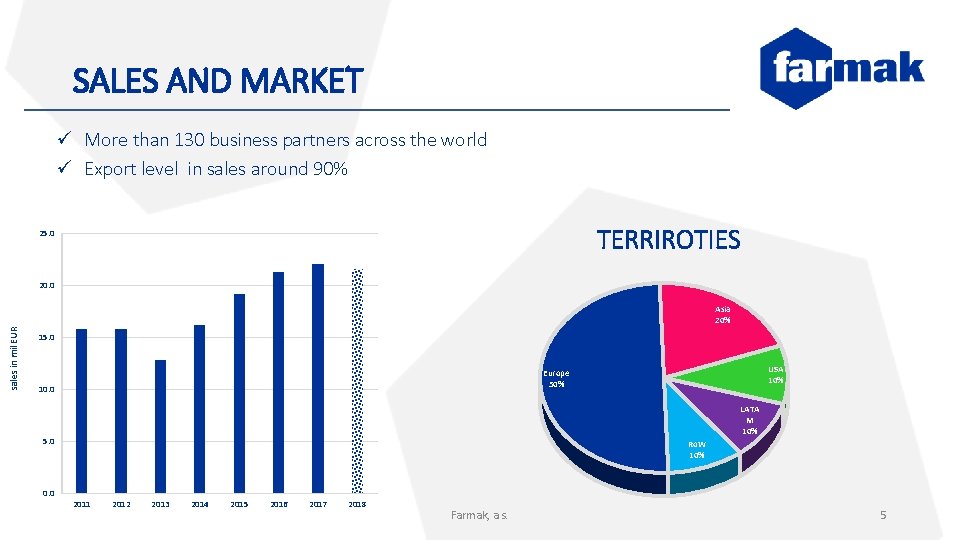
SALES AND MARKET ü More than 130 business partners across the world ü Export level in sales around 90% TERRIROTIES 25. 0 20. 0 sales in mil EUR Asia 20% 15. 0 10. 0 LATA M 10% 5. 0 0. 0 USA 10% Europe 50% Ro. W 10% 2011 2012 2013 2014 2015 2016 2017 2018 Farmak, a. s. 5
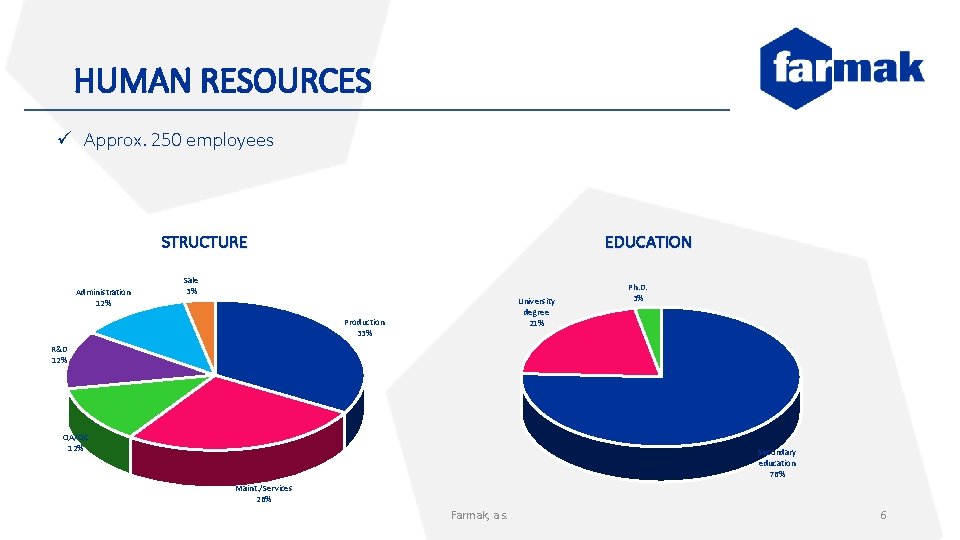
HUMAN RESOURCES ü Approx. 250 employees STRUCTURE Administration 12% EDUCATION Sale 3% University degree 21% Production 33% Ph. D. 3% R&D 12% QA/QC 12% Secondary education 76% Maint. /Services 26% Farmak, a. s. 6

QUALITY MANAGEMENT SYSTEM üCompliance with international c. GMP standards: EU GMP Eudralex Volume 4, Part 2; ICH Q 7; US FDA CFR’s and customer’s Quality Assurance Agreements üInspected by authorities on regular basis Czech State Institute for Drug Control US Food and Drug Administration üApproved by üISO 9 001 & ISO 14 001 International Standarts Certified Integrated System of QC and Environmental Protection üDMF & CEP Regulatory Documentation available Farmak, a. s. 7

CHEMICAL EXPERTISE üChemistry of heterocyclic compounds üGrignard reactions üVarious condensations üFriedel - Crafts reactions üHalogenations üOxidations üCatalytic hydrogenations üEsterifications üRacemic mixture separations üReductions (Zn, catalytic hydrogenations) Farmak, a. s. 8

PRODUCTION FACILITIES üFacilities area: approx. 36, 000 m 2 ü 2 GMP production halls (one is FDA approved) Facility is ready to expand! üHall of Final Operations: • • milling micronizing sieving packaging üSupporting facilities: • warehousing • waste water treatment • water purification Farmak, a. s. 9

PRODUCTION HALLS reactors Pfaudler ü Glass-lined, stainless steel jacketed reactors (typical volume is 1, 600 litres) ü Total reactor capacity: 100 m 3 ü Batch size 5 - 250 kg ü Reaction temperatures: between -20 and +300 °C ü Reaction pressure: between -80 and +300 k. Pa ü Product separation - centrifuges, filters, filter/driers (some of them of Hastelloy C 22) ü Drying equipment (some of them of Hastelloy C 22 Farmak, a. s. bag filters MAAP 10

KILOLAB highest level of c. GMP requirements ü Büchi Chem. Reactor CR: 2 x 60 L, 1 x 15 L ü Cryogenic technology (down to -90°C) ü Glove box ü Mobile pressure filter ü HVAC system ü Utility systems: non-water heating/cooling medium process nitrogen, process vacuum, hot water ü Protected area for raw material dispensing ü 0. 2 – 5 kg batches Farmak, a. s. 11

HALL OF FINAL OPERATIONS ü Milling/micronization ü Sieving ü Custom packaging ü HVAC system Farmak, a. s. 12

R&D AND QA/QC CENTRE ü Analytical Centre – QC Department • production sample service – raw materials, intermediates, final samples • documentation – SOP’s, analytical instructions, Co. A’s • standards, stability studies, validation of methods, water and air monitoring ü R&D Analytical Department • structural analyses • purity analyses/impurity profile • development of analytical methods ü Instrumentation • LCMS, GC, HPLC/TLC • Capillary Electrophoresis • NMR, UV/VIS a IR Spectrometry, XRay • DSC • Laser Particle Size Analyses (Cilas+Malvern) Farmak, a. s. 13

ECOLOGY AND SAFETY üEnvironmental policy in line with FARMAK’s strategy üInvolvement in the national System of Assistance in Accidents Related to Transportation of Dangerous Materials üCertified integrated system ISO 9001 and ISO 14001 üOwn waste water treatment plant üRESPONSIBLE CARE certificate holder for 20 years üApproved by SEDEX – Ethical and Responsible Supply Chain since 2015, site reference ZS 1073996 Farmak, a. s. 14

CONTACTS Feel free to contact us through our websites www. farmak. cz Phone: +420 587 430 111, +420 585 547 111 Fax: +420 585 231 424, +420 585 231 735 E-mail: Address: ceo@farmak. cz FARMAK, a. s. Na vlčinci 16/3, Klášterní Hradisko 779 00 OLOMOUC, CZECH REPUBLIC You can also follow us on Linked. In FARMAK, a. s
- Slides: 15