Falcon A SecondGeneration Absorb Falcon Design and Development

Falcon* - A Second-Generation Absorb: Falcon Design and Development Plans Richard JRapoza, Ph. D Abbott Vascular *Product currently in development. Not available for sale. See important Safety Information referenced within. Not to be reproduced, distributed or excerpted. © 2018 Abbott. All rights reserved. SE 2944958 Rev. B

Disclosure Statement of Financial Interest Within the past 12 months, I or my spouse/partner have had a financial interest/arrangement or affiliation with the organization(s) listed below. Affiliation/Financial Relationship • Full time employment Company • Abbott Vascular 2 of 18 See important Safety Information referenced within. Not to be reproduced, distributed or excerpted. © 2018 Abbott. All rights reserved. SE 2944958 Rev. B
Three things move every innovation forward Availability of Clinical Data Rapid Product Iteration Refining Implantation Strategy BRSIS NODIFFERENT See important Safety Information referenced within. Not to be reproduced, distributed or excerpted. © 2018 Abbott. All rights reserved. SE 2944958 Rev. B 3 of 18
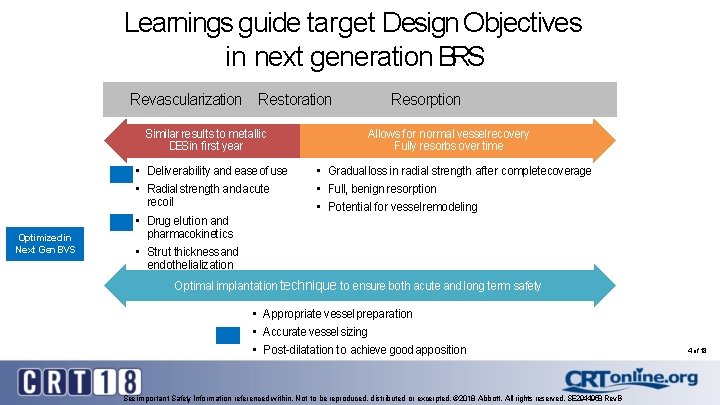
Learnings guide target Design Objectives in next generation BRS Revascularization Restoration Similar results to metallic DESin first year Optimized in Next Gen BVS • Deliverability and ease of use • Radial strength and acute recoil • Drug elution and pharmacokinetics • Strut thickness and endothelialization Resorption Allows for normal vessel recovery Fully resorbs over time • Gradual loss in radial strength after completecoverage • Full, benign resorption • Potential for vessel remodeling Optimal implantation technique to ensure both acute and long term safety • Appropriate vessel preparation • Accurate vessel sizing • Post-dilatation to achieve good apposition See important Safety Information referenced within. Not to be reproduced, distributed or excerpted. © 2018 Abbott. All rights reserved. SE 2944958 Rev. B 4 of 18
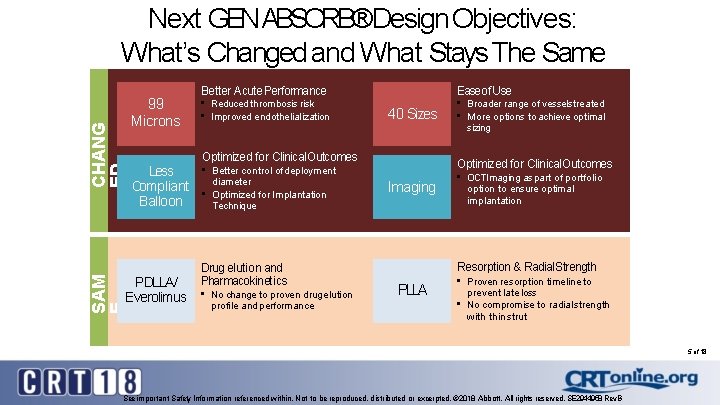
CHANG ED Next GENABSORB®Design Objectives: What’s Changed and What Stays The Same 99 Microns • Reduced thrombosis risk • Improved endothelialization Ease of Use 40 Sizes Optimized for Clinical. Outcomes Less Compliant Balloon PDLLA/ Everolimus SAM E Better Acute Performance • Better control of deployment diameter • Optimized for Implantation Technique Drug elution and Pharmacokinetics • No change to proven drugelution profile and performance • Broader range of vesselstreated • More options to achieve optimal sizing Optimized for Clinical. Outcomes Imaging • OCTImaging as part of portfolio option to ensure optimal implantation Resorption & Radial. Strength PLLA • Proven resorption timeline to prevent late loss • No compromise to radial strength with thin strut 5 of 18 See important Safety Information referenced within. Not to be reproduced, distributed or excerpted. © 2018 Abbott. All rights reserved. SE 2944958 Rev. B
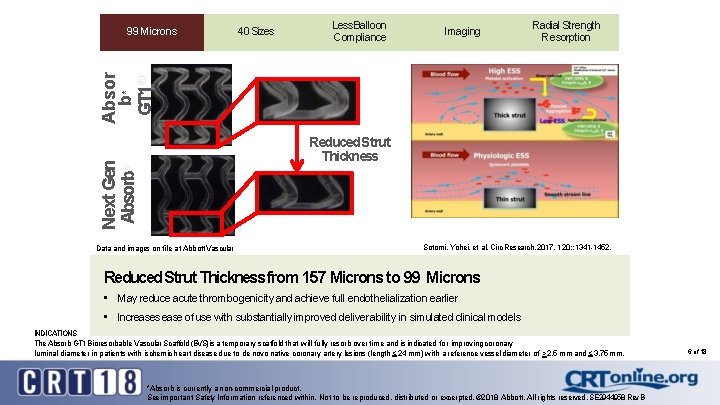
Sizes 9940 Microns Less Balloon 99 Microns Compliance Imaging Radial Strength Resorption Absor b* GT 1 ® 99 Microns Next Gen Absorb® Reduced Strut Thickness Data and images on file at Abbott Vascular Sotomi, Yohei, et al. Circ Research. 2017, 120: : 1341 -1452. Reduced Strut Thickness from 157 Microns to 99 Microns • May reduce acute thrombogenicity and achieve full endothelialization earlier • Increases ease of use with substantially improved deliverability in simulated clinical models INDICATIONS The Absorb GT 1 Bioresorbable Vascular Scaffold (BVS) is a temporary scaffold that will fully resorb over time and is indicated for improving coronary luminal diameter in patients with ischemic heart disease due to de novo native coronary artery lesions (length < 24 mm) with a reference vessel diameter of > 2. 5 mm and < 3. 75 mm. *Absorb is currently a non-commercial product. See important Safety Information referenced within. Not to be reproduced, distributed or excerpted. © 2018 Abbott. All rights reserved. SE 2944958 Rev. B 6 of 18
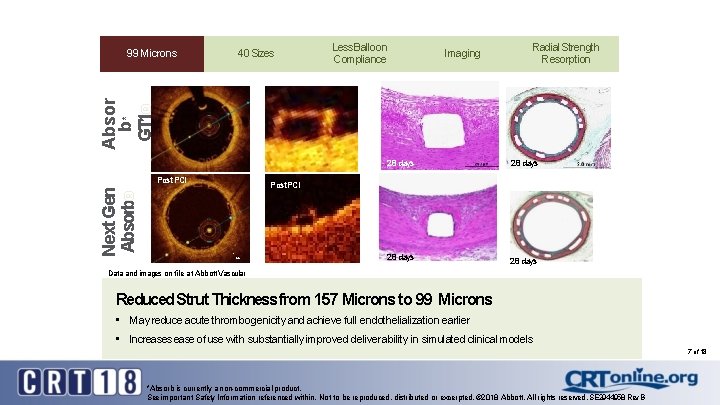
Sizes 9940 Microns Less Balloon 99 Microns Compliance Imaging Radial Strength Resorption Absor b* GT 1® 99 Microns 28 days Next Gen Absorb® Post PCI 28 days Post PCI 1 mm 28 days Data and images on file at Abbott Vascular Reduced Strut Thickness from 157 Microns to 99 Microns • May reduce acute thrombogenicity and achieve full endothelialization earlier • Increases ease of use with substantially improved deliverability in simulated clinical models 7 of 18 *Absorb is currently a non-commercial product. See important Safety Information referenced within. Not to be reproduced, distributed or excerpted. © 2018 Abbott. All rights reserved. SE 2944958 Rev. B
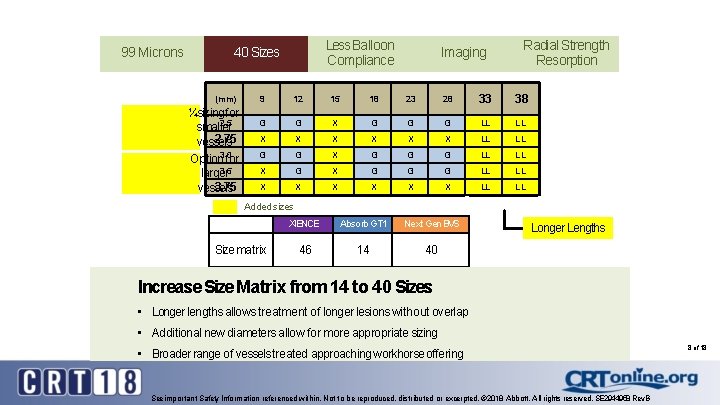
99 Microns Less Balloon 99 Microns Compliance 99 40 Microns Sizes (mm) ¼sizing for 2. 5 smaller 2. 75 vessels Option 3. 0 for 3. 5 larger 3. 75 vessels Imaging Radial Strength Resorption 9 12 15 18 23 28 33 38 G G X G G G LL LL X X X LL LL G G X G G G LL LL X X X X LL LL Added sizes Size matrix XIENCE Absorb GT 1 Next Gen BVS 46 14 40 Longer Lengths Increase Size Matrix from 14 to 40 Sizes • Longer lengths allows treatment of longer lesions without overlap • Additional new diameters allow for more appropriate sizing • Broader range of vessels treated approaching workhorse offering See important Safety Information referenced within. Not to be reproduced, distributed or excerpted. © 2018 Abbott. All rights reserved. SE 2944958 Rev. B 8 of 18
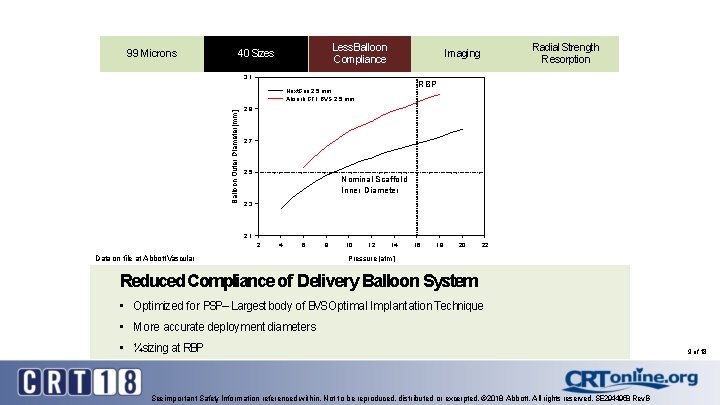
99 Microns Less Balloon 99 Microns Compliance Sizes 9940 Microns 3. 1 RBP Next. Gen 2. 5 mm Absorb GT 1 BVS 2. 5 mm Balloon Outer Diameter [mm] Radial Strength Resorption Imaging 2. 9 2. 7 2. 5 Nominal Scaffold Inner Diameter 2. 3 2. 1 2 4 6 Data on file at Abbott Vascular 8 10 12 14 16 18 20 22 Pressure [atm] Reduced Compliance of Delivery Balloon System • Optimized for PSP– Largest body of BVSOptimal Implantation Technique • More accurate deployment diameters • ¼sizing at RBP See important Safety Information referenced within. Not to be reproduced, distributed or excerpted. © 2018 Abbott. All rights reserved. SE 2944958 Rev. B 9 of 18
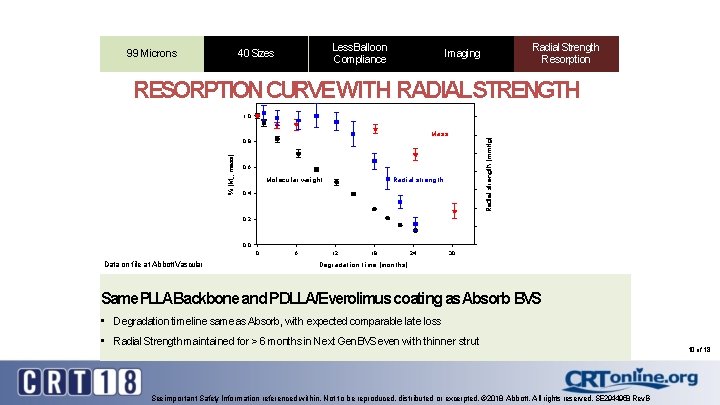
99 Microns Less Balloon 99 Microns Compliance Sizes 9940 Microns Radial Strength Resorption Imaging RESORPTION CURVEWITH RADIALSTRENGTH 1. 0 Radial strength [mm. Hg] Mass % [Mn, mass] 0. 8 0. 6 Molecular weight Radial strength 0. 4 0. 2 0. 0 0 Data on file at Abbott Vascular 6 12 18 24 30 Degradation time [months] Same PLLABackbone and PDLLA/Everolimus coating as Absorb BVS • Degradation timeline same as Absorb, with expected comparable late loss • Radial Strength maintained for > 6 months in Next Gen BVS even with thinner strut See important Safety Information referenced within. Not to be reproduced, distributed or excerpted. © 2018 Abbott. All rights reserved. SE 2944958 Rev. B 10 of 18
99 Microns Sizes 9940 Microns Less Balloon 99 Microns Compliance Imaging LEADING BVS FOROPTIMALOUTCOMES Radial Strength Resorption ENHANCEDVISUALIZATION & DIAGNOSTICS Accuracy and Precision are a MUST for BRSimplantation • Accurate vessel sizing and morphology assessment PREimplantation • Precise size selection and post-dilation based on OCTintraluminal data Data and images on file at Abbott Vascular See important Safety Information referenced within. Not to be reproduced, distributed or excerpted. © 2018 Abbott. All rights reserved. SE 2944958 Rev. B 11 of 18
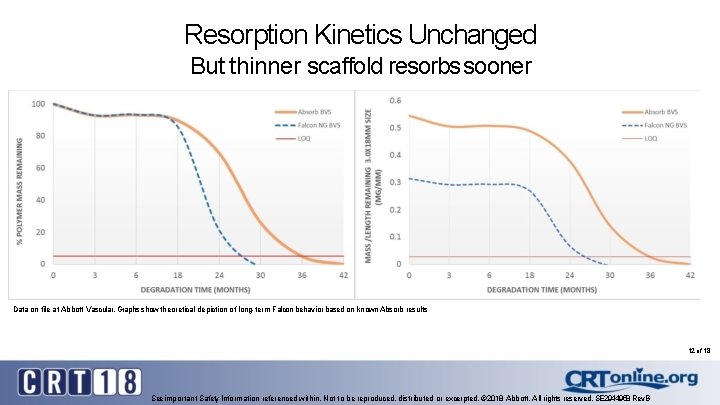
Resorption Kinetics Unchanged But thinner scaffold resorbs sooner Data on file at Abbott Vascular. Graphs show theoretical depiction of long-term Falcon behavior based on known Absorb results 12 of 18 See important Safety Information referenced within. Not to be reproduced, distributed or excerpted. © 2018 Abbott. All rights reserved. SE 2944958 Rev. B

Next Steps • Drive Falcon technical development to completion • Examine results in the ABSORB®family of trials to: – Confirm with greater volume of data that event rates are lower after complete resorption – Confirm that implantation technique has apositive influence on long term outcomes 13 of 18 See important Safety Information referenced within. Not to be reproduced, distributed or excerpted. © 2018 Abbott. All rights reserved. SE 2944958 Rev. B

Important Safety Information Absorb. GT 1 TM Bioresorbable Vascular Scaffold System INDICATIONS The Absorb GT 1 Bioresorbable Vascular Scaffold (BVS) is a temporary scaffold that will fully resorb over time and is indicated for improving coronary luminal diameter in patients with ischemic heart disease due to de novo native coronary artery lesions (length ≤ 24 mm) with a reference vessel diameter of ≥ 2. 5 mm and ≤ 3. 75 mm. CONTRAINDICATIONS The Absorb GT 1 BVS System is contraindicated for use in: • Patients who cannot tolerate, including allergy or hypersensitivity to, procedural anticoagulation or the post -procedural antiplatelet regimen. • Patients with hypersensitivity or contraindication to everolimus or structurally-related compounds, or known hypersensitivity to scaffold components (poly(L-lactide), poly(D, L-lactide), platinum) or with contrast sensitivity. WARNINGS • For single use only. Do not resterilize or reuse. Note the product "Use by" date on the package. • Careful assessment of the target lesion reference vessel diameter and selection of the appropriate scaffold diameter relative to the target lesion reference vessel diameter are required to minimize potential damage to the scaffold during post-dilatation and to ensure adequate scaffold apposition and an appropriate post- implantation minimum lumen diameter. • In small vessels (visually assessed reference vessel diameter ≤ 2. 75 mm), on-line QCA or intravascular imaging with intravascular ultrasound or optical coherence tomography is strongly recommended to accurately measure and confirm appropriate vessel sizing (reference vessel diameter ≥ 2. 5 mm). (See Section 8. 1. 6 – Implantation of Absorb in Small Coronary Arteries (Post Hoc Analysis). • If quantitative imaging determines a vessel size < 2. 5 • • • mm, do not implant the Absorb GT 1 BVS. Implantation of the device in vessels < 2. 5 mm may lead to an increased risk of adverse events such as myocardial infarction and scaffold thrombosis. Adequate lesion preparation prior to scaffold implantation is required to ensure safe delivery of the scaffold across the target lesion. It is not recommended to treat patients having a lesion that prevents complete inflation of an angioplasty balloon. It is strongly recommended to achieve a residual stenosis between 20% and 40% after pre-dilatation to enable successful delivery and full expansion of the scaffold. Ensure the scaffold is not post-dilated beyond the allowable expansion limits (see Absorb GT 1 IFU Section 12. 7 - Clinician Use Information, Further Expansion of the Deployed Scaffold). Antiplatelet therapy should be administered postprocedure (see Absorb GT 1 IFU Section 9. 1 Patient Selection and Treatment, Individualization of Treatment). This product should not be used in patients who are not likely to comply with the recommended antiplatelet therapy. Judicious selection of patients is necessary, since the use of this device carries the associated risk of scaffold thrombosis, vascular complications, and / or bleeding events. PRECAUTIONS • Implantation of the scaffold should be performed only by • Care must be taken to properly size the scaffold to ensure • • • • Do not exceed the Rated Burst Pressure (RBP) as • • • Post-dilatation is strongly recommended for optimal • • physicians who have received appropriate training. indicated on the product label. scaffold apposition. When performed, post-dilatation should be at high pressure (> 16 atm) with a noncompliant balloon. that the scaffold is in full contact with the arterial wall upon deflation of the balloon. All efforts should be made to ensure that the scaffold is not under dilated. Refer to Absorb GT 1 IFU Section 12. 7 - Clinical Use Information, further Expansion of the Deployed Scaffold. Balloon dilatation of any cells of a deployed Absorb GT 1 BVS may cause scaffold damage. Avoid scaffolding across any side branches ≥ 2. 0 mm in diameter. Placement of a scaffold has the potential to compromise side branch patency. It is not recommended to treat patients having a lesion with excessive tortuosity proximal to or within the lesion. Non-clinical testing has demonstrated the Absorb GT 1 BVS is MR Conditional. A patient with this device can be safely scanned in all MR environments 3 T or less. The safety and effectiveness of the Absorb GT 1 BVS have not been established for subject populations with the following characteristics: Coronary artery reference vessel diameters < 2. 5 mm or > 3. 75 mm Lesion lengths > 24 mm Lesions located in arterial or saphenous vein grafts Lesions located in unprotected left main artery Ostial lesions Lesions located at a bifurcation Previously stented lesions Moderate to severe calcification Chronic total occlusion or poor flow (< TIMI 1) distal to the identified lesions Three-vessel disease Unresolved thrombus at the lesion site or anywhere in the vessel to be treated Excessive tortuosity proximal to or within the lesion Recent acute myocardial infarction (AMI) (continued) See important Safety Information referenced within. Not to be reproduced, distributed or excerpted. © 2018 Abbott. All rights reserved. SE 2944958 Rev. B 14 of 18

Important Safety Information (continued) Absorb. GT 1 TM Bioresorbable Vascular Scaffold System POTENTIAL ADVERSE EVENTS Adverse events that may be associated with PCI, treatment procedures and the use of a coronary scaffold in native coronary arteries include the following, but are not limited to: • Allergic reaction or hypersensitivity to latex, contrast agent, anesthesia, device materials (platinum, or polymer [poly(L-lactide) (PLLA), polymer poly(D, L-lactide) (PDLLA)]), and drug reactions to everolimus, anticoagulation, or antiplatelet drugs, Vascular access complications which may require transfusion or vessel repair, including: Catheter site reactions, Bleeding (ecchymosis, oozing, hematoma, hemorrhage, retroperitoneal hemorrhage), Arteriovenous fistula, pseudoaneurysm, dissection, perforation / rupture, Embolism (air, tissue, plaque, thrombotic material or device), Peripheral nerve injury, Peripheral ischemia, Coronary artery complications which may require additional intervention, including: Total occlusion or abrupt closure, Arteriovenous fistula, pseudoaneurysm, dissection, perforation / rupture, Tissue prolapse / plaque shift, Embolism (air, tissue, plaque, thrombotic material or device), Coronary or scaffold thrombosis (acute, subacute, late, very late), Stenosis or restenosis, Pericardial complications which may require additional intervention, including: Cardiac tamponade, Pericardial effusion, Pericarditis, Cardiac arrhythmias (including conduction disorders, atrial and ventricular arrhythmias), Cardiac ischemic conditions (including myocardial ischemia, myocardial infarction [including acute], coronary artery spasm and unstable or stable angina pectoris), Stroke / Cerebrovascular accident (CVA) and Transient Ischemic Attack (TIA), System organ failures: Cardio-respiratory arrest, Cardiac failure, Cardiopulmonary failure (including pulmonary edema), Renal insufficiency / failure, Shock, Blood cell disorders (including Heparin Induced Thrombocytopenia [HIT]), Hypotension / hypertension, Infection, Nausea and vomiting, Palpitations, dizziness, and syncope, Chest pain, Fever, Pain, Death. See important Safety Information referenced within. Not to be reproduced, distributed or excerpted. © 2018 Abbott. All rights reserved. SE 2944958 Rev. B 15 of 18

Important Safety Information The XIENCE V®, XIENCE nano®, XIENCE PRIME® LL, XIENCE Xpedition ®, XIENCE Xpedition® SV and XIENCE Xpedition® LL , XIENCE Alpine. TM (XIENCE Family) of Everolimus Eluting Coronary Stents on the MULTI-LINK VISION® or MULTI-LINK MINI VISION® Delivery Systems • This product should not be used in patients who are not likely to comply with the recommended antiplatelet therapy. PRECAUTIONS • Stent implantation should only be performed by INDICATIONS The XIENCE Family of Everolimus Eluting Coronary Stent Systems are indicated for improving coronary luminal diameter in patients, including those with diabetes mellitus, with symptomatic heart disease due to de novo native coronary artery lesions for XIENCE V (length ≤ 28 mm), XIENCE PRIME, XIENCE Xpedition and XIENCE Alpine (lengths ≤ 32 mm) with reference vessel diameters of ≥ 2. 25 mm to ≤ 4. 25 mm. Additionally, the entire XIENCE Family is indicated for treating de novo chronic total coronary occlusions. • CONTRAINDICATIONS The XIENCE Family of stents is contraindicated for use in patients: • Who cannot receive antiplatelet and/or anti-coagulant therapy • With lesions that prevent complete angioplasty balloon inflation or proper placement of the stent or stent delivery system • With hypersensitivity or contraindication to everolimus or structurally-related compounds, cobalt, chromium, nickel, tungsten, acrylic, and/or fluoropolymers. • WARNINGS • Ensure that the inner package sterile barrier has not been opened or damaged prior to use. • Judicious patient selection is necessary because the use of this device carries the associated risk of stent thrombosis, vascular complications, and/or bleeding events. • • • physicians who have received appropriate training. Stent placement should be performed at hospitals where emergency coronary artery bypass graft surgery is accessible. Subsequent restenosis may require repeat dilatation of the arterial segment containing the stent. Long-term outcomes following repeat dilatation of the stent are presently unknown. Risks and benefits should be considered in patients with severe contrast agent allergies. Care should be taken to control the guiding catheter tip during stent delivery, deployment and balloon withdrawal. Before withdrawing the stent delivery system, visually confirm complete balloon deflation by fluoroscopy to avoid guiding catheter movement into the vessel and subsequent arterial damage. Stent thrombosis is a low-frequency event that is frequently associated with myocardial infarction (MI) or death. When DES are used outside the specified Indications for Use, patient outcomes may differ from the results observed in the SPIRIT family of trials Compared to use within the specified Indications for Use, the use of DES in patients and lesions outside of the labeled indications may have an increased risk of adverse events, including stent thrombosis, stent embolization, MI, or death. Orally administered everolimus combined with cyclosporine is associated with increased serum cholesterol and triglycerides levels. A patient’s exposure to drug and polymer is proportional to the number and total length of implanted stents. See Instructions for Use for current data on multiple stent implantation. • Safety and effectiveness of the XIENCE Family of stents have not been established for subject populations with the following clinical settings: • Patients with prior target lesion or in-stent restenosis related brachytherapy, patients in whom mechanical atherectomy devices or laser angioplasty catheters are used in conjunction with XIENCE Family stents, women who are pregnant or lactating, men intending to father children, pediatric patients, unresolved vessel thrombus at the lesion site, coronary artery reference vessel diameters < 2. 25 mm or > 4. 25 mm or lesion length > 32 mm, lesions located in saphenous vein grafts, unprotected left main coronary artery, ostial lesions, lesions located at a bifurcation or previously stented lesions, diffuse disease or poor flow (TIMI < 1) distal to the identified lesions, excessive tortuosity proximal to or within the lesion, recent acute myocardial infarction (AMI) or evidence of thrombus in target vessel multivessel disease, and instent restenosis • Everolimus has been shown to reduce the clearance of • • some prescription medications when administered orally along with cyclosporine (Cs. A). Formal drug interaction studies have not been performed with the XIENCE Family of stents because of limited systemic exposure to everolimus eluted from the stent. Everolimus is an immunosuppressive agent. Consideration should be given to patients taking other immunosuppressive agents or who are at risk for immune suppression. Oral everolimus use in renal transplant patients and advanced renal cell carcinoma patients was associated with increased serum cholesterol and triglycerides, which in some cases required treatment. Nonclinical testing has demonstrated that the XIENCE Family of stents, in single and in overlapped configurations are MR conditional up to 68 mm in length for XIENCE V and XIENCE nano only and up to 71 mm in length for all other XIENCE Family stents. It can be scanned safely under the conditions in the Instructions for Use. The XIENCE Family of stents should be handled, placed, implanted, and removed according to the Instructions for Use. (continued) 16 of 18 See important Safety Information referenced within. Not to be reproduced, distributed or excerpted. © 2018 Abbott. All rights reserved. SE 2944958 Rev. B

Important Safety Information (continued) The XIENCE V®, XIENCE nano®, XIENCE PRIME® LL, XIENCE Xpedition ®, XIENCE Xpedition® SV and XIENCE Xpedition® LL , XIENCE Alpine. TM (XIENCE Family) of Everolimus Eluting Coronary Stents on the MULTI-LINK VISION® or MULTI-LINK MINI VISION® Delivery Systems POTENTIAL ADVERSE EVENTS Adverse events (in alphabetical order) which may be associated with percutaneous coronary and treatment procedure including coronary stent use in native coronary arteries include, but are not limited to: • Abrupt closure, Access site pain, hematoma, or hemorrhage, Acute myocardial infarction, Allergic reaction or hypersensitivity to contrast agent or cobalt, chromium, nickel, tungsten, acrylic and fluoropolymers; and drug reactions to antiplatelet drugs or contrast agent, Aneurysm, Arterial perforation and injury to the coronary artery, Arterial rupture, Arteriovenous fistula, Arrhythmias, atrial and ventricular, Bleeding complications, which may require transfusion, Cardiac tamponade, Coronary artery spasm, Coronary or stent embolism, Coronary or stent thrombosis, Death, Dissection of the coronary artery, Distal emboli (air, tissue or thrombotic), Emergent or non-emergent coronary surgery, Fever, Hypotension and / or hypertension, Infection and pain at insertion site, Injury to the coronary artery, Ischemia (myocardial), Myocardial infarction (MI), Nausea and vomiting, Palpitations, Peripheral ischemia (due to vascular injury), Pseudoaneurysm, Renal Failure, Restenosis of the stented segment of the artery, Shock/pulmonary edema, Stroke / cerebrovascular accident (CVA), Total occlusion of coronary artery, Unstable or stable angina pectoris, Vascular complications including at the entry site which may require vessel repair, Vessel dissection Adverse events associated with daily oral administration of everolimus to organ transplant patients include but are not limited to: • Abdominal pain (including upper abdominal pain); Anemia; Angioedema; Anorexia; Asthenia; Constipation; Cough; Delayed wound healing/fluid accumulation; Diarrhea; Dyslipidemia (including hyperlipidemia and hypercholesterolemia); Dyspnea; Dysgeusia; Dyspepsia; Dysuria; Dry skin; Edema (peripheral); Epistaxis; Fatigue; Headache; Hematuria; Hyperglycemia (may include new onset of diabetes); Hyperlipidemia; Hyperkalemia; Hypertension; Hypokalemia; Hypomagnesemia; Hypophosphatemia; Increased serum creatinine; Infections and serious infections: bacterial, viral, fungal, and protozoal infections (may include herpes virus infection, polyoma virus infection which may be associated with BK virus associated nephropathy, and/or other opportunistic infections); Insomnia; Interaction with strong inhibitors and inducers of CY 3 PA 4 or Pg. P; Leukopenia; Lymphoma and other malignancies (including skin cancer); Male infertility (azospermia and/or oligospermia); Mucosal inflammation (including oral ulceration and oral mucositis); Nausea; Neutropenia; Non-infectious pneumonitis; Pain: extremity, incision site and procedural, back, chest, and musculoskeletal; Proteinuria; Pruritus; Pyrexia; Rash; Stomatitis; Thrombocytopenia; Thrombotic microangiopathy (TMA)/Thrombotic thrombocytopenic purpura (TTP)/ Hemolytic uremic syndrome (HUS); Tremor; Urinary tract infection; Upper respiratory tract infection; Vomiting • Live vaccines should be avoided and close contact with those that have had live vaccines should be avoided. Fetal harm can occur when administered to a pregnant woman. There may be other potential adverse events that are unforeseen at this time. 17 of 18 See important Safety Information referenced within. Not to be reproduced, distributed or excerpted. © 2018 Abbott. All rights reserved. SE 2944958 Rev. B

Caution: These products are intended for use by or under the direction of a physician. Prior to use, reference the Instructions for Use provided inside the product carton (when available), at eifu. abbottvascular. com or at Manuals. sjm. com for more detailed information on Indications, Contraindications, Warnings, Precautions and Adverse. Events Tests performed by and data on file at Abbott. Illustrations are artist’s representations only and should not be considered as engineering drawings or photographs. Abbott 3200 Lakeside Dr. , Santa Clara, CA. 95054 USA, Tel: 1. 800. 227. 9902 Absorb and Absorb GT 1 are trademarks of the Abbott Group of Companies. www. Vascular. Abbott © 2018 Abbott. All rights reserved. SE 2944958 Rev. B 18 of 18
- Slides: 18