Faculty of Pharmacy Department of Pharmaceutical Chemistry Practical







- Slides: 10

Faculty of Pharmacy Department of Pharmaceutical Chemistry Practical Analytical Chemistry (1) Practical (5)

What are the Anions ? Anions are ions bearing a negative charge; they are formed from non-metallic elements due to gain of electrons. Why we should analyze Anions ? Ø Some anions are necessary for human life. Ø Some anions are used for different pharmaceutical purposes as sulphates and carbonates. Ø Some anions are environmental hazards that cause great risk to human health as cyanides and nitrites. The conc. in the body is related to the health status

Classification of Anions are classified by 2 different modes: A. according to the identification by the volatile products obtained on treatment with acids. 1. Gases evolved with dil. HCl or H 2 SO 4 2. Gases or vapors evolved with conc. H 2 SO 4 3. Miscellaneous group A. according to the identification by the reactions in solutions (according to their solubilities) 1. Precipitation reactions 2. oxidation-reduction reactions

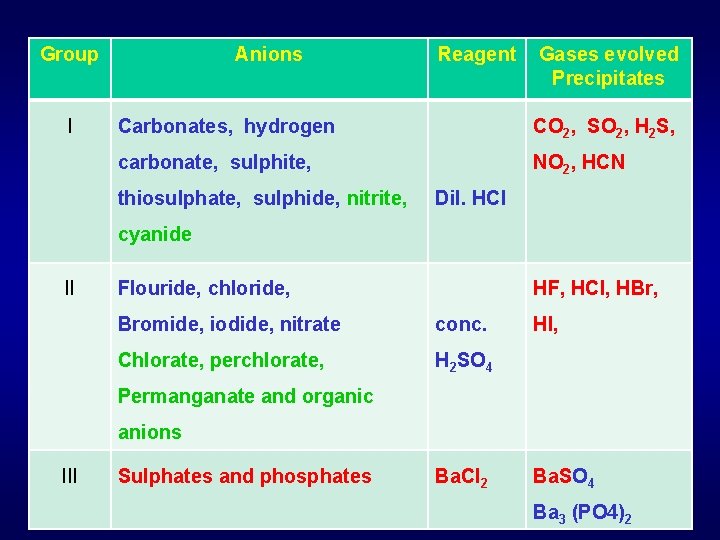
Classification of Anions Mixture of many Anions Reagent; dilute HCl Group (Group I) Volatile gases Carbonates, hydrogen carbonate, sulphite, thiosulphate, sulphide, nitrite, cyanide Reagents Confirmation (precipitate, colour, gas, …) Conc. H 2 SO 4 (Group II) conc. H 2 SO 4 Colored vapours Miscellaneous Group III Sulphate phosphate Flouride, chloride, bromide, iodide, nitrate chlorate, perchlorate, bromate Permanganate, borate, ferrocyanide, ferricyanide, Thiocyanate, organic acids

Group I Anions Reagent Gases evolved Precipitates Carbonates, hydrogen CO 2, SO 2, H 2 S, carbonate, sulphite, NO 2, HCN thiosulphate, sulphide, nitrite, Dil. HCl cyanide II Flouride, chloride, HF, HCl, HBr, Bromide, iodide, nitrate conc. Chlorate, perchlorate, H 2 SO 4 HI, Permanganate and organic anions III Sulphates and phosphates Ba. Cl 2 Ba. SO 4 Ba 3 (PO 4)2

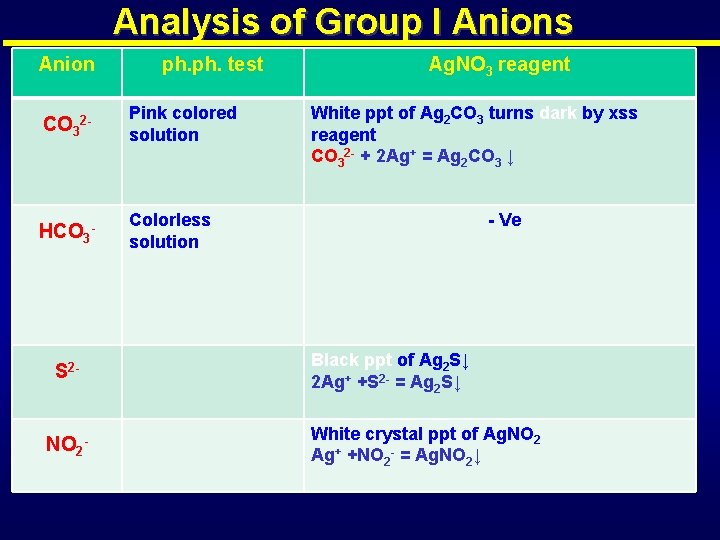
Anions of Group I Carbonate (CO 32 -), Bicarbonate (HCO 3 -), Sulphide (S 2 - ) , Thiosulphate (S 2 O 32 -), Sulphite (SO 32 -), Nitrite(NO 2 -), Cyanide(CN-) Nitrite(NO 2 -): is one of the most toxic environmental contaminant that causes great risks to general human health. Cyanide(CN-) :

Analysis of Group I Anions Anion Dil HCl equations 2 - Effervescence due to evolution of CO 2 CO 32 - + 2 H+ = CO 2 ↑ + H 2 O gas which renders lime water turbid CO 2 + Ca(OH)2 = Ca. CO 3 + H 2 O HCO 3 Effervescence due to evolution of CO 2 HCO 3 - + H+ = CO 2 ↑ + H 2 O gas which renders lime water turbid CO 2 + Ca(OH)2 = Ca. CO 3 + H 2 O CO 3 S 2 - NO 2 - - evolution of H 2 S gas characterized by its odor of bad egg which turns lead acetate paper black S 2 - + 2 H+ = H 2 S ↑ H 2 S + Pb 2+ = PBS↓ black ppt Pale blue liquid + evolution of NO 2 brown fumes NO 2 - + H+ = HNO 2 ↑ + H 2 O 3 HNO 2 = HNO 3 + H 2 O + 2 NO ↑ + O 2↑ = 2 NO 2↑ (brown)

Analysis of Group I Anions Anion ph. test CO 32 - Pink colored solution HCO 3 - Colorless solution S 2 - NO 2 - Ag. NO 3 reagent White ppt of Ag 2 CO 3 turns dark by xss reagent CO 32 - + 2 Ag+ = Ag 2 CO 3 ↓ - Ve Black ppt of Ag 2 S↓ 2 Ag+ +S 2 - = Ag 2 S↓ White crystal ppt of Ag. NO 2 Ag+ +NO 2 - = Ag. NO 2↓

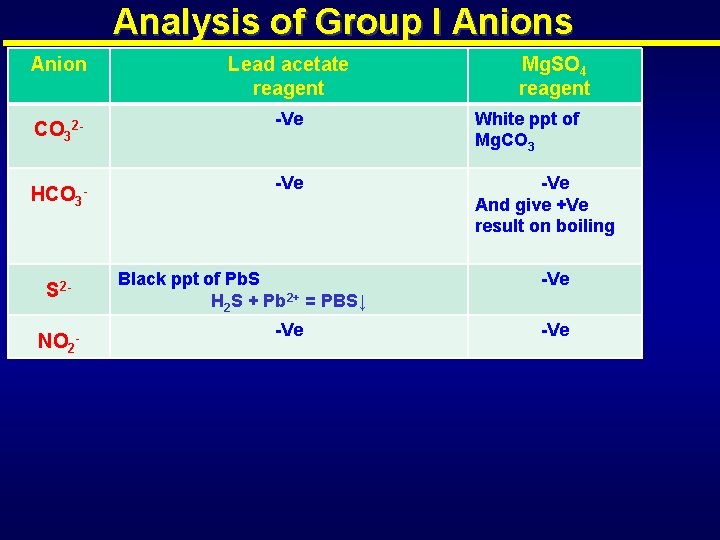
Analysis of Group I Anions Anion CO 32 HCO 3 - S 2 NO 2 - Lead acetate reagent Mg. SO 4 reagent -Ve White ppt of Mg. CO 3 -Ve And give +Ve result on boiling Black ppt of Pb. S H 2 S + Pb 2+ = PBS↓ -Ve -Ve

Points for discussion 1 -Write a flow chart for your unknown analysis in your lab notebook. You should include formulas of all reagents added, colors of all solutions and ppts, and conclusions made from these observations. For example, your original solution is colorless, you add HCl forming a white ppt and a colorless solution. At this point you do not know what the formula of the white ppt is, so do not write one.