Faculty of health sciences Medical Laboratory Sciences Department

Faculty of health sciences Medical Laboratory Sciences Department Medical parasitology lab. Concentration techniques

v. The microscopic examination of feces is required for the recognition and identification of intestinal parasites: 1. Direct Microscopy: • Advantages ØUseful for the observation of motile protozoan trophozoites. • Disadvantages ØMay not detect ova, cysts and larvae which are present in scant numbers.

• In most cases Samples contains some form of debris. Removal of such material is essential to ensure accurate and thorough screening of the specimen, it is for this reason, coupled with the fact that the parasites are often present in low numbers and need to be condenced into one area of the sample that concentration methods have been developed.

• STOOL CONCENTRATION METHODS: Where heavy infestation is present this method is not needed. Concentration method may be used to seen whether treatment of the parasites has been successful. To find ova of S. mansoné or Taenia is few of other ova and cysts if they have not been seen in routine examination (being very few) and are suspected to be present. To examine stool specimens from patients who do not come from an area where a particular parasite is found.

Ø If number of organisms in stool specimen is low, examination of a direct wet mount may not detect parasites. Ø Thus, whenever possible, the stool should be concentrated. Ø worm eggs, larvea, and protozoan cysts may be recovered by concentration but protozoan trophozoites will not be seen as they are usually destroyed during the concentration procedures. This makes direct wet mount examination obligatory as the initial phase of microscopic examination.

Principles of concentration techniques Ø The concentration procedure is indicated when the initial wet mount examination is negative despite the clinical symptoms indicating parasitic infection of a patient. Ø The principle of concentration technique is based on specific gravity. By addition of ethyle acetate to formaline fixed sample and subsequent centrifugation, the parasites present are heavier than the solution and seetle in the sediment of the tube.

• Sample debrise is typically lighter , relatively speaking , and rises to the upper layer s of the test tube. subsequent decaning of the supernatant and preparation and examination of concentrate saline or iodine wet preparation of sediment complete this process.

2. Concentration techniques : • Advantages Ø Maximizes the numbers of organisms detected which may be too scanty to be seen by direct microscopy alone. Ø Worm eggs, larvae, and protozoan cysts may be recovered. • Disadvantages Ø Destroys trophozoite stages. Most concentration methods destroy trophozoites stages. q. The purpose of concentrating feces is to increase possibility to finding ova, cyst, or larvae in samples that not be able to seen by direct microscopy.

Concentration Methods 1. Sedimentation method § Modified Formal- Ether sedimentation technique § Acid- Ether sedimentation technique 2. Flotation method § Saturated Salt Solution technique § Sheather’s Sugar Centrifugal Flotation technique § Zinc Sulphate Centrifugal Flotation technique

• In sedimentation methods, the parasites are not floated but deposited, usually by centrifuging. A small piece of stool is mixed with saline in a tube or bottle and sieved through a strainer. The sieved contents are centrifuged and the supernatant fluid poured off. The deposit is re-suspended in more saline, mixed, and centrifuged. This is repeated until the supernatant fluid is clear. The deposit is examined directly on a slide. By this simple method, parasitic cysts, eggs, and free living parasites can be concentrated.

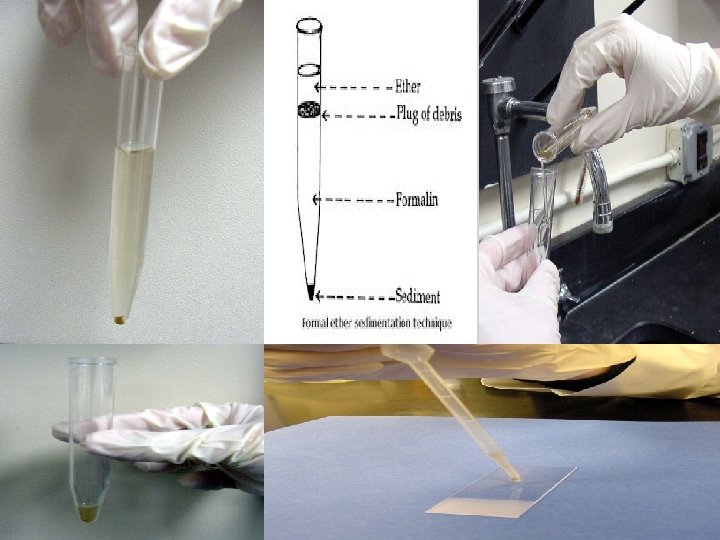
Sedimentation Methods MODIFIED FORMAL- ETHER SEDIMENTATION

Modified Formal- Ether Sedimentation • Formalin- Ether or Formalin- Ethyl acetate method is the recommended concentration procedures. • Most types of worm eggs (round worms, tapeworms, schistosomes, and other fluke eggs), larvae, and protozoan cysts may be recovered by this method. • Advantages: 1. Speed: one sample can be processed in 5 minutes. 2. Broad spectrum: it will recover most ova, cyst and larvae. 3. The morphology of most parasites is retained for easy identification. • Disadvantages: 1. Requires several pieces of apparatus which does not make it an easy. 2. The preparation contains some debris. 3. Ether is flammable. Formalin is an irritant. 4. Hymenolepis nana and Fasciola spp. do not concentrate well.

Materials and Method ØLibra ØApplicator stick ØGlass centrifugal tubes ØBeaker ØWire sieve ØVortex or whirlimixer ØCentrifuge. ØReagent: v. Reagent I: 10% formalin solution in distilled water. v. Reagent II: diethyl ether or ethyl acetate.

Caution Ether is a highly flammable compound and will ignite and explode quickly if there is a flame or spark nearby.


Procedures 1. Emulsify 1 gm. of feces in 7 ml of 10% formalin in a centrifuge tube. 2. Strain the suspension through a brass wire sieve, and collect in beaker. 3. Pour the filtrate into a 15 ml boiling tube and add 3 ml of ether, then mix well 15 sec on vortex or whirlimixer or 1 min by hand. 4. Transfer the ether- formalin suspension back into the washed centrifuge tube, and centrifuge at 3, 000 rpm for 1 min.


Procedures (cont. ) 5. Loosen the fatty layer and debris at the top of the tube with an applicator stick and invert the tube quickly to discard the supernatant. 6. On righting the tube, a few drops only should remain with the sediment, mix the sediment well and transfer one drops onto a glass slide and cover it with coverslip. 7. Scan the whole coverslip using 10 x objective, turning into 40 x for confirmation of identification of parasites.

Sedimentation Methods ACID- ETHER SEDIMENTATION TECHNIQUE

Materials and Method ØLibra ØApplicator stick ØGlass centrifugal tubes ØBeaker ØWire sieve ØVortex or whirlimixer ØCentrifuge. ØReagent: v. Reagent I: 15% Hydrochloric acid. • Conc. HCl 40 ml + 60 ml Distilled water. v. Reagent II: diethyl ether or ethyl acetate.

Procedures 1. Mix thoroughly 1 gm. feces with 3 ml of 15% of hydrochloric acid and then mix well. 2. Add and additional 5 -6 ml of 15% HCl and mix. 3. Strain the suspension through a wire sieve into beaker. 4. Place suspension in a glass centrifuge tube and make up to the 10 ml with distilled water. 5. Add 4 ml of ether, stopper the tube and shake vigorously 20 -30 sec using vortex.

Procedures (cont. ) 6. Centrifuge 2 -3 min at 1500 rpm, the suspension now will be layered. 7. Loosen plug of debris with applicator stick and immediately pour off liquid. 8. Transfer one drops onto a glass slide and cover it with coverslip. 9. Scan the whole coverslip using 10 x objective, turning into 40 x for confirmation of identification of parasites.

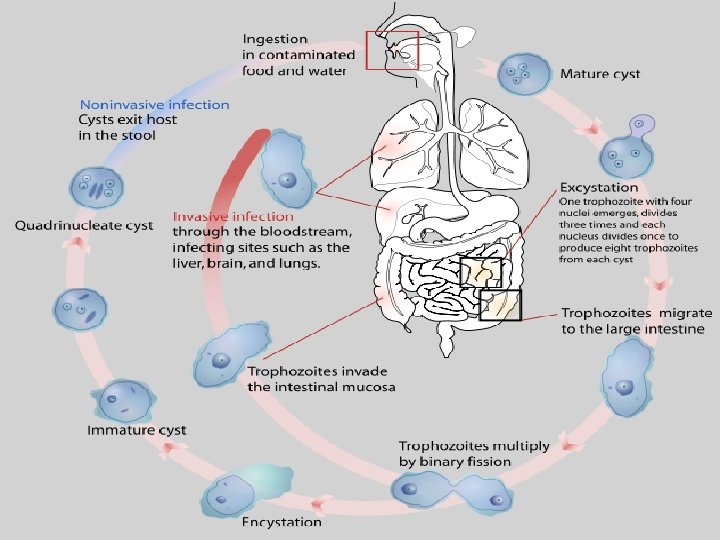
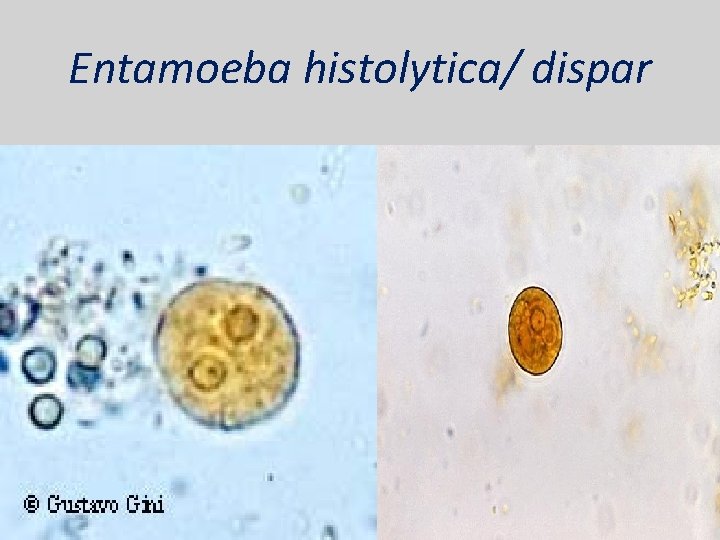
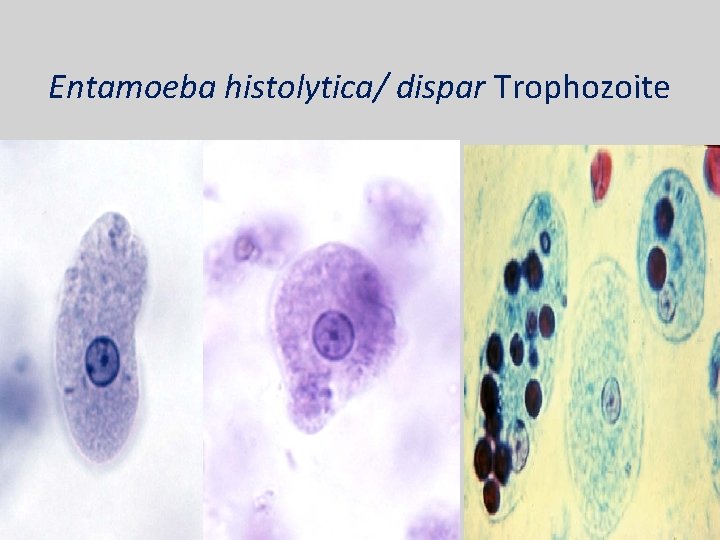
Entamoeba histolytica/ dispar • E. histolytica inhabit large intestine and cause amoebic dysentery. • There is two diagnostic stages for E. histolytica/ dispar: 1. Cyst is regular round measuring measure 10 – 20 u in diameter with 4 nuclei, and it’s the infective stage. 2. Trophozoite is the motile form, measure 15 -20 u in diameter with large nucleus. (motility by pseudopodia). • Diagnosis: – Stool examination to see cyst stage, or trophozoite stage if the sample is fresh.
Entamoeba histolytica/ dispar
Entamoeba histolytica/ dispar Trophozoite
- Slides: 27