Faculty Leonard H Calabrese DO Professor of Medicine


Faculty Leonard H. Calabrese, DO Professor of Medicine Cleveland Clinic Lerner College of Medicine RJ Fasenmyer Chair of Clinical Immunology Cleveland Clinic Cleveland, Ohio Edward Li, Pharm. D, MPH, BCOP Associate Professor Department of Pharmacy Practice University of New England College of Pharmacy Portland, Maine

Disclosures Leonard Calabrese, DO Consultant Speakers Bureau Janssen Edward Li, MD Consultant Speakers Bureau Bio. Gen, Bristol-Myers Squibb, Genentech, GSK, Regeneron Abb. Vie, Crecsendo, Geron, Eli Lilly, Pfizer Apobiologix, Pfizer

Learning Objectives Recognize manufacturing differences between biosimilars and their reference biological product as well as generic small molecule drugs Describe how the FDA uses a “totality of evidence” strategy to evaluate biosimilar compounds Consider how the availability of biosimiliars will impact clinical practice

Differences Between Biosimilar and Reference Products Leonard H. Calabrese, DO Professor of Medicine Cleveland Clinic Lerner College of Medicine RJ Fasenmyer Chair of Clinical Immunology Cleveland Clinic

Why Biosimilars Biologic therapeutics in multiple fields have revolutionized the treatment for autoimmune, autoinflammatory, and cancer Biosimilars are expensive: drugs for rheumatoid arthritis cost $50, 000/year 1 Costs have increased progressively: cost of TNFi drugs have doubled since their introduction Biosimilar TNFi in Norway in 2014 led to nearly a 60% annual saving Estimates of cost saving: >$66 billion USD over the next decade (4% of total biologics)3 TNFi, tumor necrosis factoralpha inhibitor 1. http: //www. fda. gov/downloads/advisorycommittees/committeesmeetingmaterials/drugsadvisorycommitteefo rpharmaceuticalscienceandclinicalpharmacology/ucm 315764. pdf 2. Dörner T, et al. Ann Rheum Dis. 2016; 75(6): 974 -982. 3. Mulcahy AW, et al. The Cost Savings Potential of Biosimilar Drugs in the United States. RAND Corporation.

What is a Biopharmaceutical? Any virus, therapeutic serum, toxin, antitoxin, or analogous product applicable to the prevention, treatment or cure of diseases or injuries of man. Derived from living sources • Cultured bacteria, yeast, or cells • Human or animal sources Therapeutic proteins Code of Federal Regulations Title 21, Volume 7. Subchapter F-Biologics. https: //www. accessdata. fda. gov/scripts/cdrh/cfdocs/cfcfr/CFRSearch. cfm? fr=600. 3 Accessed February 6, 2017.
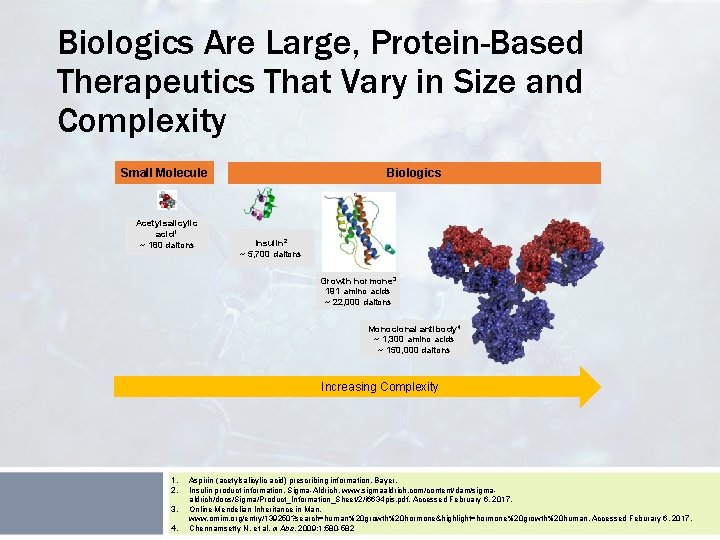
Biologics Are Large, Protein-Based Therapeutics That Vary in Size and Complexity Small Molecule Acetylsalicylic acid 1 ~ 180 daltons Biologics Insulin 2 ~ 5, 700 daltons Growth hormone 3 191 amino acids ~ 22, 000 daltons Monoclonal antibody 4 ~ 1, 300 amino acids ~ 150, 000 daltons Increasing Complexity 1. 2. 3. 4. Aspirin (acetylsalicylic acid) prescribing information, Bayer. Insulin product information, Sigma-Aldrich. www. sigmaaldrich. com/content/dam/sigmaaldrich/docs/Sigma/Product_Information_Sheet/2/i 6634 pis. pdf. Accessed February 6, 2017. Online Mendelian Inheritance in Man. www. omim. org/entry/139250? search=human%20 growth%20 hormone&highlight=hormone%20 growth%20 human. Accessed Feburary 6, 2017. Chennamsetty N, et al. m. Abs. 2009; 1: 580 -582

Why Is it Important to Understand the Biosimilar Development Process? Known DNA sequence 1 Unique manufacturing 1, 2 Unique biologic 2 Understanding the biosimilar development process allows prescribers to make informed decisions about the potential utility of biosimilars as a treatment option 1. Roger SD. Nephrology (Carlton). 2006; 11: 341 -346. 2. Mellstedt H, et al. Ann Oncol. 2008; 19: 411 -419.

What Is a Biosimilar? A biosimilar is a “copy” of a commercially available biopharmaceutical (reference product) that no longer is protected by patent which has: • Undergone rigorous analytical and clinical assessment, in comparison to its reference product • AND • Been approved by a regulatory agency according to a specific pathway for biosimilar evaluation A biosimilar is “highly similar” to its reference product in physicochemical characteristics, efficacy, and safety. Dörner T, et al. Ann Rheum Dis. 2016; 75(6): 974 -982.

Biosimilars Are Not… Second-Generation (or Biobetter) Generic Drugs → Structurally different from originally licensed biopharmaceutical → Small-molecule drugs, that are less complex than biosimilars → Intended to improve performance while preserving mechanism of action → Manufacturing process is several orders of magnitude less complex → Examples Infliximab and adalimumab Filgrastim and pegfilgrastim → Regulated under different legislation → Not considered to be biosimilar Woodcock J, et al. Nat Rev Drug Discov. 2007; 6(6): 437 -442.

US Regulatory Pathways for Small. Molecule Drugs and Biopharmaceuticals US Approval Pathways for Small Molecule Drugs vs Biologics FDCA = Food, Drug, and Cosmetic Act BPCI = Biologics Price Competition and Innovation PHSA = Public Health Service For historical reasons, some biologic products are currently approved under the FDCA. From 2020, all biologic products will be approved under the PHSA beginning in 2020. Li EC, et al. J Manag Care Spec Pharm. 2015; 21(7): 532 -539.

Goals of “Stand-Alone” and Biosimilar Development are Different “Stand-alone” Development Program, 351(a) Goal: To establish safety and efficacy of a new product Clinical Safety and Efficacy (Phases 1, 2, 3) Clinical Pharmacology “Abbreviated” Development Program, 351(k) Goal: To demonstrate biosimilarity (or interchangeability) Additional Clinical Studies Clinical Pharmacology Non-clinical Analytical Overview of the Regulatory Pathway and FDA’s Guidance for the Development and Approval of Biosimilar Products in the US. http: //www. fda. gov/downloads/Advisory. Committees/Committees Meeting. Materials/Drugs/Arthritis. Advisory. Committee/UCM 486171. pdf. Accessed February 4, 2017.

Biologics: Drug Substance Production Process Host cells to grow the protein Lengthy and complex purification process Factors influencing properties of the molecule: • Time, p. H, temperature, culture media, oxygen levels/lactic acid accumulation Properties most frequently affected: • Carbohydrates, ratios of charged species, aggregates Liu HF et al. m. Abs. 2010; 2; (5): 480 -499 Li F. et al. m. Abs 2010; 2(5): 466 -477 END Fermentation with very precise culture conditions Vectors to insert the gene START Gene sequence coding for protein Biophysical characteristics of final drug substance Factors influencing properties of the molecule/product: • Removal of host cell DNA and proteins • Removal of fragments and aggregates • Concentration of the protein • Final formulation buffer
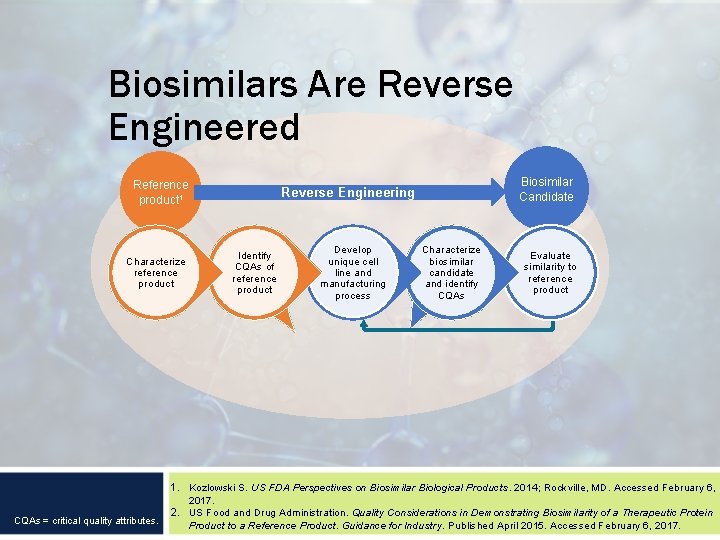
Biosimilars Are Reverse Engineered Reference product 1 Characterize reference product CQAs = critical quality attributes. Biosimilar Candidate Reverse Engineering Identify CQAs of reference product Develop unique cell line and manufacturing process Characterize biosimilar candidate and identify CQAs Evaluate similarity to reference product 1. Kozlowski S. US FDA Perspectives on Biosimilar Biological Products. 2014; Rockville, MD. Accessed February 6, 2017. 2. US Food and Drug Administration. Quality Considerations in Demonstrating Biosimilarity of a Therapeutic Protein Product to a Reference Product. Guidance for Industry. Published April 2015. Accessed February 6, 2017.

Biosimilars Development: A Stepwise Approach PK = pharmacokinetics PD = pharmacodynamics Dörner T, et al. Nat Rev Rheumatol. 2015; 11(12): 713 -724. Reprinted by permission from Macmillan Publishers Ltd

Demonstrating Biosimilarity: General Principles Clinical efficacy and safety of reference biopharmaceutical have already been demonstrated Biosimilar must demonstrate no significant difference from its reference product • Robust analytical, toxicologic, PK/PD, and immunogenicity studies in comparison to reference product • Smaller comparative effectiveness clinical trial(s) conducted in patients in a disease for which the reference product is licensed • No need to demonstrate efficacy in all indications No differences in safety or efficacy are expected between an approved biosimilar and its reference product PK = pharmacokinetics PD = pharmacodynamics http: //www. fda. gov/downloads/Drugs. Guidance. Compliance. Regulatory. Information/Guidances/UCM 291128. pdf. Accessed February 6, 2017.

Extensive Analytical Characterization Is Used Attributes related to the amino acid sequence and all post-translational modifications, including glycans Integrity of the secondary, tertiary, and quaternary structure Primary structure Higher order structure Biological and functional activities, including receptor binding and immunochemical properties Biological function Product-related substances and impurities Quantitative levels of product variants and their identities General properties and excipients Properties of the finished drug product, including strength and formulation CQA = critical quality attribute Process-related impurities Impurities from host cells and downstream process Receptor binding and immunochemical properties Kinetics and thermodynamics of binding, related to functional activity Stability Degradation profiles denoting stability US Food and Drug Administration. Quality Considerations in Demonstrating Biosimilarity of a Therapeutic Protein Product to a Reference Product. Guidance for Industry. April 2015.
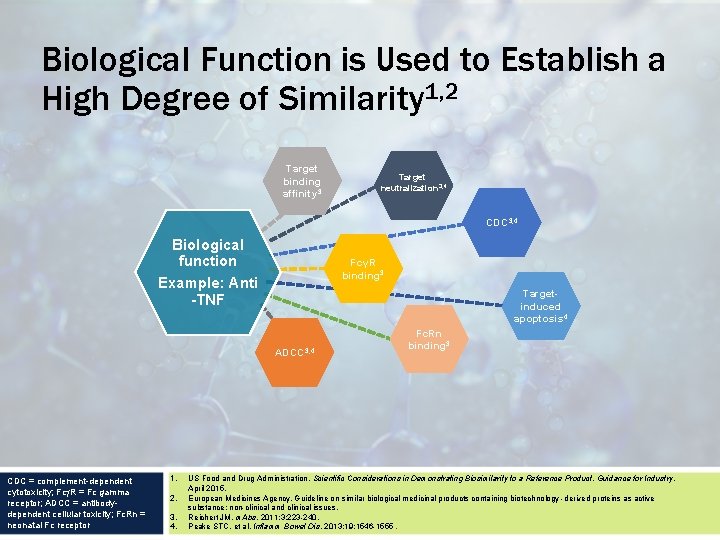
Biological Function is Used to Establish a High Degree of Similarity 1, 2 Target binding affinity 3 Target neutralization 3, 4 CDC 3, 4 Biological function Example: Anti -TNF FcγR binding 3 Targetinduced apoptosis 4 ADCC 3, 4 CDC = complement-dependent cytotoxicity; Fc R = Fc gamma receptor; ADCC = antibodydependent cellular toxicity; Fc. Rn = neonatal Fc receptor 1. 2. 3. 4. Fc. Rn binding 3 US Food and Drug Administration. Scientific Considerations in Demonstrating Biosimilarity to a Reference Product. Guidance for Industry. April 2015. European Medicines Agency. Guideline on similar biological medicinal products containing biotechnology -derived proteins as active substance: non-clinical and clinical issues. Reichert JM. m. Abs. 2011; 3: 223 -240. Peake STC, et al. Inflamm Bowel Dis. 2013; 19: 1546 -1555.

What is immunogenicity? Immunogenicity is: …the ability of a particular substance, such as an antigen or epitope, to provoke an immune response in the body of a human or animal. …the ability to induce a humoral and/or cellmediated immune responses. Woodcock J, et al. Nat Rev Drug Discov. 2007; 6(6): 437 -442.
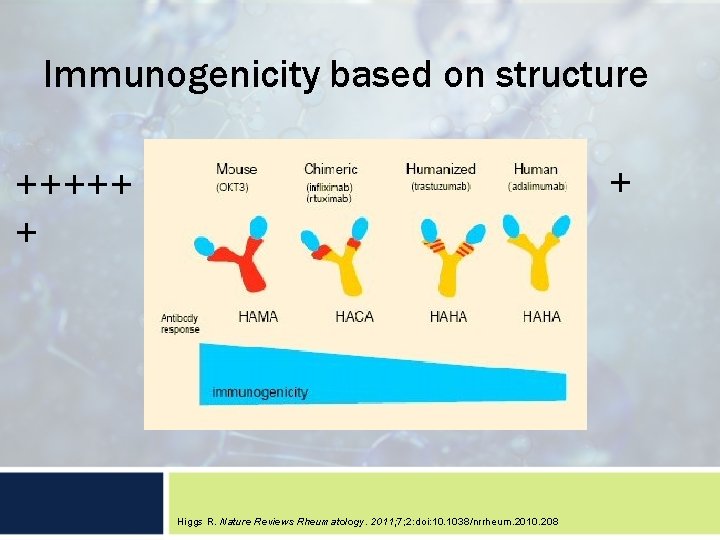
Immunogenicity based on structure + +++++ + Higgs R. Nature Reviews Rheumatology. 2011; 7; 2: doi: 10. 1038/nrrheum. 2010. 208
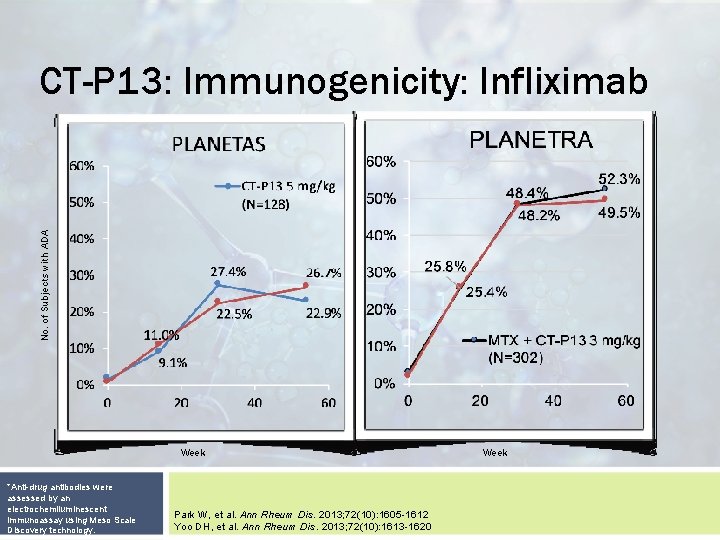
No. of Subjects with ADA CT-P 13: Immunogenicity: Infliximab Week *Anti-drug antibodies were assessed by an electrochemiluminescent immunoassay using Meso Scale Discovery technology. Park W, et al. Ann Rheum Dis. 2013; 72(10): 1605 -1612 Yoo DH, et al. Ann Rheum Dis. 2013; 72(10): 1613 -1620 Week
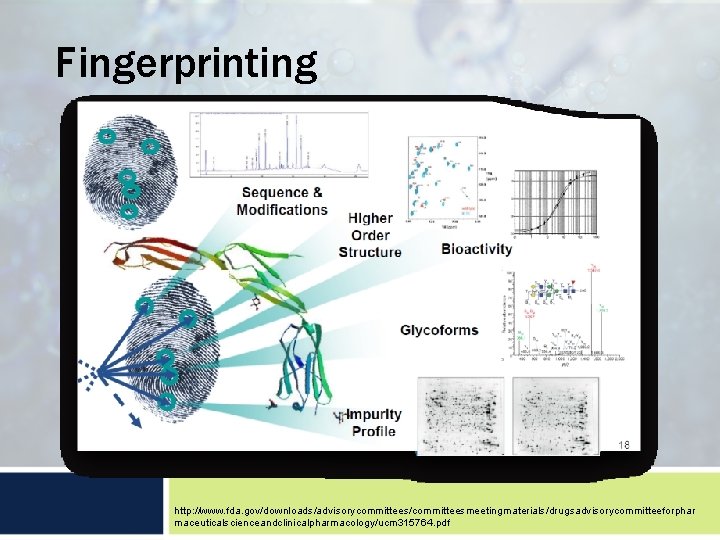
Fingerprinting http: //www. fda. gov/downloads/advisorycommittees/committeesmeetingmaterials/drugsadvisorycommitteeforphar maceuticalscienceandclinicalpharmacology/ucm 315764. pdf
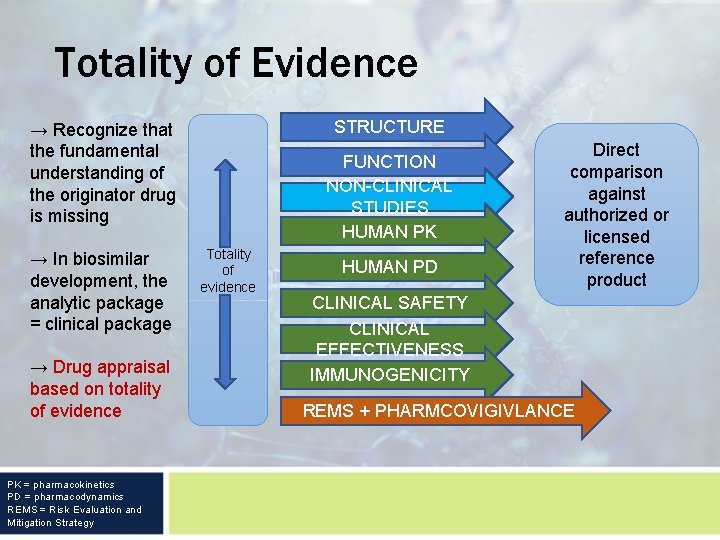
Totality of Evidence STRUCTURE → Recognize that the fundamental understanding of the originator drug is missing → In biosimilar development, the analytic package = clinical package → Drug appraisal based on totality of evidence PK = pharmacokinetics PD = pharmacodynamics REMS = Risk Evaluation and Mitigation Strategy FUNCTION NON-CLINICAL STUDIES HUMAN PK Totality of evidence HUMAN PD Direct comparison against authorized or licensed reference product CLINICAL SAFETY CLINICAL EFFECTIVENESS IMMUNOGENICITY REMS + PHARMCOVIGIVLANCE

How Biosimilars Will Impact Clinical Practice Edward Li, Pharm. D, MPH, BCOP Associate Professor University of New England College of Pharmacy
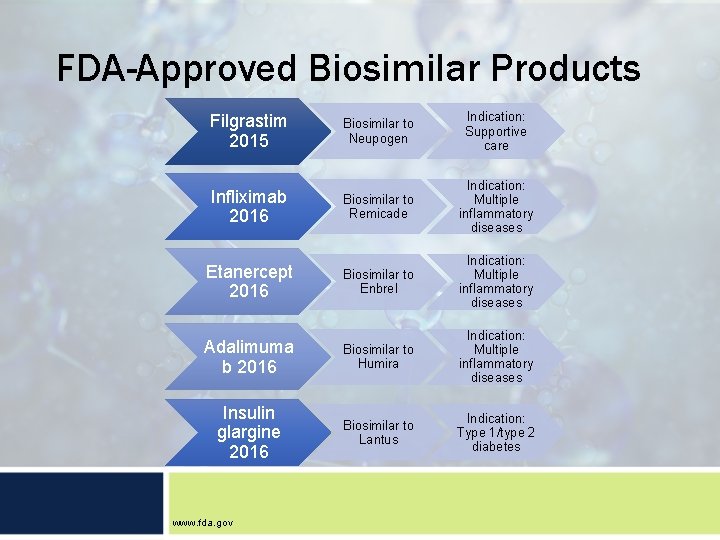
FDA-Approved Biosimilar Products Filgrastim 2015 Biosimilar to Neupogen Indication: Supportive care Infliximab 2016 Biosimilar to Remicade Indication: Multiple inflammatory diseases Etanercept 2016 Biosimilar to Enbrel Indication: Multiple inflammatory diseases Adalimuma b 2016 Biosimilar to Humira Indication: Multiple inflammatory diseases Insulin glargine 2016 Biosimilar to Lantus Indication: Type 1/type 2 diabetes www. fda. gov

Clinical Impact of Biosimilars Lower cost and increase access to medications due to prescriber behavior change Change our paradigm of considering study designs and relevant endpoints (extrapolation) Cause dispensers and prescribers to consider substituting interchangeable products Require providers to be aware of pharmacovigilance issues, such as naming and immunogenicity concerns Dörner T, et al. Ann Rheum Dis. 2016; 75(6): 974 -982

Cost Considerations
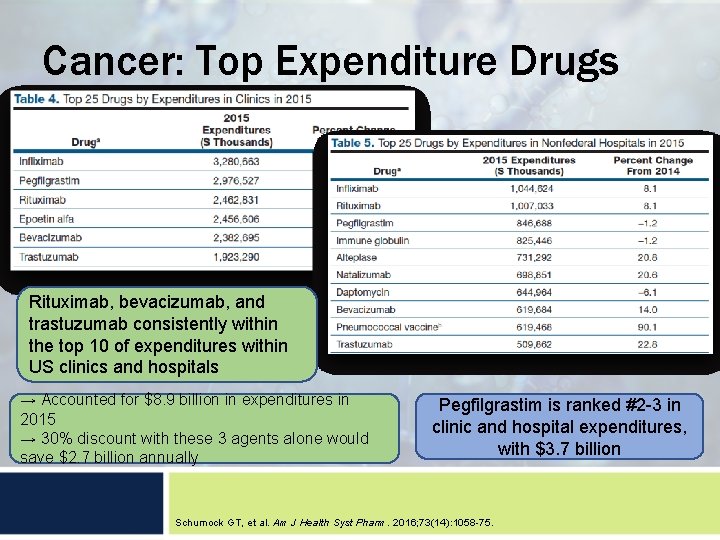
Cancer: Top Expenditure Drugs Rituximab, bevacizumab, and trastuzumab consistently within the top 10 of expenditures within US clinics and hospitals → Accounted for $8. 9 billion in expenditures in 2015 → 30% discount with these 3 agents alone would save $2. 7 billion annually Pegfilgrastim is ranked #2 -3 in clinic and hospital expenditures, with $3. 7 billion Schumock GT, et al. Am J Health Syst Pharm. 2016; 73(14): 1058 -75.
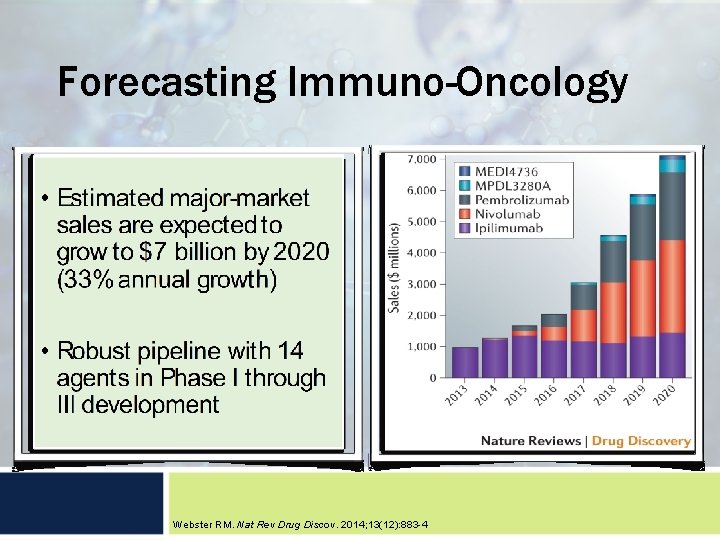
Forecasting Immuno-Oncology Webster RM. Nat Rev Drug Discov. 2014; 13(12): 883 -4

Trends in Oncology Drug Expenditures: 2010 -2014 Schumock GT, et al. Am J Health Syst Pharm. 2015; 72: 717 -736 Schumock GT, et al. Am J Health Syst Pharm. 2014; 71: 482 -499 Hoffman JM, et al. Am J Health Syst Pharm. 2013; 70: 525 -539 Hoffman JM, et al. Am J Health Syst Pharm. 2012; 69: 405 -421.
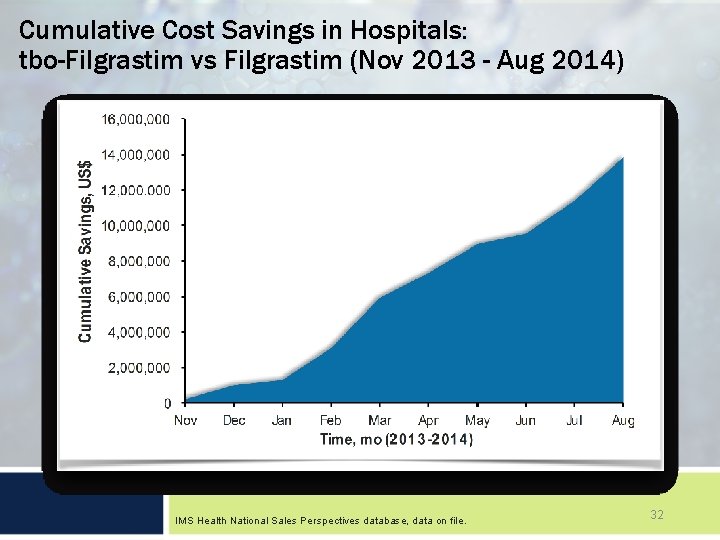
Cumulative Cost Savings in Hospitals: tbo-Filgrastim vs Filgrastim (Nov 2013 - Aug 2014) IMS Health National Sales Perspectives database, data on file. 32
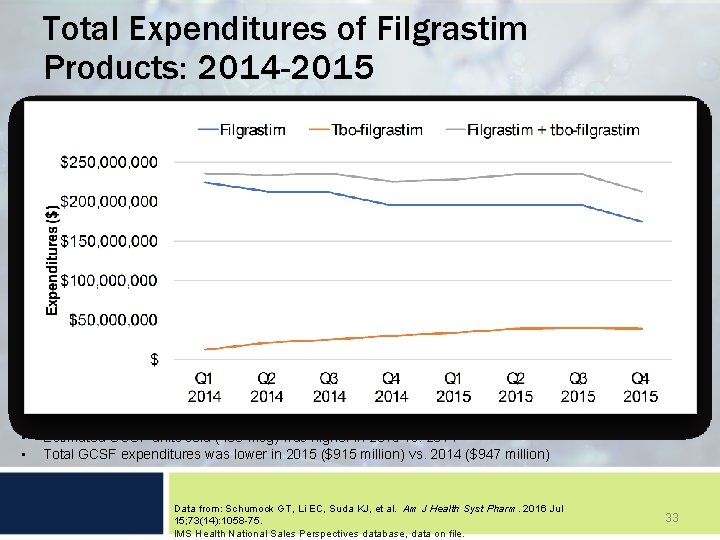
Total Expenditures of Filgrastim Products: 2014 -2015 • • Estimated GCSF units sold (480 -mcg) was higher in 2015 vs. 2014 Total GCSF expenditures was lower in 2015 ($915 million) vs. 2014 ($947 million) Data from: Schumock GT, Li EC, Suda KJ, et al. Am J Health Syst Pharm. 2016 Jul 15; 73(14): 1058 -75. IMS Health National Sales Perspectives database, data on file. 33

Extrapolation Paradigm
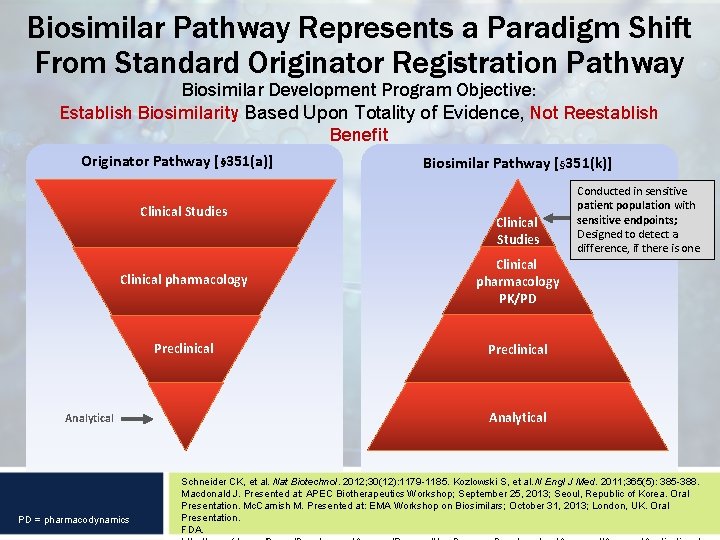
Biosimilar Pathway Represents a Paradigm Shift From Standard Originator Registration Pathway Biosimilar Development Program Objective: Establish Biosimilarity Based Upon Totality of Evidence, Not Reestablish Benefit Originator Pathway [§ 351(a)] Clinical Studies Biosimilar Pathway [§ 351(k)] Clinical Studies Clinical pharmacology PK/PD Preclinical Analytical PD = pharmacodynamics Conducted in sensitive patient population with sensitive endpoints; Designed to detect a difference, if there is one Analytical Schneider CK, et al. Nat Biotechnol. 2012; 30(12): 1179 -1185. Kozlowski S, et al. N Engl J Med. 2011; 365(5): 385 -388. Macdonald J. Presented at: APEC Biotherapeutics Workshop; September 25, 2013; Seoul, Republic of Korea. Oral Presentation. Mc. Camish M. Presented at: EMA Workshop on Biosimilars; October 31, 2013; London, UK. Oral Presentation. FDA.
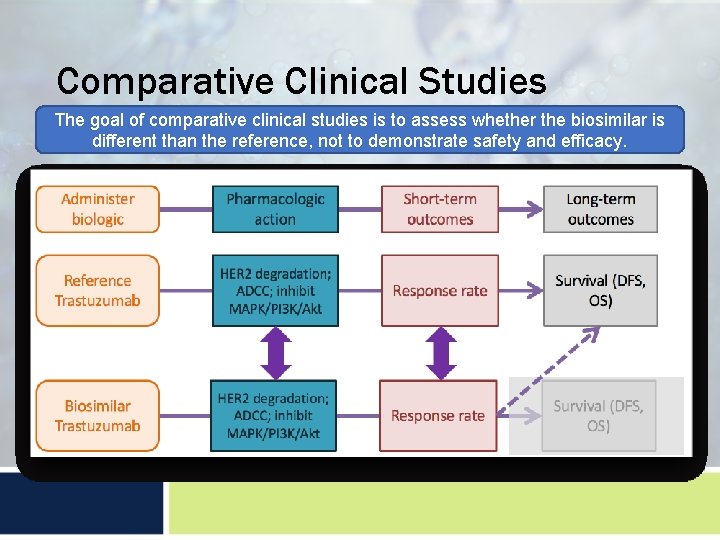
Comparative Clinical Studies The goal of comparative clinical studies is to assess whether the biosimilar is different than the reference, not to demonstrate safety and efficacy.

Extrapolation and FDA-Labeled Indications FDA-approved indications for biosimilars do not require a clinical trial for each indication. Extrapolation is scientifically justified based on specifications of the product made by manufacturer A to product made by manufacturer B. Totality of the evidence (including physiochemical, functional, PK/PD studies) informs the appropriate indications. http: //www. fda. gov/downloads/Advisory. Committees/Committees. Meeting. Materials/Drugs/Oncologic. Drugs. Advis ory. Committee/UCM 428782. pdf

Extrapolation Impact When reviewing a formulary consideration, the paradigm of evaluating Phase 3 RCTs does not apply to biosimilars • The clinical comparison uses an adequately sensitive endpoint in an adequately sensitive population and is not necessarily the relevant clinical endpoint The totality of the evidence should be reviewed, including physiochemical, functional, PK/PD similarity Focus on nonclinical considerations, such as cost, product presentation and user interface, storage, stability, and product supply reliability

Substitution

Interchangeable FDA Designation Requires Different Data § Interchangeable is an FDA designation § Requires different data standards than “biosimilarity” alone § Dedicated switching study and postmarketing monitoring § Study endpoints to evaluate PK/PD, immunogenicity, and safety (efficacy is not adequately sensitive at therapeutic doses) § The actual data package of study design and endpoints depends on the complexity of the molecule and degree of analytical similarity § The product presentation and user interface must be similar to the reference. http: //www. fda. gov/Drugs/Guidance. Compliance. Regulatory. Information/Guidances/ucm 290967. htm http: //www. ncsl. org/research/health/state-laws-and-legislation-related-to-biologic-medicationsand-substitution-of-biosimilars. aspx.

Interchangeable FDA Designation: Additional Considerations § Product with an interchangeable designation may be substituted without intervention of prescribing provider § State substitution laws will impact practice § Any biological product under consideration for substitution must first be approved by FDA as "interchangeable“ http: //www. fda. gov/Drugs/Guidance. Compliance. Regulatory. Information/Guidances/ucm 290967. htm http: //www. ncsl. org/research/health/state-laws-and-legislation-related-to-biologic-medicationsand-substitution-of-biosimilars. aspx.

Pharmacist Substitution State law gives pharmacists the authority to act independently of the prescriber to dispense the lowestcost, equivalent medicinal product Framework • • • Product criteria DAW Communication with prescriber/patient Record keeping Hospital/health system exemption Li EC, et al. J Manag Care Spec Pharm. 2015; 21(7): 532 -39

Enacted Biosimilar Substitution Laws: Sample DAW = dispense as written http: //delcode. delaware. gov/title 24/c 025/sc 06/index. shtml http: //www. flsenate. gov/Session/Bill/2013/0365/Bill. Text/er/PDF https: //leg 1. state. va. us/cgi-bin/legp 504. exe? 131+ful+CHAP 0412 http: //www. foleyhoag. com/publications/alerts-and-updates/2014/june/massachusetts-enacts-new-biosimilars-substitution-law

Common Elements of Interchangeability Rules for Biologics üBiological product under consideration for substitution must first be approved as "interchangeable" by the FDA üPrescriber (physician, specialist, PA, etc) would be able to prevent substitution by stating “dispense as written” or “brand medically necessary” üPrescriber must be notified of any substitution. In 2015 bills, language adjusted to say “communicate with” üPatient must be notified that a substitute or switch was made. In some cases, state law requires patient consent prior to switch State-to-state variations possible National Conference of State Legislatures

Pharmacovigilance

Biosimilar Pharmacovigilance Risk Identification and Characterization FDA Approval • • Pharmacovigilance Risk Minimization • Practical to encourage health care provider reporting • Real-time data • Ensure traceability • Health care provider communication • Recalls and alerts • REMS? Naming Integration into EHR Drug codes: HCPCS, NDC, etc Prospective registries Zuñiga L, Calvo B. Pharmacoepidemiol Drug Saf. 2010 Jul; 19: 661 -9. Casadevall N, et al. Expert Opin Biol Ther. 2013; 13: 1039 -47.
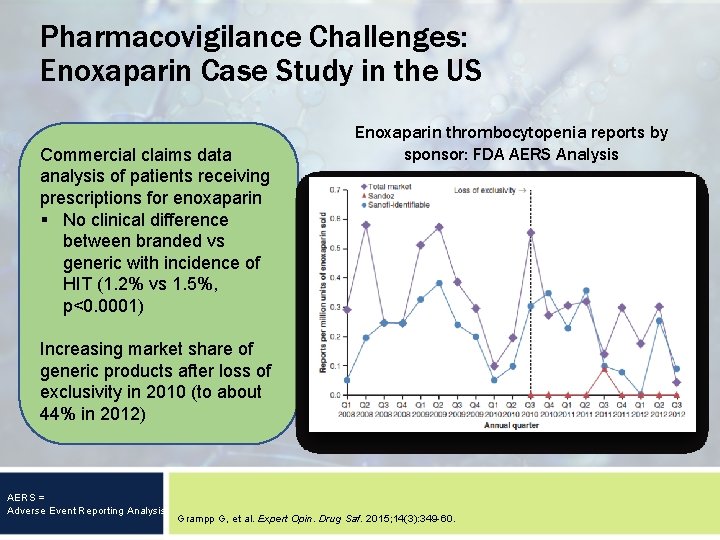
Pharmacovigilance Challenges: Enoxaparin Case Study in the US Commercial claims data analysis of patients receiving prescriptions for enoxaparin § No clinical difference between branded vs generic with incidence of HIT (1. 2% vs 1. 5%, p<0. 0001) Enoxaparin thrombocytopenia reports by sponsor: FDA AERS Analysis Increasing market share of generic products after loss of exclusivity in 2010 (to about 44% in 2012) AERS = Adverse Event Reporting Analysis Grampp G, et al. Expert Opin. Drug Saf. 2015; 14(3): 349 -60.

FDA Guidance on Naming Goal: facilitate pharmacovigilance and prevent inadvertent substitution INN + random 4 -letter suffix for all biologics • • • Unique Devoid of meaning 4 lower case letters, at least 3 are distinct Nonproprietary Examples • replicamab-cznm • replicamab-hixf Benefits • Common INN will group similar biologics in electronic systems • Having suffix for all products reduces perception that biosimilar is inferior to reference product http: //www. fda. gov/downloads/drugs/guidancecomplianceregulatoryinformation/guidances/ucm 459987. pdf

Biosimilar Pharmacovigilance: Role of the Provider Monitor and report • Adverse events: FDA Medwatch • Medication errors Correct attribution of safety event • What was ordered vs what did the patient receive? • Maintenance of electronic medical record • Bar code administration • Medication reconciliation • Consider transitions of care

Tips for Practice Be aware of which biosimilar product is being prescribed and used Prescribe using the proper name or trade name with suffix Contribute to local pharmacovigilance efforts (registries) Monitor long-term safety (pharmacovigilance) Encourage transparency in drug characterization Dörner T, et al. Ann Rheum Dis. 2016; 75(6): 974 -982

Thank you for participating.
- Slides: 51