Factors that affect electron energies Shielding provided by
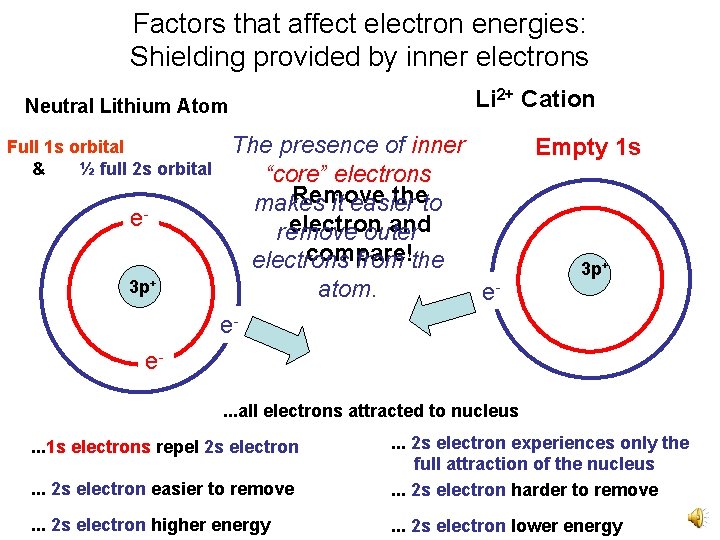
Factors that affect electron energies: Shielding provided by inner electrons Neutral Lithium Atom Full 1 s orbital & ½ full 2 s orbital e 3 p+ Li 2+ Cation The presence of inner “core” electrons Remove theto makes it easier electron and remove outer compare! electrons from the atom. ee- Empty 1 s 3 p+ e. . . all electrons attracted to nucleus . . . 2 s electron easier to remove . . . 2 s electron experiences only the full attraction of the nucleus. . . 2 s electron harder to remove . . . 2 s electron higher energy . . . 2 s electron lower energy . . . 1 s electrons repel 2 s electron
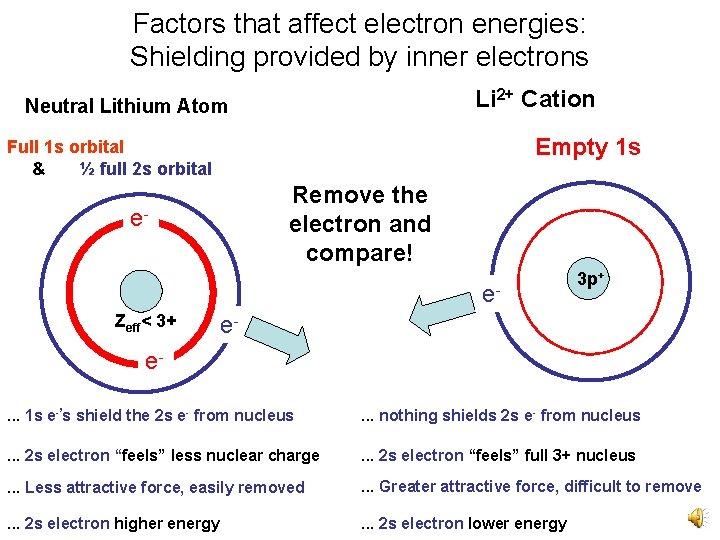
Factors that affect electron energies: Shielding provided by inner electrons Li 2+ Cation Neutral Lithium Atom Empty 1 s Full 1 s orbital & ½ full 2 s orbital Remove the electron and compare! e- e. Zeff< 3+ 3 p+ e- e. . . 1 s e-’s shield the 2 s e- from nucleus . . . nothing shields 2 s e- from nucleus . . . 2 s electron “feels” less nuclear charge . . . 2 s electron “feels” full 3+ nucleus . . . Less attractive force, easily removed . . . Greater attractive force, difficult to remove . . . 2 s electron higher energy . . . 2 s electron lower energy
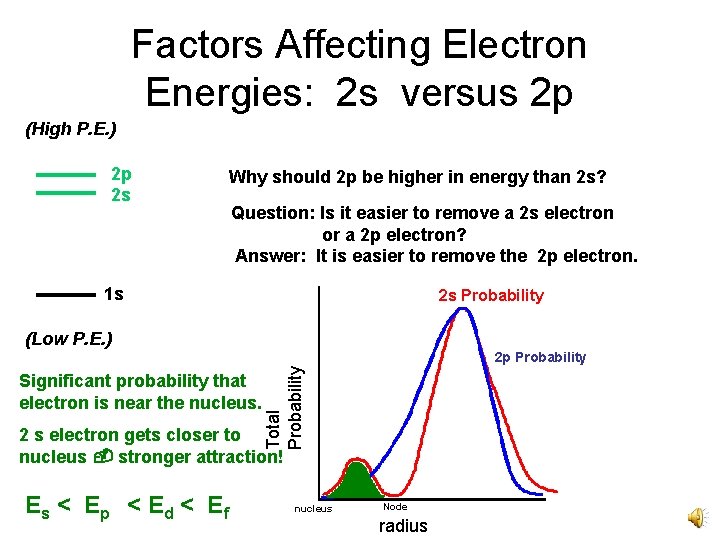
Factors Affecting Electron Energies: 2 s versus 2 p (High P. E. ) 2 p 2 s Why should 2 p be higher in energy than 2 s? Question: Is it easier to remove a 2 s electron or a 2 p electron? Answer: It is easier to remove the 2 p electron. 1 s 2 s Probability (Low P. E. ) Significant probability that electron is near the nucleus. Total Probability 2 p Probability 2 s electron gets closer to nucleus stronger attraction! Es < E p < E d < E f nucleus Node radius
- Slides: 3