Factor Label Conversions Dimensional Analysis ORGANIZED APPROACH TO

Factor Label/ Conversions/ Dimensional Analysis ORGANIZED APPROACH TO PROBLEM SOLVING USING UNITS

1. ALWAYS START WITH "? units wanted” ? Units wanted =

2. Write Given quantity on top ? Units wanted = Given quantity

3. Write matching conversion factor on bottom of next parenthesis ? Units wanted = Given quantity Matching conversion factor

4. Put wanted unit of conversion factor on top! ? Units wanted = Wanted unit of Given quantity conversion factor Matching conversion factor

5. Multiply everything on top, and divide by everything on bottom! ? Units wanted = Wanted unit of Given quantity conversion factor Matching conversion factor *In calculator type: (multiply everything on top) / (multiply everything on bottom) = your answer with units wanted!

*You must have the same unit on top in the first parenthesis, as you do in the bottom of the next parenthesis!! *Diagonal Units: must cancel out! ? Units wanted = given quantity Units wanted Matching conversion factor: MUST be in SAME UNIT as your Given Quantity!!!!!

• You can also do this with multiple conversion factors! ? Units Given quantity Some unit Units wanted! wanted = Matching Some unit conversion factor • Start with same steps as before, but your first conversion factor may not have the wanted unit on top. • You may need to use many conversion factors to get to the unit wanted!!

1 year = ______ mins ? mins = 1 year 365 days 24 hrs 60 min 1 hr 1 year 1 day

1 Molex GFMx 1 Molex

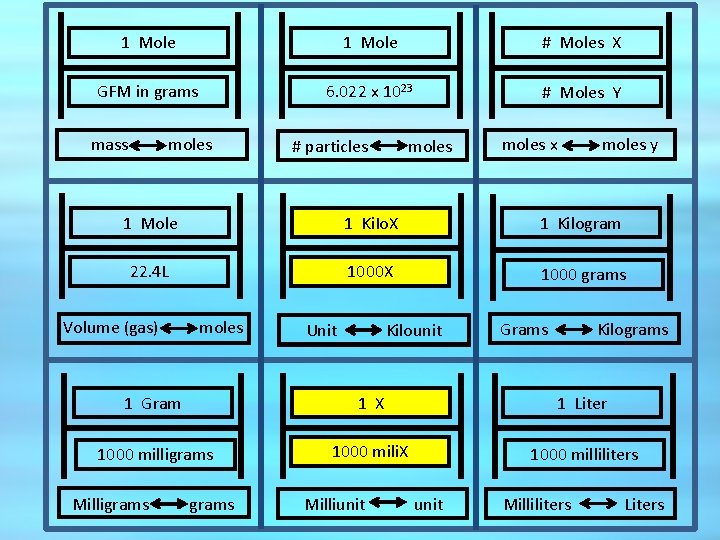
1 Mole # Moles X GFM in grams 6. 022 x 1023 # Moles Y mass moles # particles moles x moles y 1 Mole 1 Ki. Io. X 1 Kilogram 22. 4 L 1000 X 1000 grams Volume (gas) moles Unit Kilounit Grams Kilograms 1 Gram 1 X 1 Liter 1000 milligrams 1000 mili. X 1000 milliliters Milligrams Milliunit Milliliters Liters

Example conversion Na 2 SIO 3(s) + 8 HF(aq) H 2 Si. F 6(aq) + 2 Na. F(aq) + 3 H 2 O(l) How many grams of Na 2 Si. O 3 can react with 0. 800 g of HF?

Na 2 SIO 3(s) + 8 HF(aq) H 2 Si. F 6(aq) + 2 Na. F(aq) + 3 H 2 O(l) How many grams of Na 2 Si. O 3 can react with 0. 800 g of HF? Mass in grams 1 Molex GFMx 1 Moley 2 Molex GFMy 1 Moley
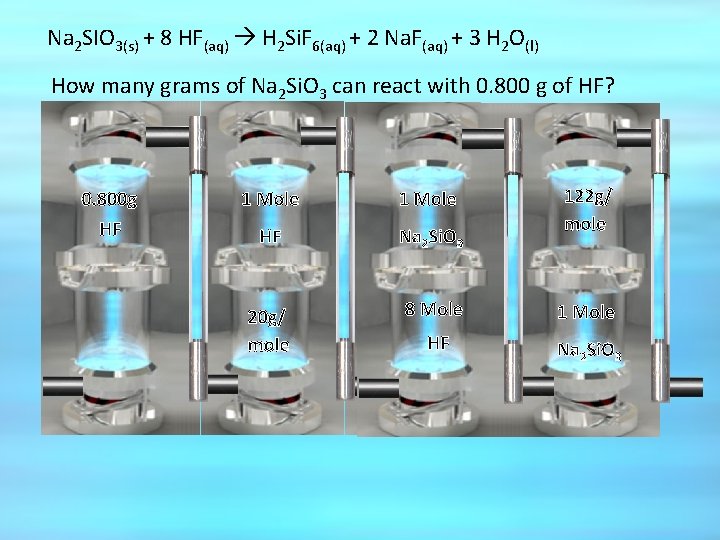
Na 2 SIO 3(s) + 8 HF(aq) H 2 Si. F 6(aq) + 2 Na. F(aq) + 3 H 2 O(l) How many grams of Na 2 Si. O 3 can react with 0. 800 g of HF? 122 g/ mole 0. 800 g 1 Mole HF HF Na 2 Si. O 3 20 g/ mole 8 Mole 1 Mole HF Na 2 Si. O 3

Mass in grams x 1 Moley GFMx 2 Molex 1 Moley Mass in gramsy
- Slides: 16