Factor Eight Inhibitor Bypassing Activity FEIBA for the

Factor Eight Inhibitor Bypassing Activity (FEIBA) for the Rapid Reversal of Major Bleeding in Patients with Warfarin Induced Coagulopathy: A Pilot Study David Barounis, MD 1 Catherine Knight, MD 2 Ben-Paul Umunna, MD 2 Mary Hormese, Pharm. D, BCPS 2 Elise Lovell, MD 2 1 Stanford University, Stanford, CA; 2 Advocate Christ Medical Center, Oak Lawn, IL

No financial disclosures or conflict of interest

Background • Fresh frozen plasma (FFP) + vitamin K to reverse major bleeding due to warfarin associated coagulopathy • FFP shortcomings ▫ ▫ ▫ incomplete reversal time delay for ABO compatibility, thawing large volume time to transfuse risk of TRALI • Prothrombin complex concentrates (PCC) recommended since 2012

FEIBA • • • Plasma derived 4 coagulation factors (II, VII, IX, X) FDA approved in hemophilia Small volume No blood typing/thawing

Clinical Experience with FEIBA • Retrospective comparison of 72 pts receiving FEIBA to 69 patients receiving FFP • Median time to reversal of INR < 1. 4 was 2 hours in FEIBA group, 25 hours in FFP group • 7% TAE, 22% mortality in FEIBA group Wojcik C, Schymik ML, Cure EG. Activated prothrombin complex concentrate factor VIII inhibitor bypassing activity (FEIBA) for the reversal of warfarininduced coagulopathy. Int J Emerg Med 2009; 2: 217 -225.

Study Purpose • To evaluate the efficacy and safety of FEIBA and IV vitamin K for the reversal of warfarinassociated coagulopathy in patients with major hemorrhage

Hypothesis • The use of FEIBA and IV vitamin K will result in the rapid reversal of warfarin associated coagulopathy in patients with major bleeding • Adverse event rate will be low

Methods - Study Setting • Tertiary care suburban community teaching hospital • 100, 000 ED visits per year • 700 inpatient beds

Methods - Study Design • Ongoing prospective, observational study • ED patients on warfarin presenting with major bleeding • INR ≥ 5. 0 1000 U of FEIBA • INR < 5. 0 500 U of FEIBA IV • 10 mg IV vitamin K • Repeat INR 30 minutes, 4 and 24 hours

Inclusion Criteria • Age > 18 • Present to the ED with major hemorrhage while on warfarin ▫ life or limb threatening bleed or ▫ bleed in critical location (intracranial, ophthalmic, intraspinal) or ▫ 2 gram fall in hemoglobin • Initial INR >1. 5

Exclusion Criteria • • Coagulopathy not due to warfarin On warfarin, but INR ≤ 1. 5 No major hemorrhage Received additional reversal agents prior to/in ED (FFP, a. Factor VII, PCCs, vitamin K PO/SQ/IM)

Methods of Measurement • All eligible patients identified by M/W/F review of FEIBA dispensed to ED

Outcome Measures • Primary Outcome: Time to INR ≤ 1. 5 • Secondary outcomes: ▫ ▫ Thrombotic adverse events, allergic reaction FEIBA dose required to reverse Units of PRBCs transfused Mortality

Statistical Analysis • Reporting of descriptive measures (means, medians, IQRs, as appropriate)

Results • Between 2/8/2013 and 8/30/2013, 44 ED patients received FEIBA • 9 did not meet inclusion criteria ▫ 6 not major bleed ▫ 3 INR ≤ 1. 5 • 14 patients excluded ▫ ▫ ▫ 6 FFP or alternate route vitamin K 4 died before consent obtained 1 no POA 1 withdrew consent 2 ethical issue to approach for consent

Results • 21 patients enrolled ▫ 11 CNS bleed, 8 GI bleed, 1 Hematuria, 1 Pulmonary hemorrhage • • Mean initial INR was 5. 5 16/21 patients admitted to ICU for at least 1 day Achieved INR ≤ 1. 5 in all patients Mean time to INR ≤ 1. 5 was 127 minutes ▫ 1 patient’s repeat INR was drawn ~12 hrs after FEIBA ▫ if this patient excluded, mean time to INR ≤ 1. 5 was 98 minutes

Results • Four (19%) patients died (none with TAE) ▫ 2 GI bleed ▫ 2 CNS bleed • Four thrombotic adverse events (TAE) in 3 (14%) patients ▫ 2 ischemic CVAs ▫ 2 extremity DVTs

Results: Mortality Age Diagnosis Cause of death Hospital Day 78 yo male CNS bleed herniation/withdr 2 awal of care 41 yo male GI bleed/arrest 12 76 yo male GI bleed/arrest 18 88 yo female CNS bleed withdrawal of care 8
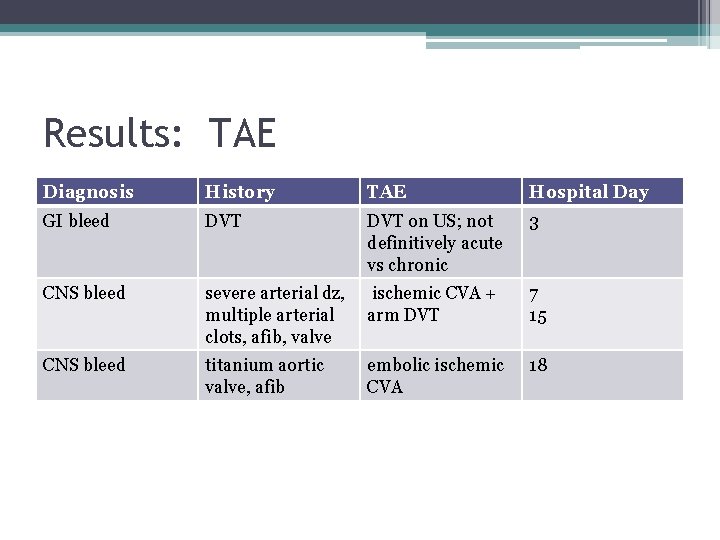
Results: TAE Diagnosis History TAE Hospital Day GI bleed DVT on US; not definitively acute vs chronic 3 CNS bleed severe arterial dz, ischemic CVA + multiple arterial arm DVT clots, afib, valve 7 15 CNS bleed titanium aortic valve, afib 18 embolic ischemic CVA

TAE: Background • Kcentra: 9% TAE vs. 5. 5% TAE in FFP group (ns) • Wojcik: 1. 4% TAE in FFP group • MEDENOX trial: 15% rate of DVT in patients admitted without SQ enoxaparin • FEIBA in hemophilia: TAE rate of 4 per 100, 000 infusions

Limitations • • Single center Observational study design Disease oriented primary outcome 4 patients died before consent obtained (impacts mortality rate) • Contribution of FEIBA to thrombotic adverse events uncertain

Conclusions • FEIBA and IV vitamin K result in rapid reversal of warfarin-induced coagulopathy in patients with major bleeding • Thrombotic adverse event rate was 14%

23

What about Kcentra? • FDA approved April 30, 2013 • PCC given along with Vitamin K ▫ Factors II, VII, IX, X, with proteins C and S, antithrombin III ▫ Includes heparin • Dosing based on INR, body weight • No repeat dosing • Known risks

Cost of FEIBA vs FFP • Feiba: $1. 48 per unit ▫ Hospital cost: $740 for 500 units, $1480 for 1000 units ▫ Patient cost: $3, 496 and $5, 920 • FFP: $65 -70 per unit, start with 10 cc/kg, 200 cc in unit of FFP, so 100 kg pt needs 5 units FFP = $350

4 Patients with 3 TAEs • Leg DVT, 2 ischemic CVAs, and arm DVT. One pt with both arm DVT and CVA. • Pt with leg DVT admitted with GI Bleed, had h/o DVT. US hospital day 3 with DVT which could not be definitively called acute versus chronic. • Pt with arm DVT and CVA admitted with acute on chronic subdural hematoma, h/o severe arterial disease/multiple peripheral arterial clots, also mitral valve replacement, afib • Pt with other CVA (thought to be embolic) admitted with SAH, had h/o titanium aortic valve and afib.

Clinical Experience with FEIBA • FEIBA usage summarized in 16 patients at community hospital • 87% of patients survived to discharge • No signs or symptoms of TAE Stewart WS, Pettit H. Experiences with an activated 4 -factor prothrombin complex concentrate (FEIBA) for reversal of warfarin-related bleeding. AJEM 2013; 31: 1251 -1254.
- Slides: 27