Facilities Development Division Californias Building Department for Hospitals

Facilities Development Division California’s Building Department for Hospitals Paul A. Coleman, Architect, Deputy Director Chris Tokas, S. E. , Deputy Division Chief Gordon Oakley, Fire Marshal, Deputy Division Chief Roy Lobo, Ph. D. , S. E. , Principal Structural Engineer Glenn Gall, Supervisor, Building Standards Unit Nanci Timmins, Fire Marshal, Chief Fire Life Safety Officer California Healthcare Association May 9, 2017 1

Facilities Development Division California’s Building Department for Hospitals Plan Review and Field Performance Update 2
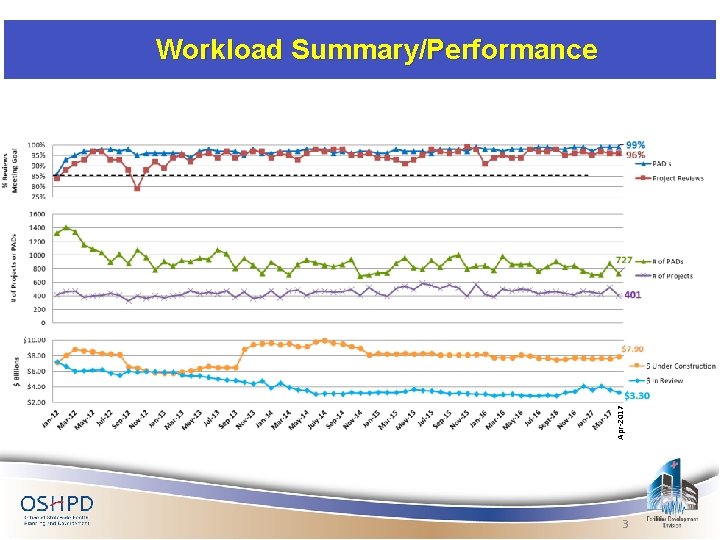
Apr-2017 Workload Summary/Performance 3
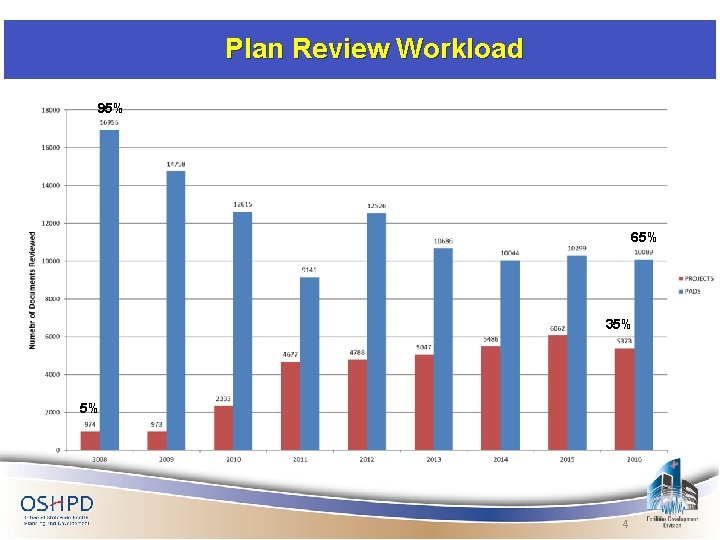
Plan Review Workload 95% 65% 35% 5% 4
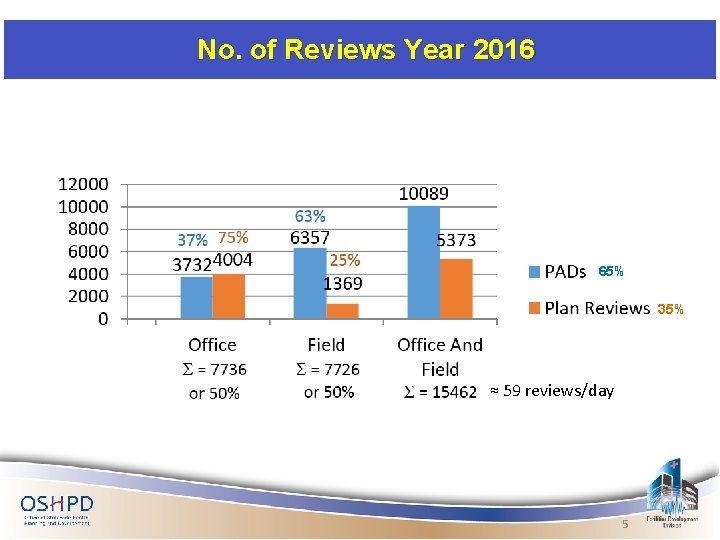
No. of Reviews Year 2016 65% 35% ≈ 59 reviews/day 5
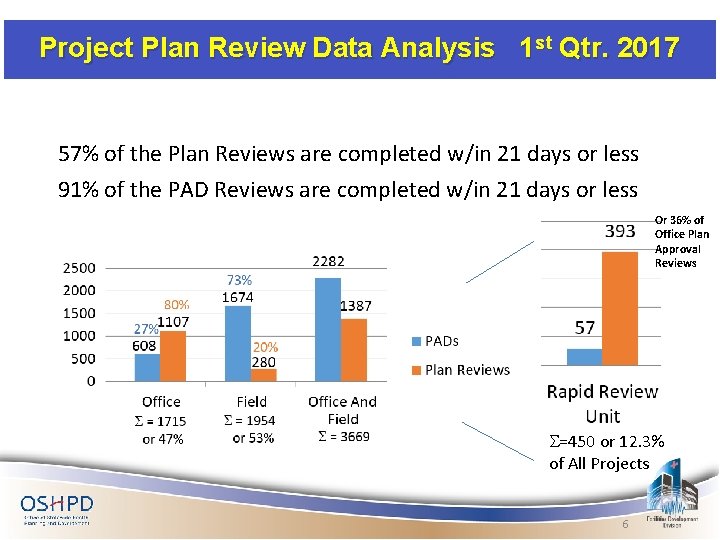
Project Plan Review Data Analysis 1 st Qtr. 2017 57% of the Plan Reviews are completed w/in 21 days or less 91% of the PAD Reviews are completed w/in 21 days or less Or 36% of Office Plan Approval Reviews S=450 or 12. 3% of All Projects 6
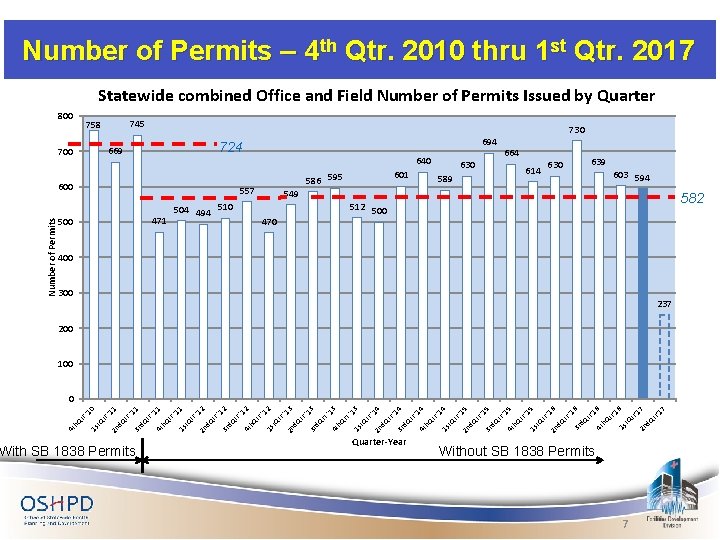
Number of Permits – 4 th Qtr. 2010 thru 1 st Qtr. 2017 Statewide combined Office and Field Number of Permits Issued by Quarter 800 745 758 730 724 669 700 640 471 500 589 630 614 603 594 549 582 512 500 510 504 494 664 630 601 586 595 557 470 400 300 237 200 100 With SB 1838 Permits 7 r'1 17 7 d. Q t tr' Without SB 1838 Permits 2 n t. Q 1 s 6 tr' 16 h. Q r'1 4 t 6 16 d. Q t 3 r r ' '1 d. Q t tr 2 n '1 5 t. Q tr 1 s '1 5 h. Q 4 t 5 15 tr 3 r d. Q r ' '1 d. Q t tr 2 n '1 4 1 s t. Q 4 tr h. Q tr '1 Quarter-Year 4 t 4 14 3 r d. Q r ' '1 tr d. Q t 2 n '1 3 1 s t. Q 3 rt '1 h. Q 4 t 13 r ' d. Q 3 r 3 '1 tr d. Q t 2 n '1 2 1 s t. Q 2 tr '1 h. Q 4 t 12 r ' d. Q 3 r 2 '1 tr d. Q t 2 n '1 1 1 s t. Q 1 tr '1 h. Q 4 t 11 r ' d. Q 3 r 1 '1 tr d. Q t 2 n t. Q 1 s h. Q tr '1 0 0 4 t Number of Permits 694
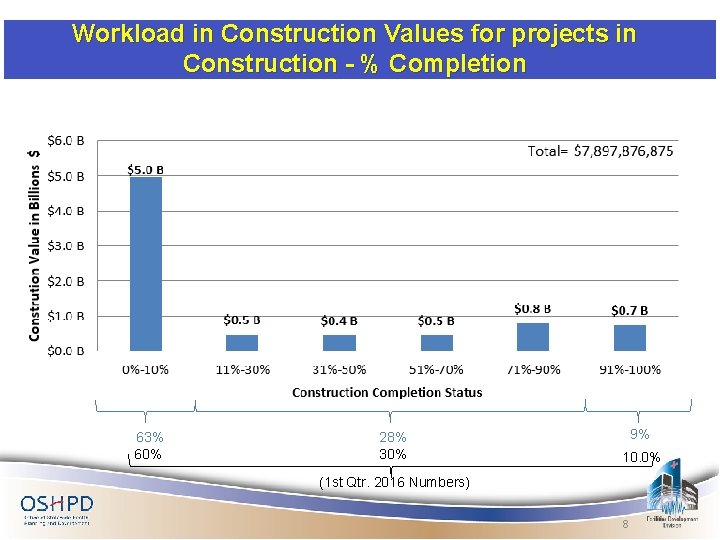
Workload in Construction Values for projects in Construction - % Completion 63% 60% 28% 30% 9% 10. 0% (1 st Qtr. 2016 Numbers) 8
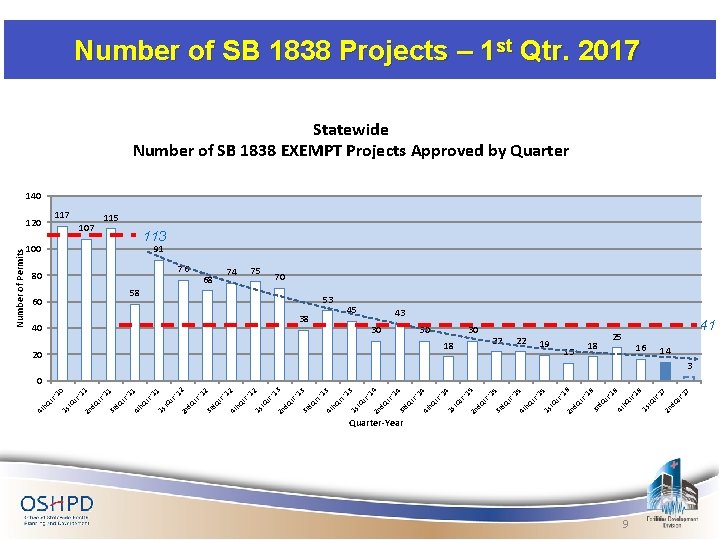
Quarter-Year 9 d. Q t 7 r'1 17 tr' 16 2 n t. Q 25 1 s 6 tr' 16 h. Q r'1 16 18 4 t d. Q t r ' 13 3 r 6 '1 tr 19 d. Q t 2 n '1 5 tr 5 22 t. Q 1 s h. Q tr '1 22 4 t 15 18 d. Q r ' 30 3 r 5 '1 tr 30 d. Q t 2 n '1 4 tr 4 20 t. Q 1 s h. Q tr '1 14 30 4 t d. Q r ' 4 '1 45 3 r d. Q t tr 38 2 n '1 3 rt 3 53 t. Q 1 s h. Q rt '1 13 58 4 t d. Q r ' 40 3 r 3 '1 tr 75 d. Q t 2 n '1 2 tr 2 74 t. Q 1 s h. Q tr '1 12 68 4 t d. Q r ' 76 3 r 2 '1 tr '1 1 80 d. Q t 2 n t. Q tr 1 100 1 s h. Q 60 4 t tr '1 11 107 d. Q r ' 117 3 r 1 '1 '1 0 tr d. Q t 2 n t. Q 120 1 s tr h. Q 4 t Number of Permits Number of SB 1838 Projects – 1 st Qtr. 2017 Statewide Number of SB 1838 EXEMPT Projects Approved by Quarter 140 115 91 113 70 43 41 14 3 0
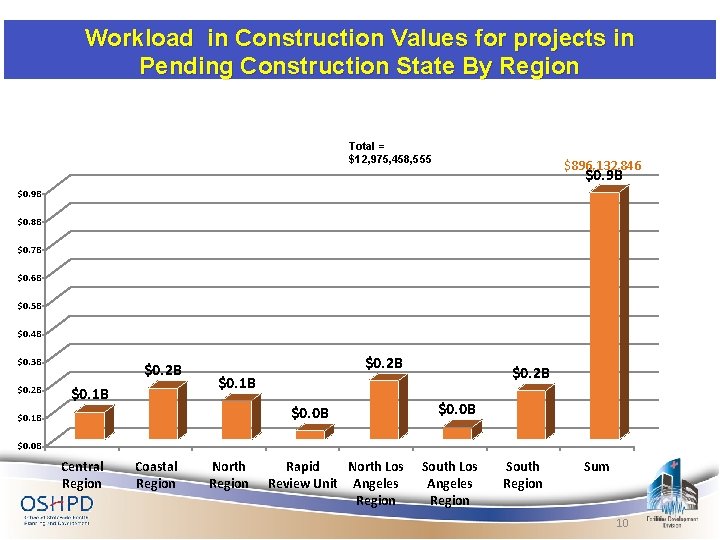
Workload in Construction Values for projects in Pending Construction State By Region Total = $12, 975, 458, 555 $896, 132, 846 $0. 9 B $0. 8 B $0. 7 B $0. 6 B $0. 5 B $0. 4 B $0. 3 B $0. 2 B $0. 1 B $0. 0 B Central Region Coastal Region North Region Rapid North Los Review Unit Angeles Region South Los Angeles Region South Region Sum 10
![OSHPD Special Seismic Certification Preapproval (OSP) Overall OSP Review Status OSP Status [CATEGORY NAME] OSHPD Special Seismic Certification Preapproval (OSP) Overall OSP Review Status OSP Status [CATEGORY NAME]](http://slidetodoc.com/presentation_image_h/9fa8d62a85a580ff4e00cc001f2f50f4/image-11.jpg)
OSHPD Special Seismic Certification Preapproval (OSP) Overall OSP Review Status OSP Status [CATEGORY NAME] [PERCENTAGE] Approved Expired Remarked Total 517 Approved 353 Expired 113 Remarked 31 In-Review 20 In-Review 11

Expired OSPs on December 31, 2016 Ø 155 Expired OSPs on December 31, 2016 Ø OSPs expired on December 31, 2016 can be used for all projects submitted prior to January 1, 2017 Ø 52 OSP renewal and 24 new OSPs approved since January 1, 2017 Ø OSP review is still meeting 60/30/30 turn around goals 12

OSP Review Workload Ø In a Typical Year OSHPD receives 75 OSPs Ø In 2016, OSHPD received 157 OSPs (more than double the typical) Ø In 2017, OSHPD has already received 75 OSPs & is on pace to receive about 300 OSPs (about 4 -times the typical) Ø Currently there are 51 OSPs either in review at OSHPD or in correction with RDP 13
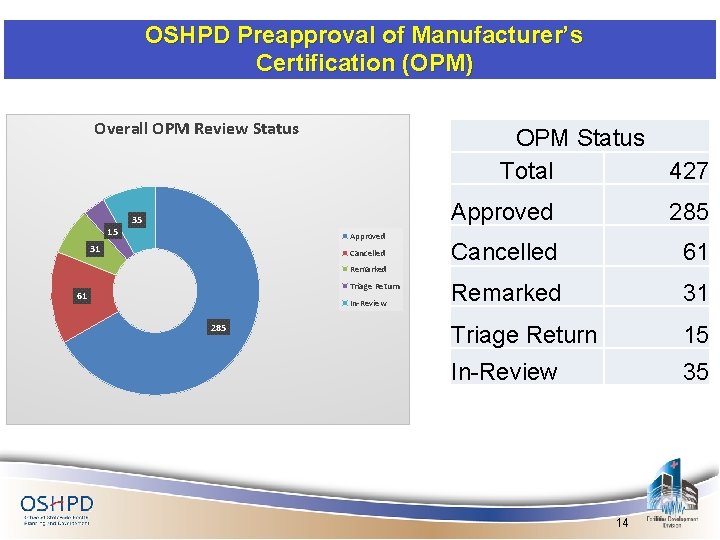
OSHPD Preapproval of Manufacturer’s Certification (OPM) Overall OPM Review Status 15 OPM Status Total 35 Approved 31 Cancelled Remarked Triage Return 61 In-Review 285 427 Approved 285 Cancelled 61 Remarked 31 Triage Return 15 In-Review 35 14

OPM Review Status Ø 285 OPMs approved including 6 -Distribution System OPMs (Mason, B-line/Tolco, Sausee, Erico & Anvil) Ø Currently there are 81 -OPMs either in review at OSHPD or in correction with RDP including 3 -distribution system OPMs (ISAT, Unistrut/Power-Strut) Ø OPM review is still meeting 80/40/40 turn around goals 15

Facilities Development Division California’s Building Department for Hospitals Seismic Compliance Update
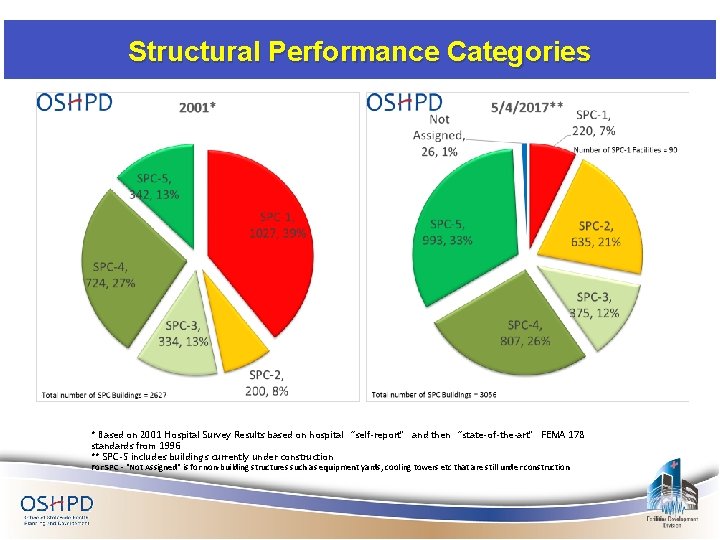
Structural Performance Categories * Based on 2001 Hospital Survey Results based on hospital “self-report” and then “state-of-the-art” FEMA 178 standards from 1996 ** SPC-5 includes buildings currently under construction For SPC - "Not Assigned" is for non-building structures such as equipment yards, cooling towers etc that are still under construction
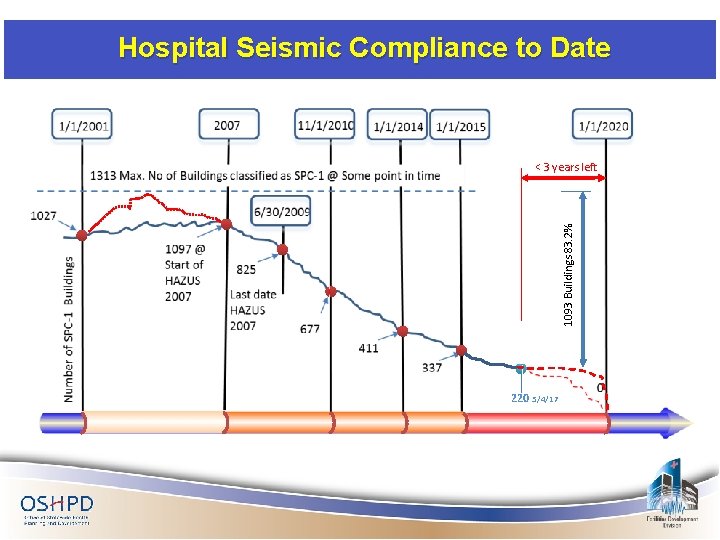
Hospital Seismic Compliance to Date 1093 Buildings 83. 2% < 3 years left 220 5/4/17

SPC -1 Buildings = 220 SPC-1 Buildings with Approved SB 90 Extensions 166 SPC-1 Buildings with only SB 1661/AB 2557/SB 81 Extensions 18 SPC-1 Buildings with only SB 306 Extensions 26 SPC-1 Buildings that currently have an extension 210 SPC-1 Buildings that do not have any extension beyond 2013* 10 Total 220 *No acute care services – some of these building have removal-of-acute-care-services projects – all have been sent a letter urging removing building from inventory.

SPC-1 Buildings with Extensions beyond 1/1/2013 SPC-1 Buildings with expired SB 90 Extensions 16 SPC-1 Buildings with expired SB 1661 Extensions 18 SPC-1 Buildings that have expired extensions 34 SPC-1 Buildings with expired extensions that have occupancy/construction final - Remaining* * 2 of 9 have active revisions in OSHPD/FDD for extending the SB 90 extension. 2 are in process of getting Certificate of Occupancy. 25 9

Buildings Without Approved Extensions Condition applied to 12 buildings with no extensions, 18 expired SB 1661 extensions, 4 expired SB 90 extensions thus far. A letter was sent to all facilities where condition was applied.
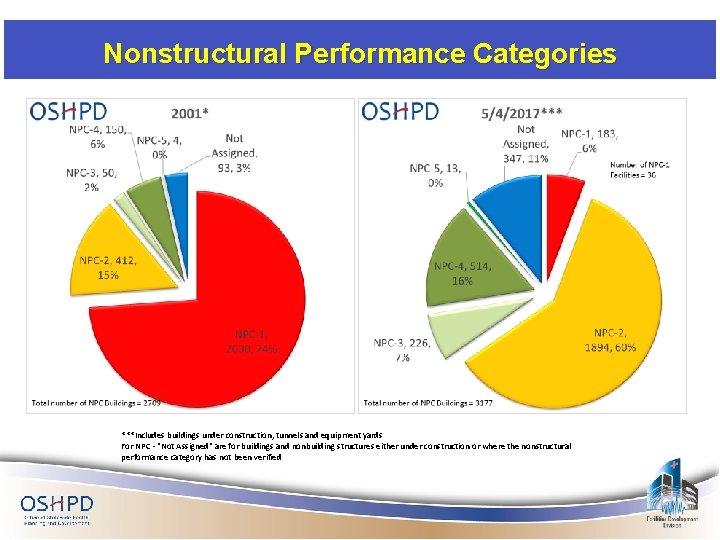
Nonstructural Performance Categories ***Includes buildings under construction, tunnels and equipment yards For NPC - "Not Assigned" are for buildings and nonbuilding structures either under construction or where the nonstructural performance category has not been verified

Facilities Development Division California’s Building Department for Hospitals Project Closure and Invoicing
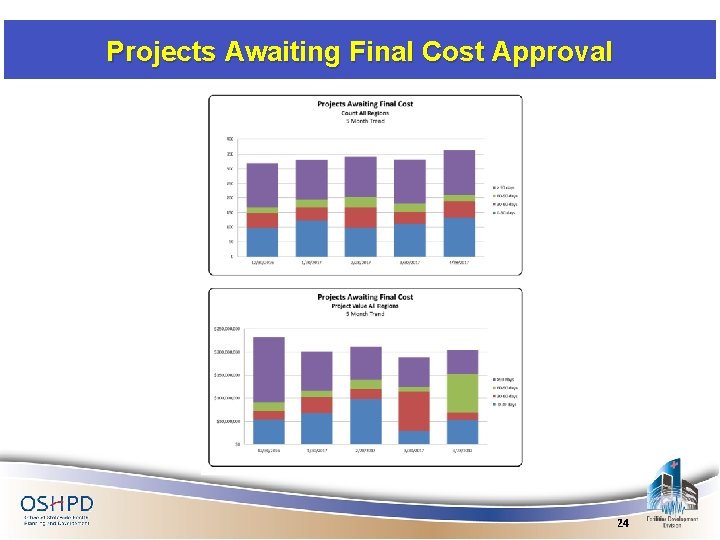
Projects Awaiting Final Cost Approval 24
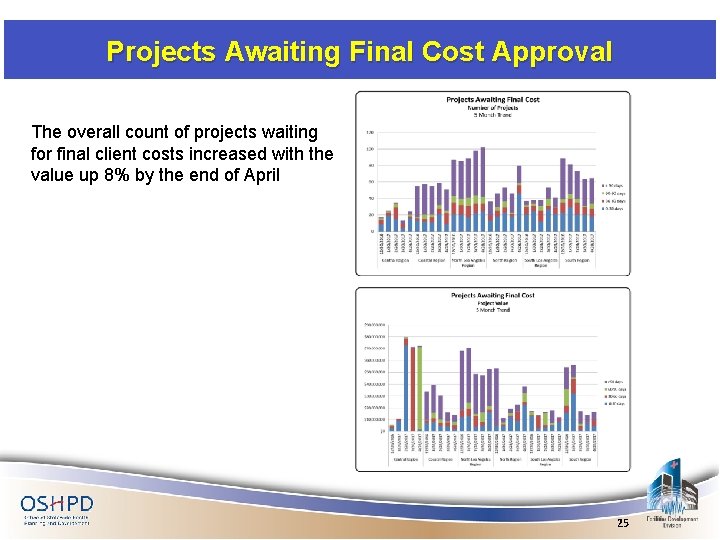
Projects Awaiting Final Cost Approval The overall count of projects waiting for final client costs increased with the value up 8% by the end of April 25
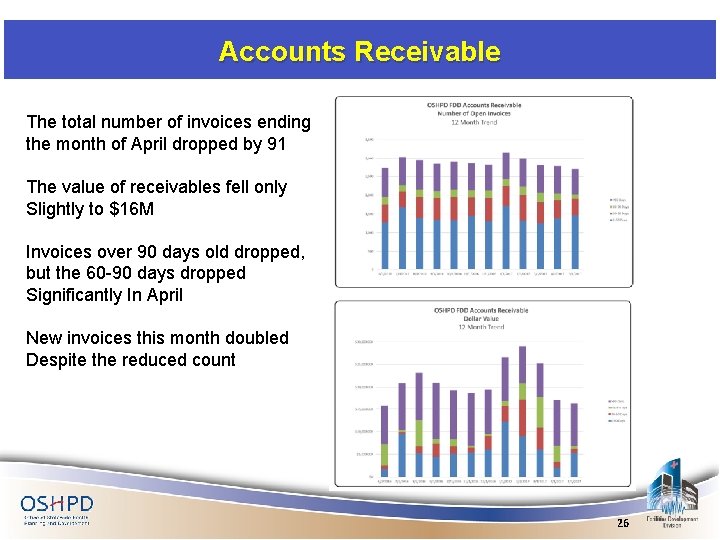
Accounts Receivable The total number of invoices ending the month of April dropped by 91 The value of receivables fell only Slightly to $16 M Invoices over 90 days old dropped, but the 60 -90 days dropped Significantly In April New invoices this month doubled Despite the reduced count 26
Invoice Reports https: //www. oshpd. ca. gov/FDD/Forms/Keyplans/index. html https: //www. oshpd. ca. gov/FDD/Forms/Keyplans/index. SNFs. html 27
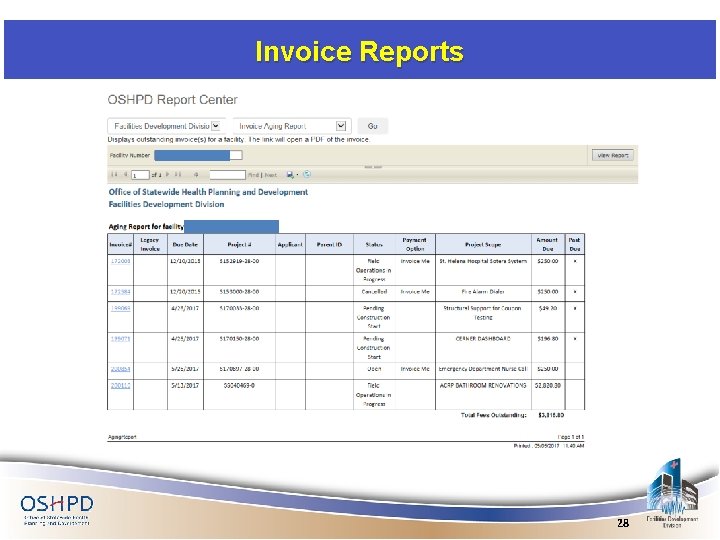
Invoice Reports 28

Facilities Development Division California’s Building Department for Hospitals e. Services

e. RAD 30

e. RAD The Resolution of Approved Documents (RAD) process is an escalation path for OSHPD Field or Office Staff to resolve seeming errors and omissions on the approved construction documents prior to directing changes to the approved documents. 31

Functional Program - Pharmacy Hospitals that perform sterile compounding must meet new regulatory requirements from the California State Board of Pharmacy. Beyond updating processes and procedures, hospitals will be required to improve or reconfigure facilities for ventilation, install new equipment for sterility and ensure employee protections. This process is new and requires collaboration, coordination and a focused approach for successful implementation. CHA has gathered representatives from three California government entities — Board of Pharmacy, Office of Statewide Health Planning and Development (OSHPD) Facilities Development Division, and Department of Public Health Licensing and Certification Program — to share insights crucial to compliant implementation of the sterile compounding regulations. 32

Functional Program - Pharmacy Each pharmacy project that involves sterile compounding requires a Functional Program to be included with the project submittal in accordance with California Administrative Code 7 -119. The Functional Program will automatically be delivered to CDPH for their review and input using an “Ad Hoc Task” in e. SP. When the Ad Hoc Task is created, CDPH receives an email notification with a hyperlink to the Functional Program that can be downloaded and reviewed. 33

IOR Workload Report Expedited Building Permits have been added to the IOR Individual Workload Report to better reflect all of the work an IOR is responsible for. 34

Plan Submittal Reminder With the code cycle change this year there were many clients who submitted an application for a new project but never submitted or uploaded construction document – essentially trying to ‘lock-in’ the 2013 CBSC code. CBC Section 105. 3 requires construction documents to be submitted at the same time the application is submitted. There are currently 401 open records, some dating back 4½ years. A concerted effort is underway to close all of these records due to inactivity. 35

Plan Submittal Reminder A new Code Application Notice or Policy Intent Notice will be developed to address this problem. In the mean time, a notice has been added to the validation message automatically generated when a new application is successfully submitted through e. SP. 36

ACD Initiated By In an effort to better understand the number of ACDs submitted on projects, a new field has been added to the application to capture the reason the ACD was initiated. This information will also be used to develop future training programs. 37

Document Upload Confirmation Many clients have requested an email notification confirming the upload of documents to e. SP. Within the next few weeks this feature will be added to e. SP. 38
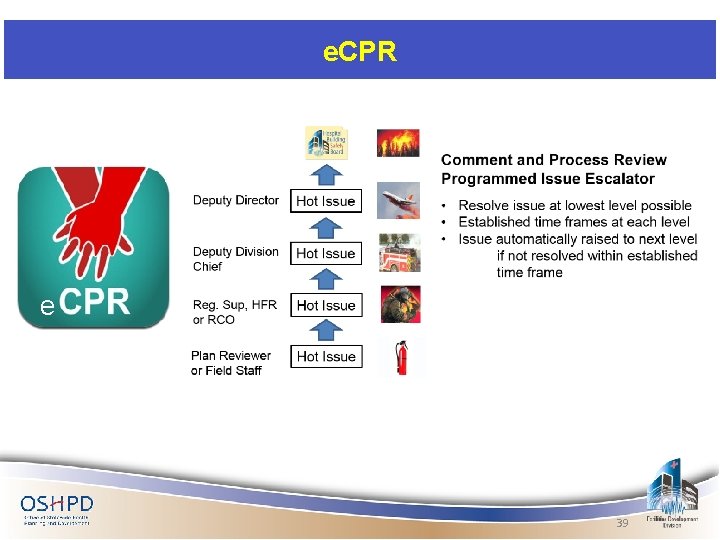
e. CPR Auto Codes e 39

e. CPR A new record type is being added to the various amendments that may be made to a project (such as ACD, DSI, BP, etc. ). The new record type will be formal appeals to OSHPD using the Comment and Process Review currently codified in CAC Section 7 -161. 40

e. CPR Any person associated with the project will be able to create the e. CPR. Upon successful completion of the application, the CPR will be automatically routed to the correct reviewer or field staff. The time limitations specified in CAC 7 -161 will be tracked; if the first level reviewer does not reply within 10 calendar days, the CPR is automatically escalated to the Supervisor or RCO. 41

e. CPR Acceptance or denial of the appeal is documented with an email and a formal letter which will include the reason for any denial. If the appellant wishes to escalate to the next level, a hyperlink is provided in the letter which will be automatically addressed to the appropriate next-level individual. 42

Facilities Development Division California’s Building Department for Hospitals Upcoming Educational Opportunities

Webinars New Pharmacy Standards/Regulations • CHA Sponsored, including Board of Pharmacy, CDPH, and FDD 2016 California Building Standards Code • In conjunction with the HBSB – 1: 30 – 3: 30 pm • • • Fire/life Safety – May 1 Administrative Regulations and Architectural, Mechanical and Electrical – May 15 Structural – May 22 44

AB 1732 - Chapter 818, Statutes of 2016

Signage - OSHPD FREER Manual * *Revised 02/01/2017

DSA Bulletin - All Gender Toilet Facilities Requirements (AB 1732) https: //www. documents. dgs. ca. gov/dsa/bulletins/BU_17 -01. pdf

Advisory Guide for Sterile Compounding Purpose: The California State Board of Pharmacy (Bo. P) has changed its regulations to ensure they reflect changes in current law, as a result of SB 294. Bo. P regulations are now aligned with compounding standards of the United States Pharmacopeia USP <797> Pharmaceutical Compounding – Sterile Preparations, and USP <800> Hazardous Drugs – Handling in Healthcare Settings. Specifically, the Bo. P has recently revised Title 16: – § 1735 “Compounding in Licensed Pharmacies” – § 1751 “Sterile Compounding” California State Board of Pharmacy regulations became enforceable as of January 1, 2017, whereas USP <800> regulations will become effective on July 1, 2018.

Advisory Guide for Sterile Compounding Joint effort of: • California Department of Public Health (CDPH) • California State Board of Pharmacy (Bo. P) • Office of Statewide Health Planning and Development (OSHPD) Issuance Date – April 25, 2017 Contents: • Purpose & History • Submittal Requirements • Definitions • Architectural, Mechanical & Electrical Requirements • Code References 49

Advisory Guide for Sterile Compounding Environments: Non-Hazardous Sterile Compounding environment requirements create an appropriate environment for mixing injectable solutions that present no hazard to the compounding technician/pharmacy staff. Hazardous Sterile Compounding environment requirements create an appropriate sterile environment for mixing injectable solutions that do present a health hazard to the compounding technician/pharmacy staff, and must also limit outside environmental exposure to adjoining rooms and all ventilation discharge locations. Chemotherapy medications, for example, would be compounded in this environment. Essential Design Elements: Primary Engineering Control (PEC) – The compounding workstation hood. (Typically ISO Class 5) Secondary Engineering Control (SEC) – The cleanroom/buffer room where the hood is located. (Typically ISO Class 7) Ante Room – The room leading to the cleanroom/buffer room. (Typically ISO Class 7 or 8)

Advisory Guide for Sterile Compounding PEC SEC Miscellaneous Equipment Miscellaneous Ante Room Ante Scrub Sink Air Pressure Relationships Sample Diagram

Facilities Development Division California’s Building Department for Hospitals Other Stuff

HBSB Stuff NOTICE OF PUBLIC MEETING HOSPITAL BUILDING SAFETY BOARD Administrative Processes, Code Changes, and Standard Details Committee and Energy Conservation and Management Committee JOINT MEETING

HBSB Stuff

FDD’s Top 3 Objectives for 2017 1. Expand Electronic Plan Review 2. Reevaluate FDD’s Plan Review Performance Goals 3. Develop an IOR Trainee/Apprentice Program 55

In Case You Haven’t Heard FDD’s Projected Move Dates = June 23 - 25

57
- Slides: 57