Fabrication of Porous Anodic Alumina Templates with Sub20

Fabrication of Porous Anodic Alumina Templates with Sub-20 nm Pores Shaud Tavakoli Sands Research Group Advisor: Manuel Da. Silva

Background o Oxide film can be grown on certain metals via anodization o Aluminum, niobium, tantalum, titanium, tungsten, zirconium o Aluminum and titanium unique – thick oxide coating with high density of tiny pores possible o Other metals – only see formation of barrier oxide o Anodized alumina referred to as Porous Anodic Alumina (PAA)

Properties of PAA o o Electrically insulating Optically transparent over wide energy band Chemical and thermal stability Factor of 2 volume expansion from aluminum to alumina o Alumina often thinner than original Al due to chemical dissolution of alumina during anodization o Pore diameter 4 -250 nm o Density of pores ranging from 108 to 1012 pores/cm 2 o Thickness up to 300µm o Brittle/fragile

Applications of PAA o Electronic and optoelectronic devices o Magnetic storage o Chemical sensors o Biochemical membranes o Carbon nanotubes o Catalysts o Metallic/semiconducting nanowires and nanorods
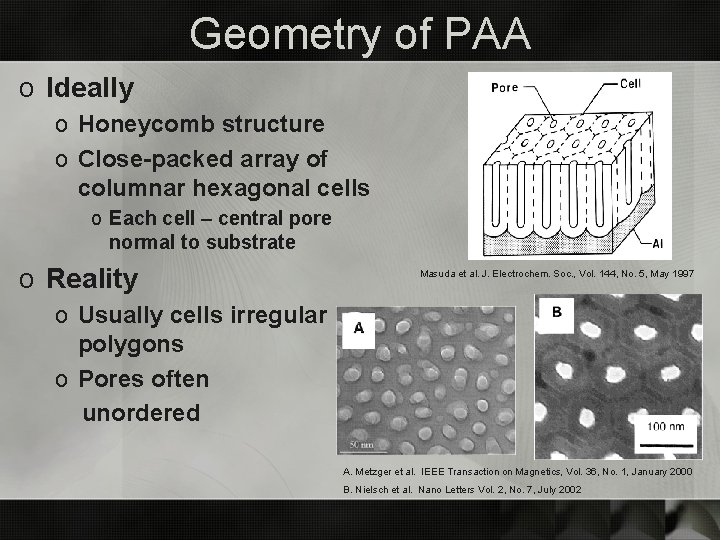
Geometry of PAA o Ideally o Honeycomb structure o Close-packed array of columnar hexagonal cells o Each cell – central pore normal to substrate o Reality Masuda et al. J. Electrochem. Soc. , Vol. 144, No. 5, May 1997 o Usually cells irregular polygons o Pores often unordered A. Metzger et al. IEEE Transaction on Magnetics, Vol. 36, No. 1, January 2000 B. Nielsch et al. Nano Letters Vol. 2, No. 7, July 2002

Relevant Reactions o Overall anodization reaction: 2 Al + 3 H 2 O Al 2 O 3 + 3 H 2 o Sum of reactions at each electrode o Metal/oxide interface: 2 Al + 3 O 2 - Al 2 O 3 + 6 eo Oxygen atoms react with metal o Oxide/electrolyte interface: Al 3+ + 3 H 2 O Al 2 O 3 + 6 H+ o Aluminum anions react with water o Reaction at cathode: 6 H+ + 6 e- 3 H 2 o Hydrogen gas evolution

Our Procedure o Electropolish sample o Removes thin native oxide o Eliminates roughness o Provides a shiny surface finish o Two-step anodization o o Anodize once Strip alumina Anodize second time Pore order develops during 1 st anodization! o Characterize sample using field emission scanning electron microscopy Yuan et al. Chem. Mater. 2004, 16, 1841 -1844

Experiments o Adjusting conditions: o acid concentration o anodization time o temperature o voltage o Using different electrolytes/voltages for 1 st and 2 nd anodization o Oxalic for 1 st anodization o Sulfuric for 2 nd anodization o Three-step anodization o Pore shrinking
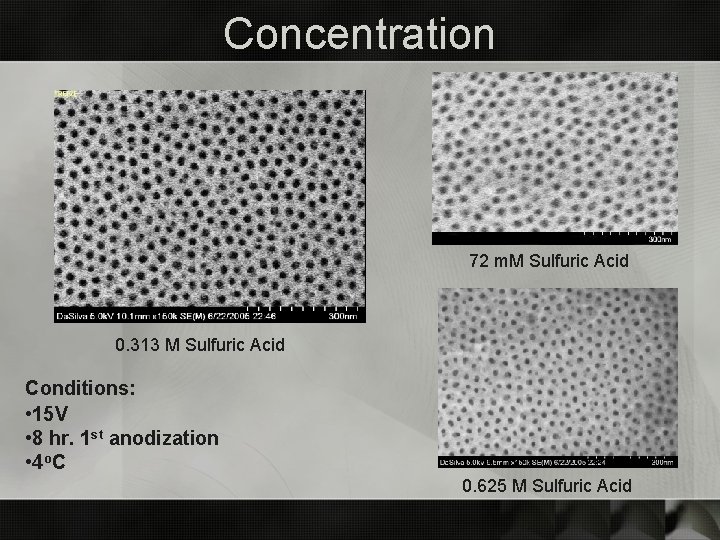
Concentration 72 m. M Sulfuric Acid 0. 313 M Sulfuric Acid Conditions: • 15 V • 8 hr. 1 st anodization • 4 o C 0. 625 M Sulfuric Acid

Anodization Time Anodized at 4 o C and 15 V in 72 m. M sulfuric acid. • 1 st Anodization: 8 h • 2 nd Anodization: 22 h • Avg. pore diameter: ~20 nm Anodized at 4 o C and 15 V in 72 m. M sulfuric acid. • 1 st Anodization: 20 h • 2 nd Anodization: 50 h • Avg. pore diameter ~20. 5 nm • Maybe result of etching

Voltage 15 V Sulfuric Acid Anodized for 8 and 22 hrs. at 4 o C in 72 m. M sulfuric acid. 20 V Sulfuric Acid Anodized for 8 and 22 hrs. at 4 o C in 72 m. M sulfuric acid. o 15 V sample avg. pore diameter ~20 nm o 20 V sample larger pores than 15 V

Two Solution Phosphoric 104 V/ Sulfuric 10 V • ~280 nm cell size • 100+ pores/cell Anodized for 5 hrs. at 4 o C in 1 M phosphoric acid, then 19 hrs. at 4 o C in 0. 313 M sulfuric acid. Sulfuric 25 V/ Sulfuric 10 V • ~60 nm cell size • 4 -5 pores/cell Anodized for 8 hrs. at 4 o C in 0. 313 M sulfuric acid, then 21 hrs. at 4 o C in 0. 313 M sulfuric acid.

Three-Step Anodization Oxalic 40 V/Sulfuric 10 V o Improved cell order with three-step o Cell order slightly decreased during 3 rd anodization All samples anodized at 4 o C. Oxalic acid concentrations: 0. 3 M; Sulfuric acid concentrations: 0. 313 M. Oxalic 40 V/Sulfuric 10 V Oxalic 40 V/ Sulfuric 10 V/ Sulfuric 10 V
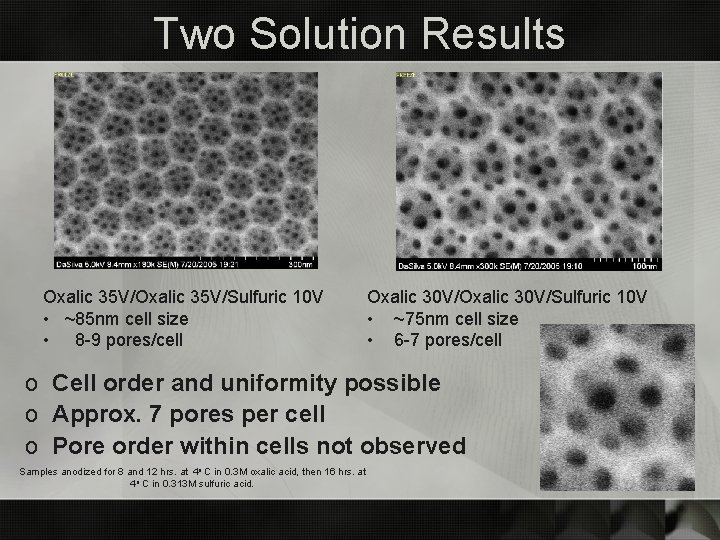
Two Solution Results Oxalic 35 V/Sulfuric 10 V • ~85 nm cell size • 8 -9 pores/cell Oxalic 30 V/Sulfuric 10 V • ~75 nm cell size • 6 -7 pores/cell o Cell order and uniformity possible o Approx. 7 pores per cell o Pore order within cells not observed Samples anodized for 8 and 12 hrs. at 4 o C in 0. 3 M oxalic acid, then 16 hrs. at 4 o C in 0. 313 M sulfuric acid.

Pore Shrinking o Put sample in boiling water to convert alumina to aluminum oxy-hydroxide (Al(O)OH) o Optimize pore order o May develop irregular pores Myung et al. Nanotechnology 15 (2004) 833 -838

Pore Shrinking Results o Conditions: o 4 o. C, 40 V o 0. 3 M oxalic acid o Anodized 9 hr. ; 12 hr. o 1 min. boil o Avg. pore diameter 40 nm o 20% reduction at surface 1 min. o Longer heat treatment o Samples ruined 5, 10, 20 min.
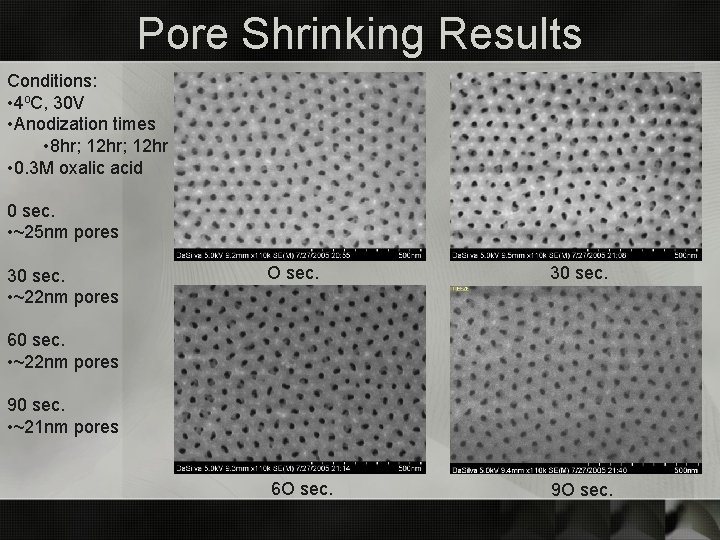
Pore Shrinking Results Conditions: • 4 o. C, 30 V • Anodization times • 8 hr; 12 hr • 0. 3 M oxalic acid 0 sec. • ~25 nm pores 30 sec. • ~22 nm pores O sec. 30 sec. 6 O sec. 9 O sec. 60 sec. • ~22 nm pores 90 sec. • ~21 nm pores

THE END.
- Slides: 18