Fabien ZOULIM How to use virological tools for

Fabien ZOULIM

How to use virological tools for the optimal management of chronic hepatitis B Fabien Zoulim INSERM U 871 & Liver Department Lyon, France

Pathobiology and Natural History of the Disease
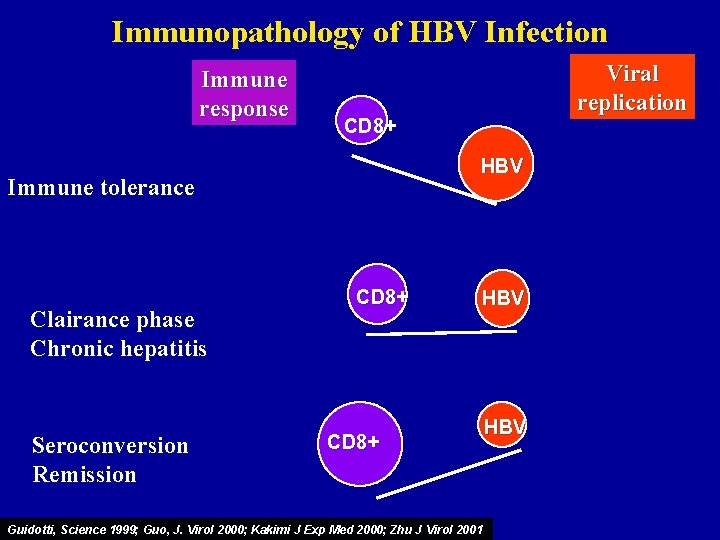
Immunopathology of HBV Infection Immune response CD 8+ HBV Immune tolerance Clairance phase Chronic hepatitis Seroconversion Remission Viral replication CD 8+ HBV CD 8+ Guidotti, Science 1999; Guo, J. Virol 2000; Kakimi J Exp Med 2000; Zhu J Virol 2001 HBV

Phases of the disease • Immunotolerance phase - High viral load and normal ALT levels • Immunoactive phase / chronic hepatitis - Viral replication and elevation of ALT levels • Inactive carrier state - Low viral load and normal ALT levels • Reactivation - Wild type virus or pre-core mutant • Resolved Infection - Clearance of HBs. Ag Fattovich, J Hepatol 2003

Natural history of hepatitis B Acute infection Recovery Chronic infection Lee, N Engl J Med 1997 Lok, Hepatology 2001 Ganem, NEJM 2004 Chronic hepatitis Immune tolerance Wild type virus (HBe. Ag+) Pre-core mutant (HBe. Ag-) Inactive carrier Reactivation Cirrhosis 30 -50 years Hepatocellular carcinoma
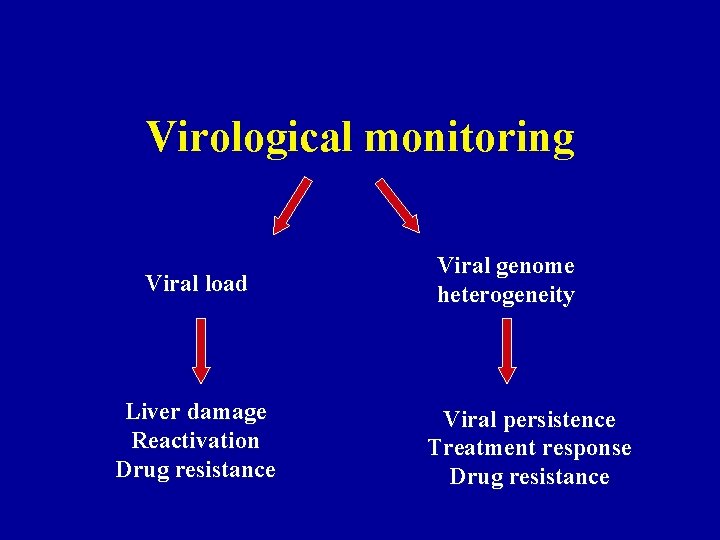
Virological monitoring Viral load Liver damage Reactivation Drug resistance Viral genome heterogeneity Viral persistence Treatment response Drug resistance

Monitoring of Viral Load
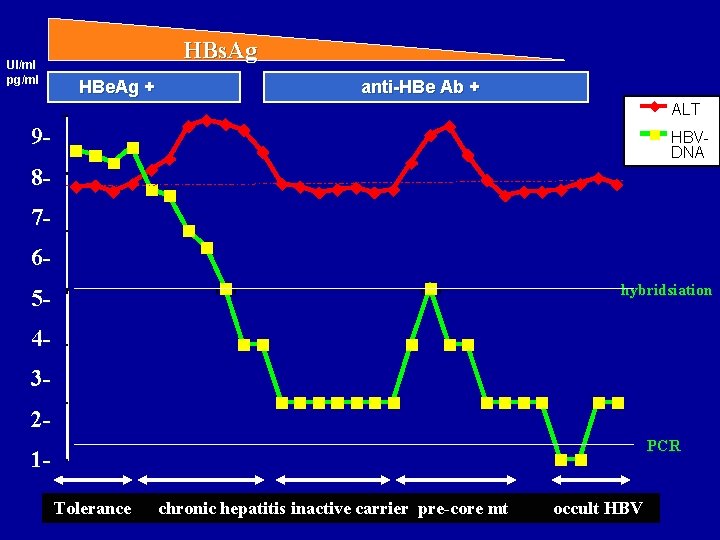
HBs. Ag UI/ml pg/ml HBe. Ag + anti-HBe Ab + ALT 1000 9 - HBVDNA 100 8 - 7 - 10 6 - 5 -1 hybridsiation 40, 1 30, 01 2 - PCR 10, 001 Tolerance chronic hepatitis inactive carrier pre-core mt occult HBV

) ) 0) 3 8 7) 1 ( 6 ( 1 ( ( s g + r g A g ie A r S A e e ar B B HB C H H t. c a In (copies/cell) Median Total HBV DNA ccc. DNA (copies/cell) Evolution of Intrahepatic ccc. DNA During the Natural History 0) ) ) 1 7) 3 8 ( ( 6 1 ( - ( rs g + e g A g ri A S r A e e a B B B H C H H t. c a In Werle et al, Gastroenterology 2004

Serum Viral Load in Chronic Hepatitis Titre vs histology in HBe. Ag-negative patients Serum titre Histology (inflammation) < 104 31/37 had HAI < 3 > 2 105 15/22 had HAI > 4 > 107 5/6 had HAI > 7 Lindh et al J Viral Hepatitis 2000; 7: 258 -67.

Pre-core mutants

HBe. Ag and Precore Mutation ATG 1762 -1764 1814 Basic Core Promoter ATG 1896 1901 Precore region Core region HBc. Ag HBe. Ag Virion Serum


Outcome of Chronic HBe. Ag Negative Hepatitis B Biochemical patterns in 164 untreated patients after 23 months (range 12 -36) monthly monitoring With flares and normalization 73 pts ( 44. 5% ) Asymptomatic flare-up: 90% of cases Without flares A L T 59 pts ( 36. 0% ) Flare-up yearly frequency: once 57. 1% twice 20% < once 22. 8% With flares but without normalization 0 12 months 32 pts ( 19. 5% ) 24 Brunetto MR et al, J Hepatol 2002

Diagnosis of inactive carrier versus HBe. Ag negative chronic hepatitis • Inactive Carrier – Persistently normal ALT levels – Persistently low levels of serum HBV DNA • Threshold : 103 or 104 copies / m. L ? – Wild type genome; sometimes pre-core mutations – The key : careful monitoring ! • HBe. Ag negative chronic hepatitis – – Fluctuation / exacerbation of ALT Fluctuations of HBV DNA levels usually below 106 copies / m. L – Presence of pre-core / core promoter mutations

HBV genotypes

HBV genotypes • Influence on the type of pre-core or BCP mutation • Impact on the outcome of infection and severity of liver disease (HCC) • Impact on IFN response • No clear impact on response to nucleoside analogs Zhang J Med Virol 1996, Orito Hepatology 2001, Mayerat J Viral Hepat 1999; Wai Hepatology 2002, Jansen Lancet 2005 Pichoud et al, Hepatology 1999; Grandjacques J Hepatol 2000; Si Ahmed et al, Hepatology 2000; Yang et al, Gastroenterology 2004

Viral genotypes and IFN response (HBe. Ag loss) 50 47% % 44% 40 28% 30 25% 20 10 0 A B n=90 n=23 C D n=39 n=103 Jansen et al, Lancet, 2005

Monitoring of Antiviral Therapy

Goals and types of response Virological response -HBV DNA < 104 copies/m. L: decreased liver damage - HBV DNA < 103 copies/m. L: decreased risk of resistance Biochemical response - normalization of ALT levels Histological response - improvement in HAI or Metavir score Combined response / Complete response Timing during therapy Initial response / Maintained response End of treatment response / Sustained reponse Hoofnagle, J Hepatol 2003

Infected liver Blood circulation Viral load NKT CD 8 CD 4 B ccc. DNA Infected hepatocytes

Infected liver Blood circulation Viral load NKT CD 8 Antivirals CD 4 B ccc. DNA Infected hepatocytes Werle et al, Gastroenterology 2004

Reductions in Serum HBV DNA, Total Intrahepatic HBV DNA and ccc. DNA During ADV Therapy Me dian m. L ies/m. L (Log 10 copies/ Log 10 copi es/cell) Werle et al, Gastroenterology 2004 § 48 weeks of ADV resulted in significant reductions in : serum HBV DNA > total intrahepatic HBV DNA > ccc. DNA -> 14 years of therapy to clear completely viral ccc. DNA


Virologic Consequences of Persistent Viremia 1) Infection of new hepatocytes slower kinetics of clearance of infected cells and ccc. DNA 2) Increases the risk of occurrence and selection of HBV mutations responsible for drug resistance 3) On-treatment prediction of HBV drug resistance Le Guerhier et al Antimicrob Agents Chemoter 2000; 44: 111 -122; Delmas et al Antimicrob Agents Chemother 2002; 46: 425 -433; Kock et al Hepatology 2003; 38: 1410 -1418; Richman Hepatology 2000; 32: 866 -867

Viral Load at Week 24 is a Predictor of Resistance at Week 104 of Therapy (Telbivudine vs. Lamivudine trial) HBe. Ag Positive, n=921 Telbivudine HBe. Ag Negative, n=446 Lamivudine Di Bisceglie et al. , Abstract #112, AASLD 2006

Clinical Definition of HBV Resistance to Antivirals • Genotypic Resistance: Detection of mutations in the HBV genome, known to confer resistance, which develop during anti-viral therapy • Virologic Breakthrough: Rebound in serum HBV DNA levels following the development of genotypic resistance • Clinical Breakthrough: Virologic breakthrough with increased ALT levels or worsening histology Laboratory Investigations • Phenotypic Resistance: Decreased susceptibility (in vitro testing) to inhibition by anti-viral drugs associated with genotypic resistance. • Cross Resistance: Mutants selected by one agent that also confer resistance to other antiviral agents Zoulim et al; Future Virology 2006

HBV drug resistance mutations Terminal protein 1 Spacer II(F) RNase. H 349 (rt 1) 183 I(G) Pol/RT A LAM / FTC ADV ETV Ld. T TDF 692 (rt 344) GVGLSPFLLA YMDD B C D 845 a. a. E V 173 L L 180 M A 181 V M 204 I/V N 236 T I 233 V I 169 T T 184 G S 202 G/I M 250 V M 204 I A 194 T ? * All ETV resistance requires background YMDD mutations Allen et al. Hepatology 1998; 27: 1670– 7; Gish et al. J Hepatol 2005; 43: 60– 6; Qi et al. J Hepatol 2004; 40(Suppl 1): 20– 1; Tenney et al. AAC 2004; 48: 3498– 507; Lai et al. Gastroenterology 2005; 129: 528– 36; Sheldon et al. Antivir Ther 2005; 10: 727– 34; Delaney et al. AAC 2006 ; Schildgen et al NEJM 2006; Villet et al J Hepatol 2007

HBV DNA (10 E+6 genome eq/ml) ALT(U/L) Line Probe Assay Versus Sequencing for the Detection of HBV Drug Resistance 180 100 140 80 100 60 60 40 20 1 100 200 300 400 595 Day 1 39 290 400 595 Codon 528 Li. PA Seq L L L/M M M Codon 552 Li. PA Seq M M M/V M V V Codon 555 Li. PA Seq V V V V V 0 Sequencing of PCR products C T C M T G G C T Can detect any new mutation T A T G G A T Line probe assay Very sensitive (minor species and low viremia) Nafa et al Hepatology 2000; Lok et al. J Clin Microbiol. 2002

HBV DNA Quantification Dynamic Range of HBV DNA Detection log copies/ml Graph adapted from J. Hepatol. , 39, S 3 -S 25, 2003


Strategies for Monitoring Treatment Response and Detecting HBV Drug Resistance Viraemia levels and ALT every 3 months - Antiviral response and potency - Persisting viraemia - Early detection of drug resistance Serologic assays - HBe. Ag/Anti-HBe. Ab: every 6 months in HBe. Ag+ patients - HBs. Ag/Anti-HBs. Ab: when HBV DNA < limit of detection Genotypic assays - In multidrug experienced patients - At the time of virologic breakthrough - When viral load is not suppressed for long period of time

Approaches to Management Depend on Cross-Resistance Data Resistance mutations

Conclusions Requirement for the most sensitive / quantitative assays • Management of chronic HBV infection – – – Low levels of replication : inactive carriers / occult infection Early detection / prediction of reactivation Treatment eligibility • Monitoring of antiviral therapy – – Early virological response Viral breakthrough / drug resistance Genotypic assays Individualized treatment adaptation for 2 nd or 3 rd line treatment to avoid multidrug resistance


% of patients with resistance mutations Incidence of Resistance in Nucleoside Naive Patients Lai et al CID 2003; Hadzyiannis et al Gastroenterology 2006; Colonno et al AASLD 2006; Di Bisceglie et al AASLD 2006

% of patients with resistance mutations Incidence of Resistance in Lamivudine Refractory Patients Lampertico et al AASLD 2006; Colonno et al AASLD 2006

Management of HBV drug resistance Adefovir switch ~20% resistance/2 year 16% resistance/3 years Adefovir add-on 0% resistance at 3 years Lamivudine 70% resistance at 5 years Entecavir Switch Lamivudine add-on Adefovir Entecavir add-on 30% resistance at 5 years 38% resistance at 3 years ? Telbivudine add-on Adefovir add-on Entecavir resistance at 5 years ? Telbivudine Tenofovir add-on* ? Adefovir add-on resistance at 5 years ? * Not yet approved for HBV therapy Tenofovir add-on* ?

Mechanisms of HBV Drug Resistance Virus Hepatocytes Viral polymerase spontaneous error rate ccc. DNA Long half-life Infected cells Long half-life Defective immune response Impairment of innate response Viral quasi-species Host Viral persistence Selection of escape mutants Selective pressure Antivirals or others Immune response Drug PK Replication fitness Replication space Treatment failure Zoulim Antivir Res 2004; 64: 1– 15

The Hepadnavirus Genome and its Variability « a » determinant vaccine/HBIg 8 genotypes A to H RT domain antivirals core mt CTL response pre-core mt anti-e response ?
- Slides: 41