f MRI Compatible Mechatronic Ankle Device Presented by

f. MRI Compatible Mechatronic Ankle Device Presented by: Danielle Doane, Ben Foss-Michaelis, Brendon Reedy, Karina Snow, and Brandon Teller Advisors: Professor Constantinos Mavroidis Ph. D Azadeh Khanicheh Ph. D Sponsor: NU Robotics Lab
Project Statement n Develop a novel device that measures force and position in a functional magnetic resonance imaging (f. MRI) environment in order to analyze the cortical response to dorsiflexion and plantarflexion ankle movements www. mritoday. net

Project Need n Lack of technology preventing research q f. MRI ankle studies exist, but no device is currently available n Benefits of device q Allows for novel approaches in rehabilitation research q Correlates data and cortical response q Standardizes study and test conditions
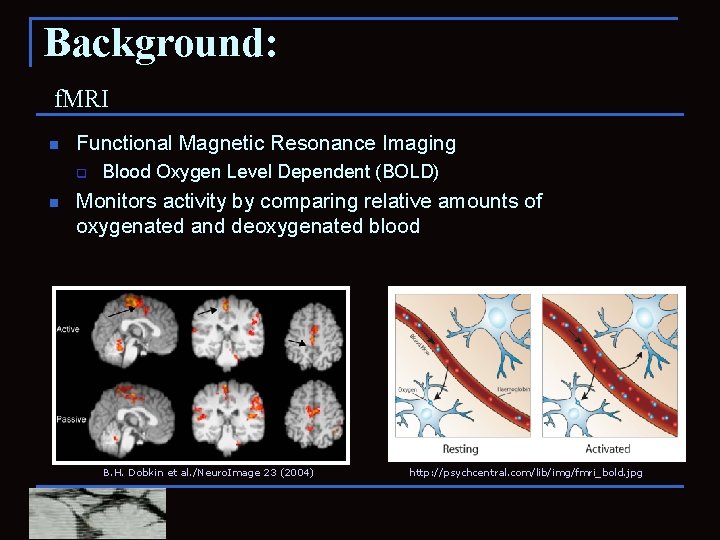
Background: f. MRI n Functional Magnetic Resonance Imaging q n Blood Oxygen Level Dependent (BOLD) Monitors activity by comparing relative amounts of oxygenated and deoxygenated blood B. H. Dobkin et al. /Neuro. Image 23 (2004) http: //psychcentral. com/lib/img/fmri_bold. jpg
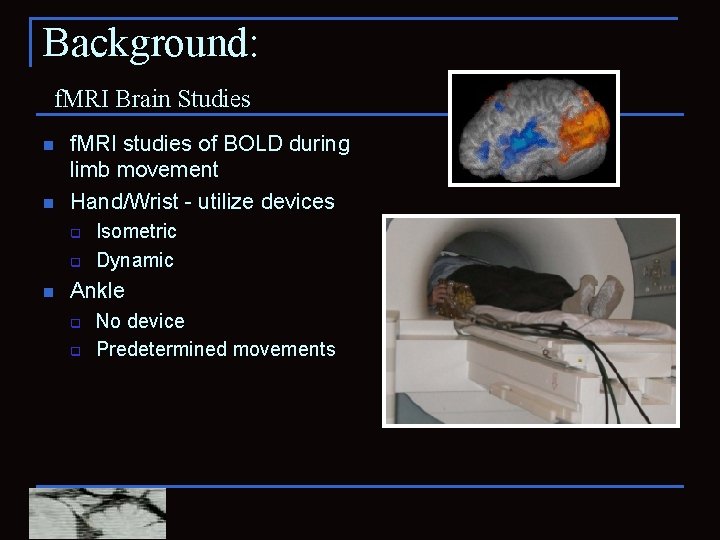
Background: f. MRI Brain Studies n n f. MRI studies of BOLD during limb movement Hand/Wrist - utilize devices q q n Isometric Dynamic Ankle q q No device Predetermined movements

Design Specifications n n MRI Compatible Size- 11 in wide by 26 in long Weight- 25 lbs Functions q q n Controlled Dorsiflexion and Plantarflexion Free Dynamic, Isometric force sensing Ranges of Motion and Force q q Dorsiflexion- 0 -10°, 26 lb Plantarflexion- 0 -40°, 100 lb n ROM Increments- 5° Dynamic Ranges of Speeds or Frequency- up to 25°/s n Expert Interviews: n n Paul Canavan, Northeastern University, Ph. D, PT, ATC, CSCS Paolo Bonato, Spaulding Rehabilitation, Ph. D Joel Stein, Spaulding Rehabilitation, MD

Concept Selection Criteria n f. MRI Compatibility of Materials (25%) n Ability to Perform Desired Exercises (20%) n Minimal Head Migration (15%) n Resistance in Plantarflexion (10%) n Modulated Design/Ease of Assembly (10%) n Foot and Lower Leg Restraints (10%) n Overall Size/Weight of Device (10%)
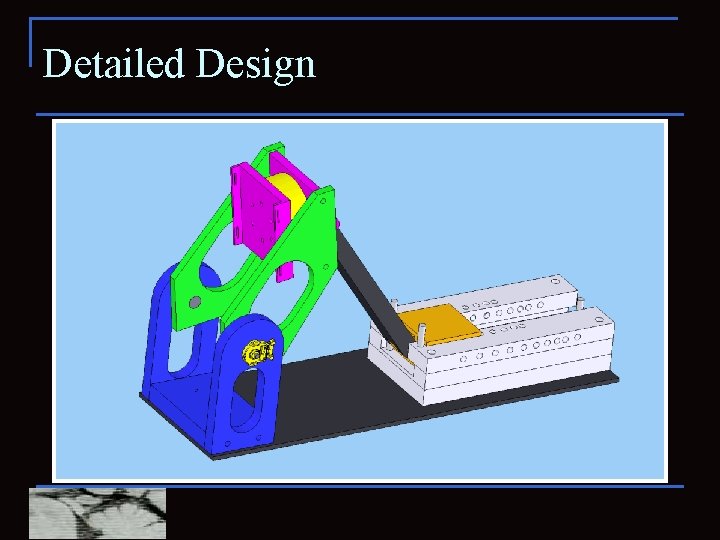
Detailed Design

Detailed Design: Material Selection/Manufacturing Plan n Delrin® q q q n Material Strength Easily Machined Low Coefficient of Friction n Sensors q Aluminum with brass bolts Manufacturing Plan All components machined at Northeastern q Sensors custom made for f. MRI compatibility q wwww. renco. com wwww. jr 3. com

Detailed Design: Component Analysis n n Analyzed all components for maximum force application of 100 lbs q Cosmos. Xpress q Deflection and maximum stress Critical Points q q Foot Pedal/Base Deflection n Max Deflection=. 001” Component Joints - Pins n Failure analysis
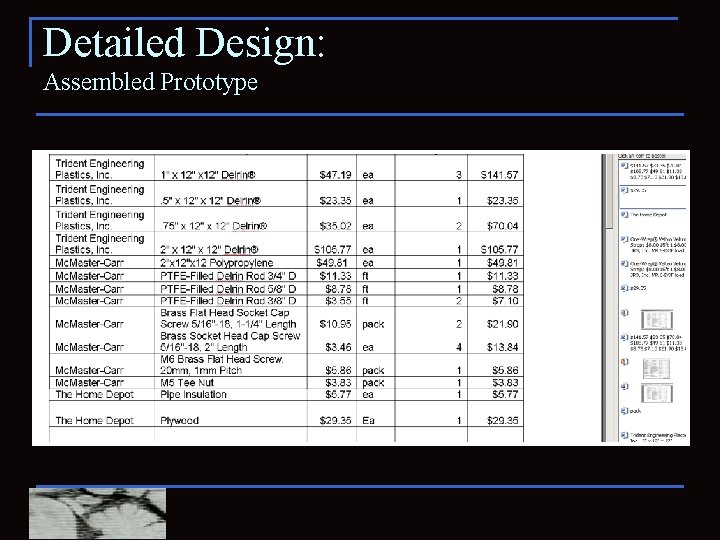
Detailed Design: Assembled Prototype
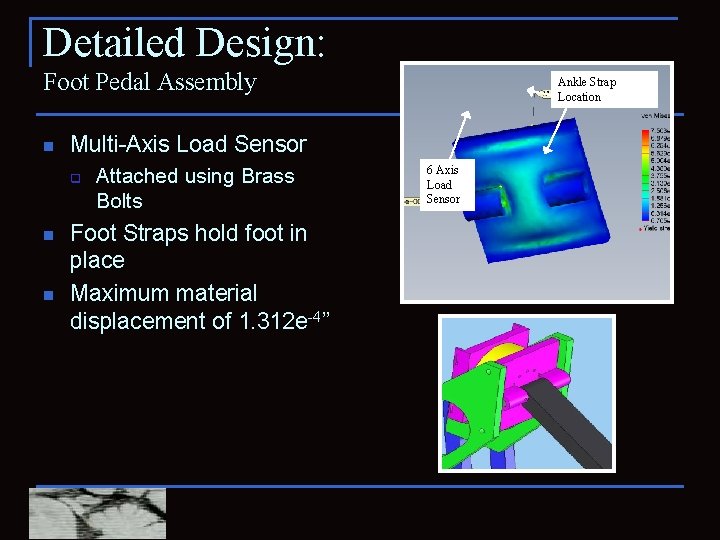
Detailed Design: Foot Pedal Assembly n Multi-Axis Load Sensor q n n Ankle Strap Location Attached using Brass Bolts Foot Straps hold foot in place Maximum material displacement of 1. 312 e-4” 6 Axis Load Sensor

Detailed Design: Slider/Track Assembly n Dynamic Pins q Limit range of motion q Incremental hole locations on top of base Isometric Pin q (. 375 dia x 7 in) q Lock device for isometric exercise q Incremental hole locations on side of base q Factor of Safety of 4. 315 q n (3 Pins. 375 dia X 3 in)

Visual Feedback System: Isometric and Dynamic n Lab. View Interface q q Promotes normalized test execution Patient Interaction n Movement dictation n Performance feedback n Conduct a variety of exercise programs
Device Function

Testing n System Performance Verification q q q n Visual Feedback Ease of use Data output f. MRI Compatibility q All components previously validated for use in f. MRI

Conclusions n Conclusions q q q Achieved project goal of designing and prototyping a mechatronic ankle device for use in f. MRI Device unsuccessfully measures isometric force in the plantarflexion and dorsiflexion direction Problems arise at the sensor-pedal interface n n q The hypothesis is misalignment and the large area of force application generate significant torques These torques are compromising the data Potential solutions include: n n Redesigning the pedal to reduce area of force application Applying a strain gauge to the Dogbone as the means of force measurement

Future Work q Potential solutions include: n n q Redesigning the pedal to reduce area of force application Applying a strain gauge to the Dogbone as the means of force measurement Pedal Redesign Solutions:

Questions? ? ? n Acknowledgements: q q www. dkimages. com Professor Mavroidis, Northeastern University Azadeh Khanicheh, Northeastern University Professor Canavan, Northeastern University Paolo Bonato, Spaulding Rehabilitation

Cost Analysis: Cost for one Ankle Device

Cost Analysis: Total Project Cost

Future Work: Testing Plan n Testing Outside f. MRI q Performance Verification n Isometric- Sensor Readings Dynamic- Different Speeds Testing Inside f. MRI (Phantom Testing) q Sensor Testing n n q Force Sensor Position Sensor Test Plan n n http: //www. medical. siemens. com/webapp/ No Device in room, All power off Device in room, Power On, Phantom in Place, Device not moving Device in room, Power On, Phantom in Place, Device in motion

Design Specifications: Sensing Components n Force q n Position q q q n Max Force- 100 lbs (will amplify down from 250 lbs) Range- 360° Static Error- <. 02° Resolution- 13 bits Frequency q q Range- 120°/s Resolution- 13 bits
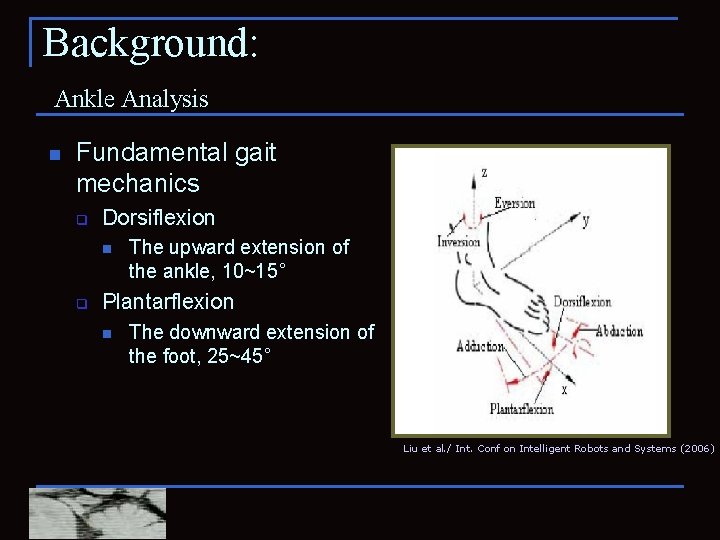
Background: Ankle Analysis n Fundamental gait mechanics q Dorsiflexion n q The upward extension of the ankle, 10~15˚ Plantarflexion n The downward extension of the foot, 25~45˚ Liu et al. / Int. Conf on Intelligent Robots and Systems (2006)
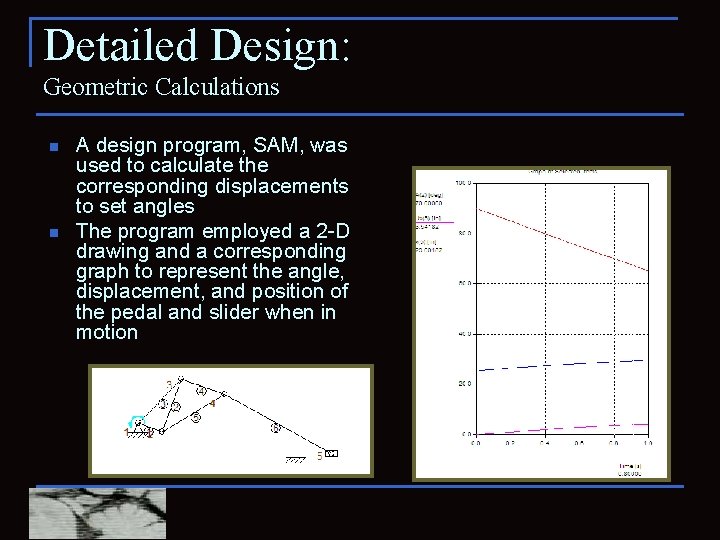
Detailed Design: Geometric Calculations n n A design program, SAM, was used to calculate the corresponding displacements to set angles The program employed a 2 -D drawing and a corresponding graph to represent the angle, displacement, and position of the pedal and slider when in motion
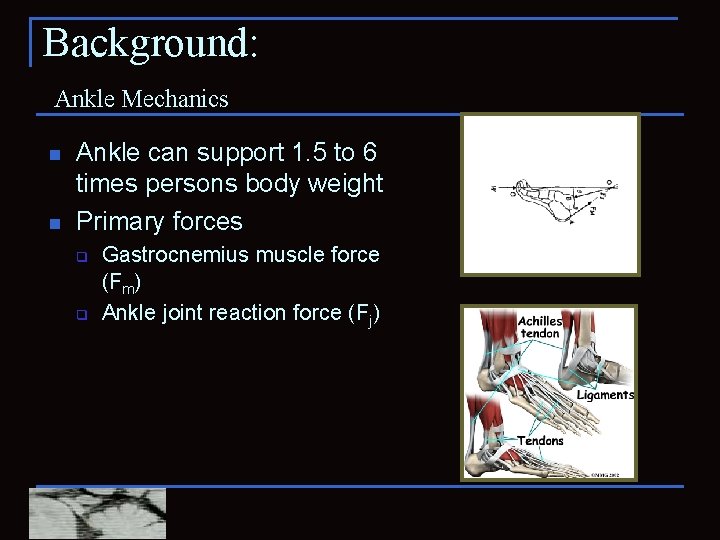
Background: Ankle Mechanics n n Ankle can support 1. 5 to 6 times persons body weight Primary forces q q Gastrocnemius muscle force (Fm) Ankle joint reaction force (Fj)

Background: f. MRI-Compatible Devices and Studies n n Gait Rehabilitation Study Lower Limb Movement Brain Function Studies

Background: f. MRI-Compatible Devices and Studies n n n Isometric Wrist Device Dynamic Hand Device Robotic Arm Device J. Diedrichsen et al. / Neuroscience (2005) J. Hidler et al. /Journal of Neuroscience (2006) Khanicheh et al. / IEEE Int Conf on Rehab Robotics (2005)
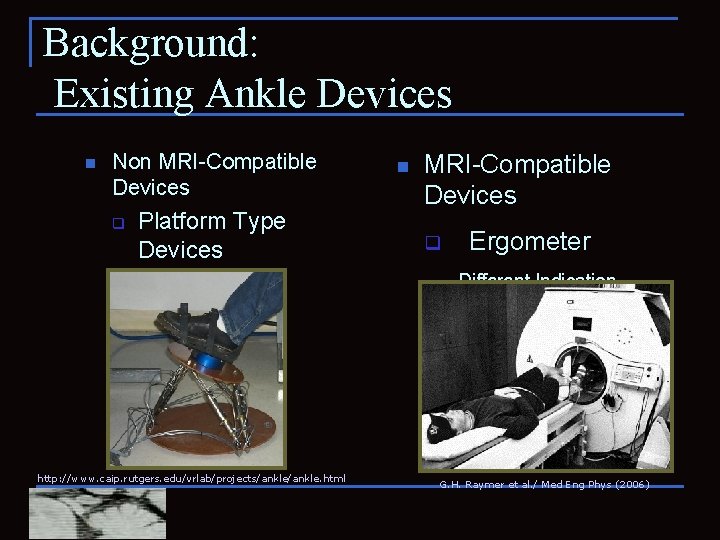
Background: Existing Ankle Devices n Non MRI-Compatible Devices q Platform Type Devices -Rutgers Ankle n MRI-Compatible Devices q Ergometer -Different Indication -Not suitable for f. MRI http: //www. caip. rutgers. edu/vrlab/projects/ankle. html G. H. Raymer et al. / Med Eng Phys (2006)
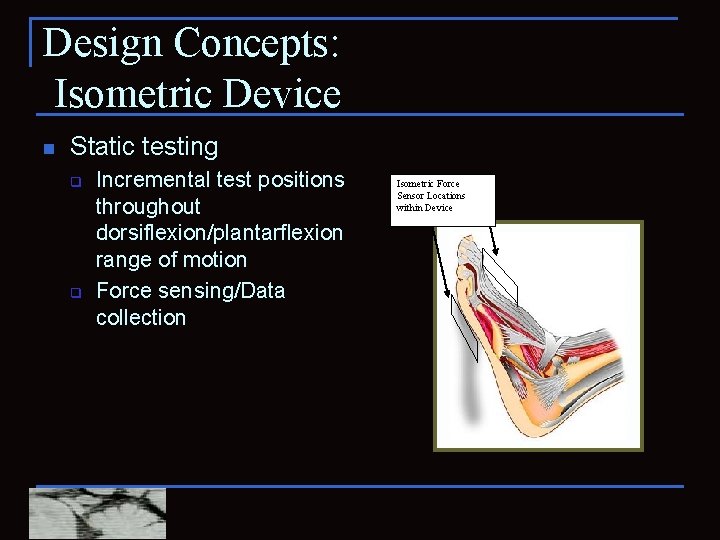
Design Concepts: Isometric Device n Static testing q q Incremental test positions throughout dorsiflexion/plantarflexion range of motion Force sensing/Data collection Isometric Force Sensor Locations within Device
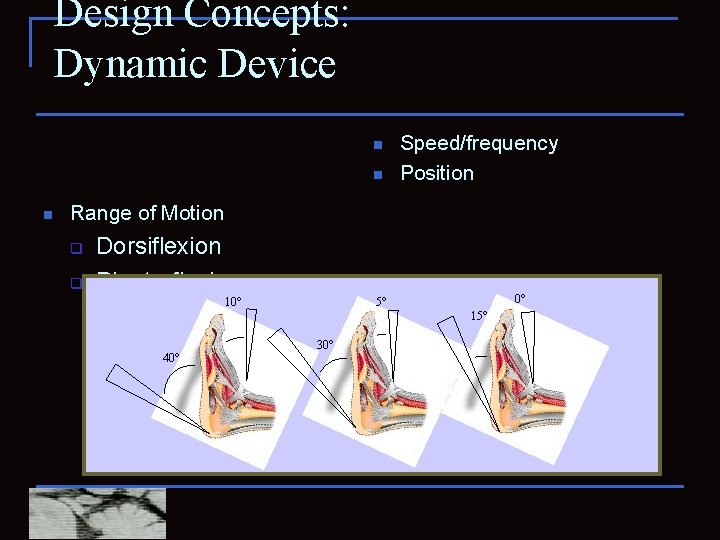
Design Concepts: Dynamic Device n n n Speed/frequency Position Range of Motion q q Dorsiflexion Plantarflexion 10° 0° 5° 15° 30° 40°

Design Concepts: Visual Force Feedback n Used during Isometric and Dynamic exercise q q Promotes normalized test execution Patient Interaction n Movement dictation Performance feedback allows researchers to conduct a variety of exercise programs
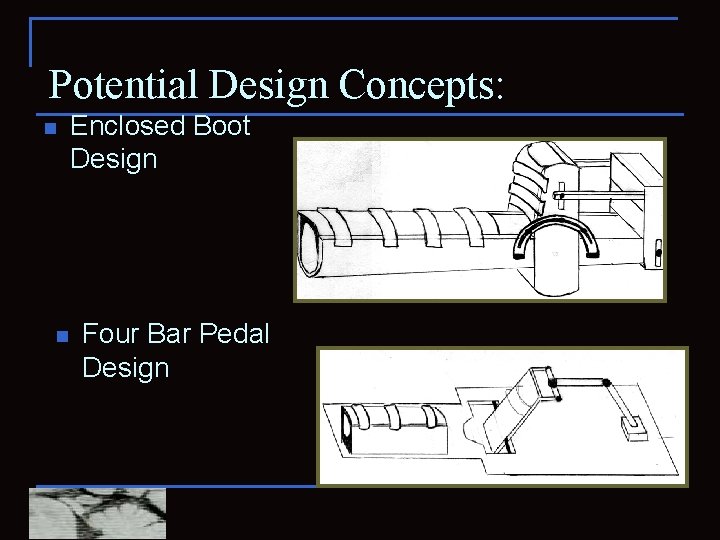
Potential Design Concepts: n n Enclosed Boot Design Four Bar Pedal Design
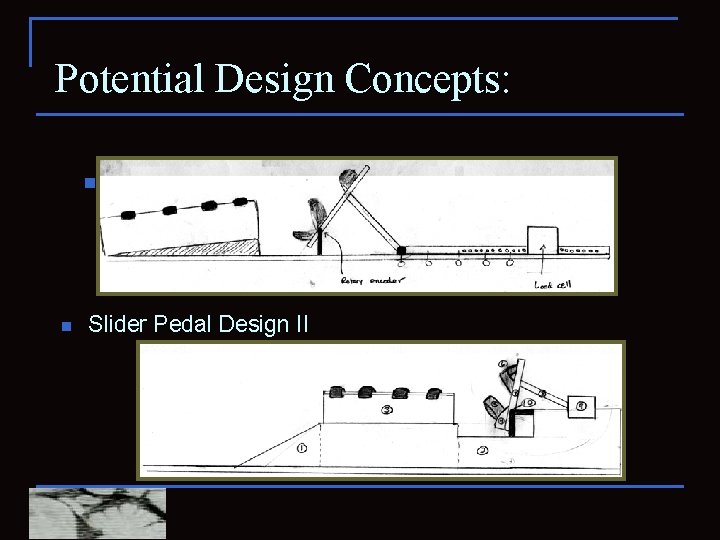
Potential Design Concepts: n n Slider Pedal Design I (Slider Crank) Slider Pedal Design II
- Slides: 34