F I R E study A Multicenter Doubleblind


F. I. R. E. study A Multicenter, Double‑blind, Randomized, Placebo‑Controlled Study to Measure the Effect of FX 06 (a fibrin‑derived peptide Bβ 15‑ 42) on Ischemia‑REperfusion Injury in Patients Undergoing Primary Percutaneous Coronary Intervention Dan Atar MD Professor and Head of Cardiology Aker University Hospital, University of Oslo, Norway

Conflicts-of-Interest DA received honoraria from Fibrex Medical Research & Development Gmb. H provided full financial support for this study

Background Early reperfusion by PCI or thrombolysis is the treatment of choice for STEMI Sudden re-initiation of blood flow achieved with PCI can lead to further endothelial and myocardial damage, termed reperfusion injury Complex underlying cellular and molecular mechanism Numerous pharmacologic strategies have been evaluated for mitigating reperfusion injury, but few have been successful in randomized controlled trials

F. I. R. E. – a Phase II trial of FX 06 in STEMI FX 06: A Novel Compound • Small peptide derived from the human fibrin sequence • Human fibrinopeptide Bß 15 -42, MW 3039 D • Prepared by solid phase peptide synthesis Mode of action • FX 06: peptide that potently inhibits the binding of fibrin E 1 fragment to vascular endothelial (VE) cadherin • Preserves endothelial barrier function, prevents capillary leak • Inhibits transmigration of inflammatory cells through endothelium • Exhibits anti-inflammatory effect F. I. R. E. - Rationale • To investigate the cardioprotective efficacy of FX 06 as an adjunct to reperfusion therapy in patients with acute ST elevation myocardial infarction (STEMI) • To assess safety and tolerability

Study design
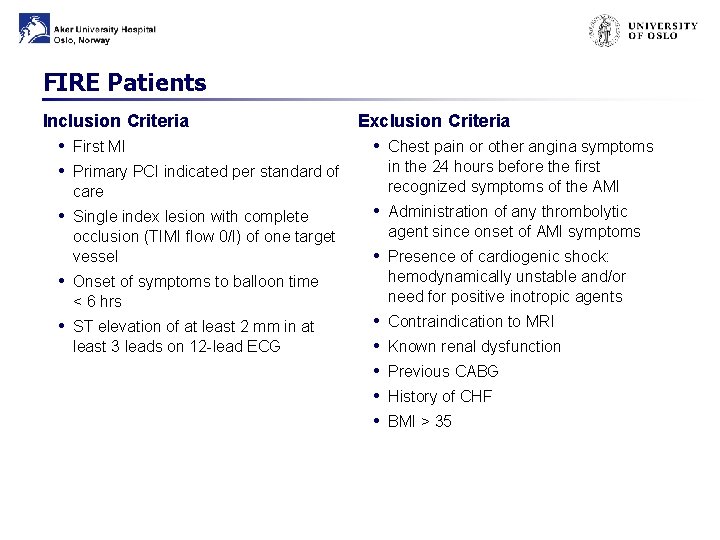
FIRE Patients Inclusion Criteria Exclusion Criteria • First MI • Chest pain or other angina symptoms in the 24 hours before the first • Primary PCI indicated per standard of care • Single index lesion with complete occlusion (TIMI flow 0/I) of one target vessel recognized symptoms of the AMI • Administration of any thrombolytic agent since onset of AMI symptoms • Presence of cardiogenic shock: hemodynamically unstable and/or need for positive inotropic agents • Onset of symptoms to balloon time < 6 hrs • ST elevation of at least 2 mm in at least 3 leads on 12 -lead ECG • • • Contraindication to MRI Known renal dysfunction Previous CABG History of CHF BMI > 35
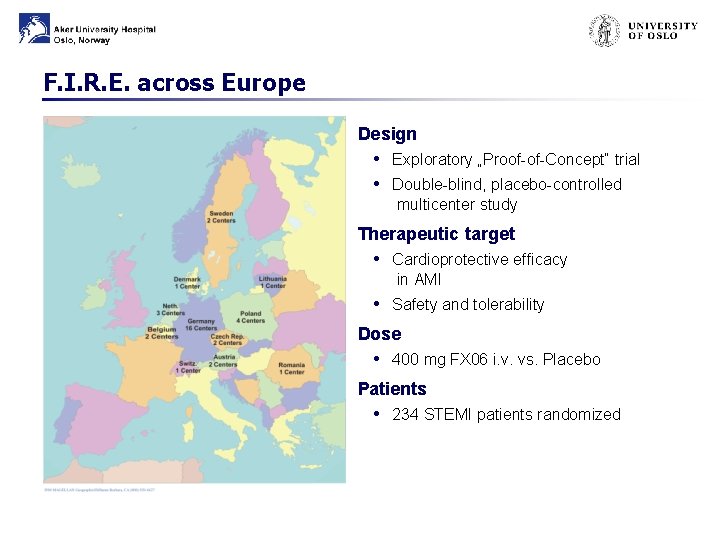
F. I. R. E. across Europe Design • Exploratory „Proof-of-Concept“ trial • Double-blind, placebo-controlled multicenter study Therapeutic target • Cardioprotective efficacy in AMI • Safety and tolerability Dose • 400 mg FX 06 i. v. vs. Placebo Patients • 234 STEMI patients randomized
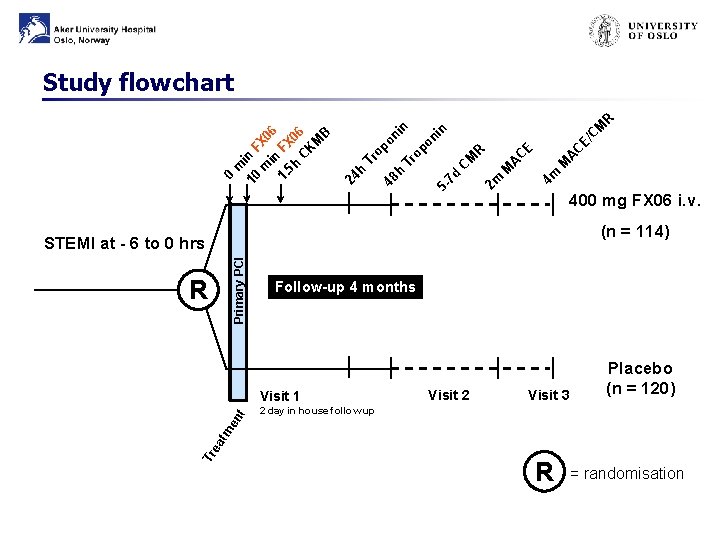
M E/ C E A M 4 m 2 m M A M 5 - 7 d C Tr h C R op on C in in on op 48 Tr h 24 0 m 10 in F m X 0 in 6 1. FX 5 h 0 C 6 K M B R Study flowchart 400 mg FX 06 i. v. (n = 114) Primary PCI STEMI at - 6 to 0 hrs R Follow-up 4 months Visit 2 Visit 3 2 day in house follow up Tr ea tm en t Visit 1 Placebo (n = 120) R = randomisation

Outcome Measures FIRE Primary Outcome Measure • Infarct size measured by C-MRI at 5 days FIRE Secondary Outcome Measures • • • Final infarct size/myocardial scar at 4 months LV function at 5 days and 4 months Troponin I Time to ST segment elevation resolution Combined major adverse cardiac event (MACE): cardiovascular death, myocardial infarction or symptom-driven revascularization plus new symptomatic heart failure (NYHA or Killip Class II or greater) plus rehospitalization for any cardiac cause
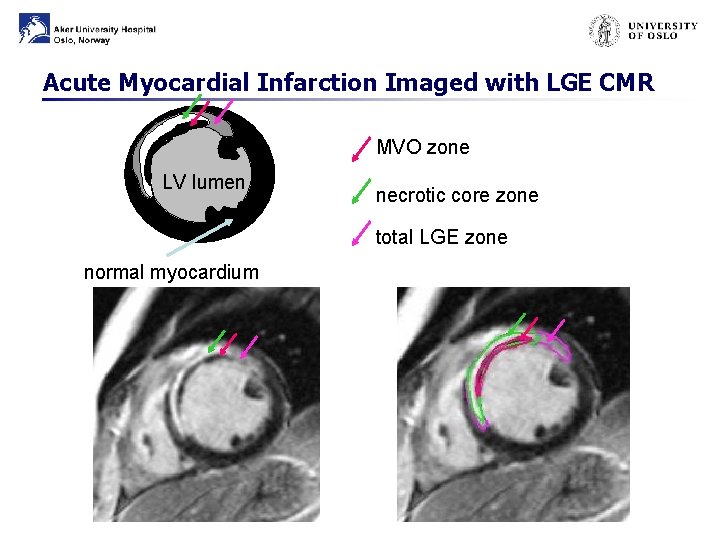
Acute Myocardial Infarction Imaged with LGE CMR MVO zone LV lumen necrotic core zone total LGE zone normal myocardium

F. I. R. E. Baseline Characteristics
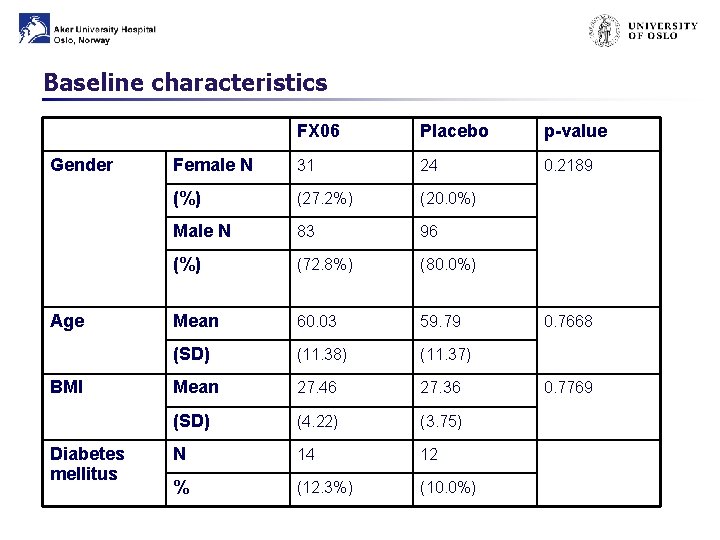
Baseline characteristics Gender Age BMI Diabetes mellitus FX 06 Placebo p-value Female N 31 24 0. 2189 (%) (27. 2%) (20. 0%) Male N 83 96 (%) (72. 8%) (80. 0%) Mean 60. 03 59. 79 (SD) (11. 38) (11. 37) Mean 27. 46 27. 36 (SD) (4. 22) (3. 75) N 14 12 % (12. 3%) (10. 0%) 0. 7668 0. 7769
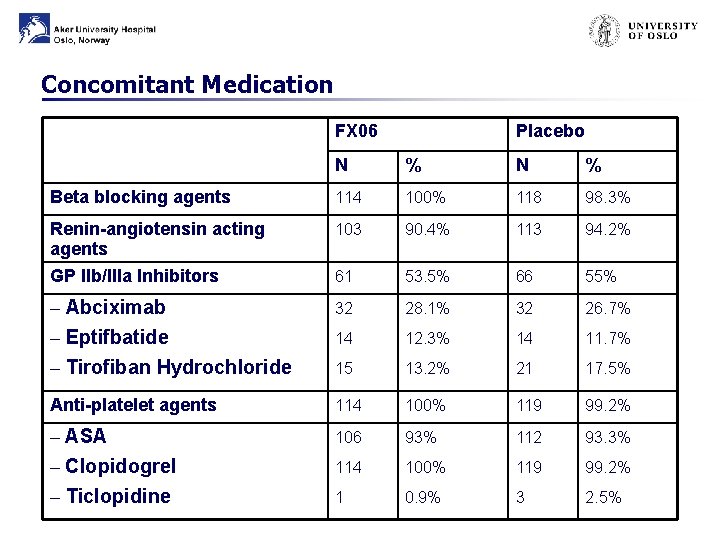
Concomitant Medication FX 06 Placebo N % Beta blocking agents 114 100% 118 98. 3% Renin-angiotensin acting agents 103 90. 4% 113 94. 2% GP IIb/IIIa Inhibitors 61 53. 5% 66 55% – Abciximab – Eptifbatide – Tirofiban Hydrochloride 32 28. 1% 32 26. 7% 14 12. 3% 14 11. 7% 15 13. 2% 21 17. 5% Anti-platelet agents 114 100% 119 99. 2% – ASA – Clopidogrel – Ticlopidine 106 93% 112 93. 3% 114 100% 119 99. 2% 1 0. 9% 3 2. 5%
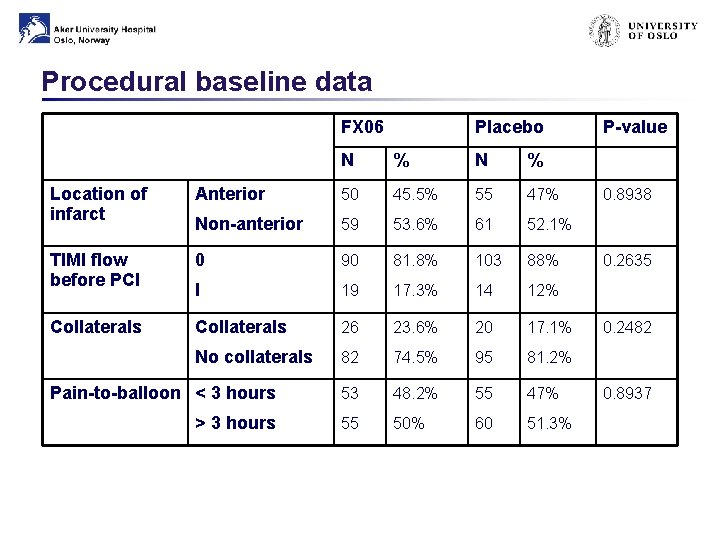
Procedural baseline data FX 06 Placebo N % Location of infarct Anterior 50 45. 5% 55 47% Non-anterior 59 53. 6% 61 52. 1% TIMI flow before PCI 0 90 81. 8% 103 88% I 19 17. 3% 14 12% Collaterals 26 23. 6% 20 17. 1% No collaterals 82 74. 5% 95 81. 2% Pain-to-balloon < 3 hours 53 48. 2% 55 47% > 3 hours 55 50% 60 51. 3% P-value 0. 8938 0. 2635 0. 2482 0. 8937

Results: Infarct size and Clinical Events
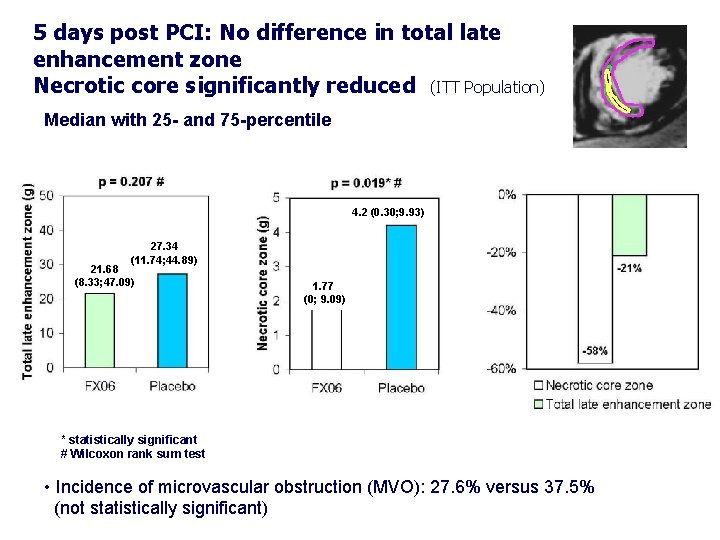
5 days post PCI: No difference in total late enhancement zone Necrotic core significantly reduced (ITT Population) Median with 25 - and 75 -percentile 4. 2 (0. 30; 9. 93) 27. 34 (11. 74; 44. 89) 21. 68 (8. 33; 47. 09) 1. 77 (0; 9. 09) * statistically significant # Wilcoxon rank sum test • Incidence of microvascular obstruction (MVO): 27. 6% versus 37. 5% (not statistically significant)
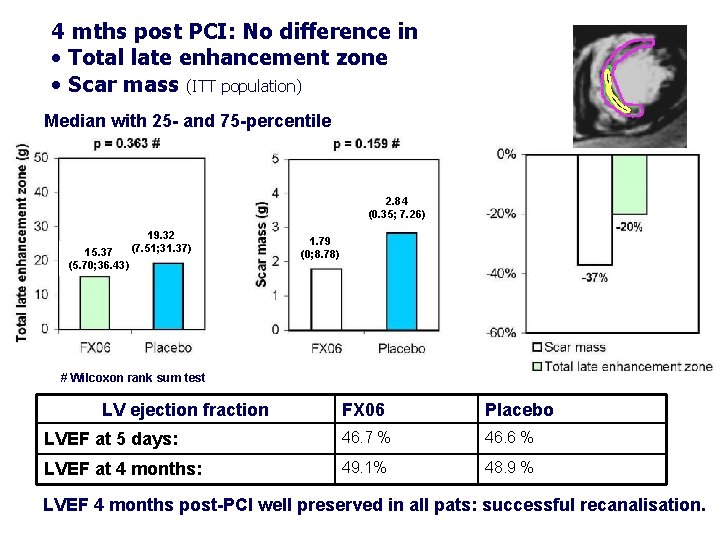
4 mths post PCI: No difference in • Total late enhancement zone • Scar mass (ITT population) Median with 25 - and 75 -percentile 2. 84 (0. 35; 7. 26) 15. 37 (5. 70; 36. 43) 19. 32 (7. 51; 31. 37) 1. 79 (0; 8. 78) # Wilcoxon rank sum test LV ejection fraction FX 06 Placebo LVEF at 5 days: 46. 7 % 46. 6 % LVEF at 4 months: 49. 1% 48. 9 % LVEF 4 months post-PCI well preserved in all pats: successful recanalisation.
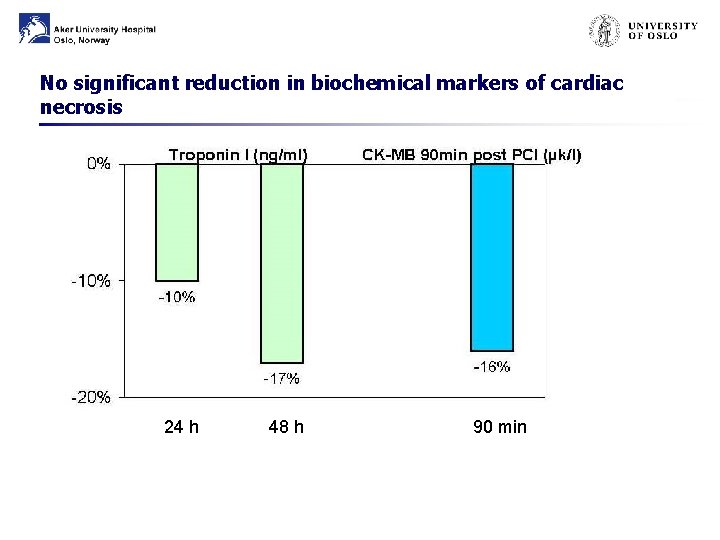
No significant reduction in biochemical markers of cardiac necrosis 24 h 48 h 90 min
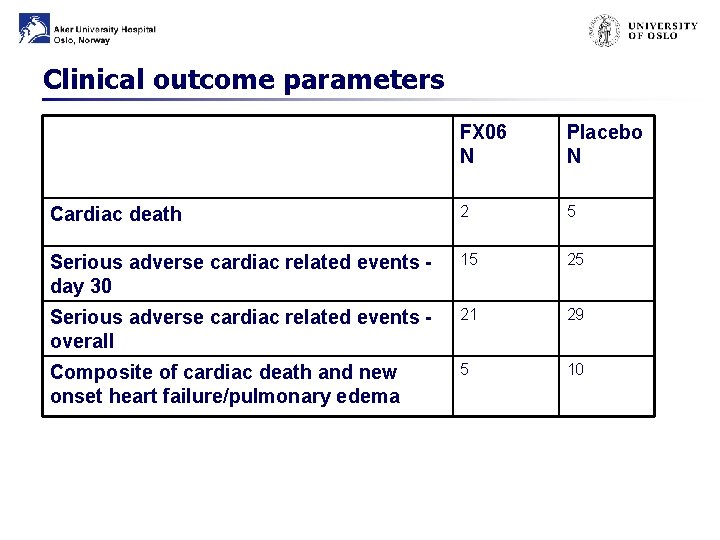
Clinical outcome parameters FX 06 N Placebo N Cardiac death 2 5 Serious adverse cardiac related events day 30 15 25 Serious adverse cardiac related events overall 21 29 Composite of cardiac death and new onset heart failure/pulmonary edema 5 10

Results – Safety
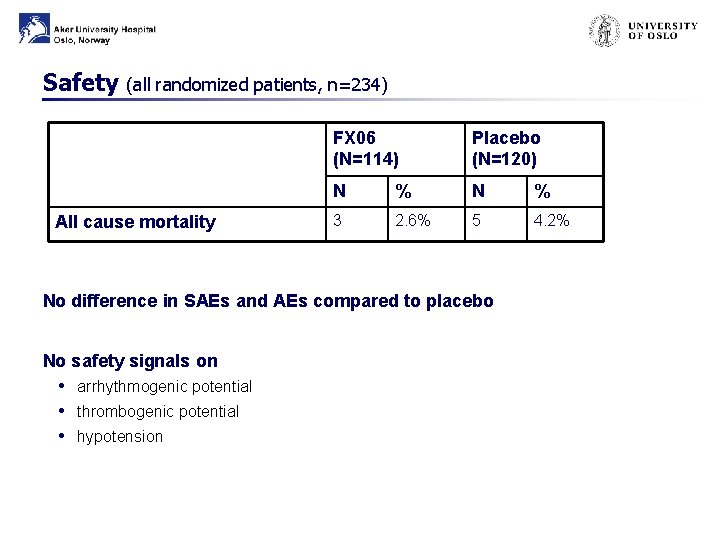
Safety (all randomized patients, n=234) All cause mortality FX 06 (N=114) Placebo (N=120) N % 3 2. 6% 5 4. 2% No difference in SAEs and AEs compared to placebo No safety signals on • arrhythmogenic potential • thrombogenic potential • hypotension

Cardiovascular SAEs (all randomized patients, n=234) Cardiac death Heart failure (non fatal)/Pulmonary oedema VT / VF/ life-threatening Arrhythmia Aute Coronary Syndromes Acute stent thrombosis Atrioventricular block IIIrd degree Revascularization Intraventricular thrombus Hypotension Non cardiac major vascular events TOTAL FX 06 Placebo 2 5 3 5 5 7 3 4 2 2 2 1 1 3 3 1 (acute peripheral ischemia) 21 1 (stroke) 29
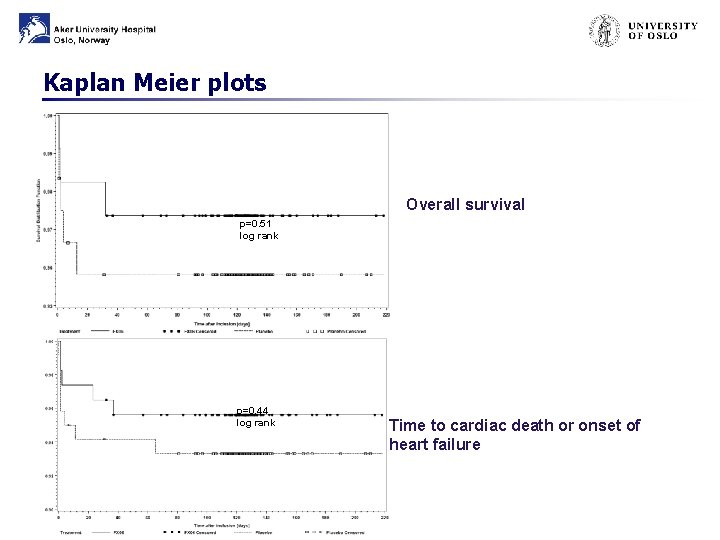
Kaplan Meier plots Overall survival p=0. 51 log rank p=0. 44 log rank Time to cardiac death or onset of heart failure

Conclusion: FX 06 results in the following effects vs. placebo…. . Imaging endpoints and biomarkers: Clinical endpoints: • Primary endpoint: Change in LGE zone not statistically significant • Necrotic core infarct zone at 5 days significantly reduced (-58%, p<0. 025) • Incidence of microvascular obstruction (27. 6% versus 37. 5%) not significantly different • Incidence of cardiac death: 2 vs. 5 • Major cardiovascular adverse events: 21 vs. 29 • Combined incidence of cardiac death and new onset CHF: 5 vs. 10 • Scar mass 4 mths post PCI not significantly different • LV-EF not significantly changed at 4 mths • No significant change in CK-MB at 90 min (-16%) and Troponin I at 24 (-10%) and 48 hrs (-17%) post PCI • This study may suggest a cardioprotective role of FX 06 as an adjunct treatment to PCI • Effect of FX 06 on necrotic infarct zone most likely achieved by amelioration of reperfusion injury • These findings from explorative study may encourage scrutinization in a larger trial

The FIRE team • FIRE Steering Committee: Dan Atar (Oslo, NO), Kurt Huber (Vienna, AU), Jürg Schwitter ( Zurich, CH), Bernard Geudelin (Vienna, AU) • CMR CORE LABORATORY Peter Buser, University Hospital Basel, Switzerland • ECG CORE LABORATORY Peter Clemmensen, Rigshospitalet University of Copenhagen, Denmark • Data Safety Monitoring Board (DSMB): Frans Van de Werf, University Hospital Gasthuisberg, Leuven (Belgium), chairman of DSMB, Lars Wallentin, University Hospital, Uppsala (Sweden), and Günter Breithardt, Universitätsklinikum Münster (Germany) • Investigators: Netherlands – Rensing B, Slagboom T, Van der Ent M; Czech Republic - Stasek J; Poland – Kasprzak JD, Tracz W ; Germany – Butter C, Süselbeck T, Wessely R, Kurz T, Lepper W, Darius H, Kastrati A, Morguet A, Buerke M, Thiele H, Muth G, Bode C ; Sweden – Grip L, Swahn E ; Denmark – Hansen PR ; Belgium – Beauloye C, Schoors D ; Austria – Huber K, Glogar D ; Lithuania – Laucevicius A • Statistician Marcos Marin-Galiano, IFE-Europe, Essen, GE (CRO) Sponsor: Fibrex Medical Research & Development Gmb. H Rainer Henning, Katherine Brandl, Nora Breit

Publication Results of the FIRE-trial: Accepted for publication upon minor revision
- Slides: 27